Abstract
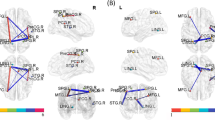
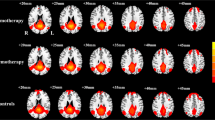
The present study aimed to explore the functional connectivity differences in Resting State Networks (RSNs) induced by cancer and chemotherapy in Lung Cancer (LC) patients using an Independent Component Analysis (ICA). Three matched groups of 15 LC patients following Chemotherapy (C+), 15 LC patients before Chemotherapy (C-) and 15 Healthy Controls (HC) were included. Analysis was performed using ICA and a multivariate pattern analysis (MVPA) to classify groups based on profiles of functional connectivity. We found significant differences in four of the RSN identified: Default Mode Network (DMN), Predominantly Left and Right Anterior Temporal Network, and Cerebellum Network. Whereas DMN showed decreased connectivity, the other RSNs exhibited increased connectivity in both LC groups compared to HC and in C+ in comparison to C-. MVPA discriminated significantly and accurately between all groups. Our study showed that disrupted functional connectivity associated with cancer and chemotherapy-induced cognitive deficits is not only related to DMN decreased connectivity abnormalities but also to an increased connectivity of other RSNs, suggesting a potential compensatory mechanism.

Graphical abstract




Similar content being viewed by others
References
Abou-Elseoud, A., et al. (2010). The effect of model order selection in group PICA. Human Brain Mapping, 31(8), 1207–1216. doi:10.1002/hbm.20929.
Ahles, T. A., & Saykin, A. J. (2002). Breast cancer chemotherapy-related cognitive dysfunction. Clinical Breast Cancer, 3(Suppl 3), S84–S90.
Ahles, T. A., & Saykin, A. J. (2007). Candidate mechanisms for chemotherapy-induced cognitive changes. Nature Reviews. Cancer, 7(3), 192–201. doi:10.1038/nrc2073.
Ahles, T. A., et al. (2002). Neuropsychologic impact of standard-dose systemic chemotherapy in long-term survivors of breast cancer and lymphoma. Journal of Clinical Oncology, 20(2), 485–493.
Altman, D. G. (1999). Practical statistics for medical research. New York: Chapman & Hall/CRC Press.
Ashburner, J. (2007). A fast diffeomorphic image registration algorithm. NeuroImage, 38(1), 95–113. doi:10.1016/j.neuroimage.2007.07.007.
Beckmann, C. F., et al. (2005). Investigations into resting-state connectivity using independent component analysis. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 360(1457), 1001–1013. doi:10.1098/rstb.2005.1634.
Bell, A. J., & Sejnowski, T. J. (1995). An information-maximization approach to blind separation and blind deconvolution. Neural Computation, 7(6), 1129–1159.
Bruno, J., et al. (2012). Altered resting state functional brain network topology in chemotherapy-treated breast cancer survivors. Neurobiology of Disease, 48(3), 329–338. doi:10.1016/j.nbd.2012.07.009.
Buckner, R. L., et al. (2008). The brain's default network: Anatomy, function, and relevance to disease. Annals of the New York Academy of Sciences, 1124, 1–38. doi:10.1196/annals.1440.011.
Calhoun, V. D., et al. (2001). A method for making group inferences from functional MRI data using independent component analysis. Human Brain Mapping, 14(3), 140–151.
Challis, E., et al. (2015). Gaussian process classification of Alzheimer's disease and mild cognitive impairment from resting-state fMRI. NeuroImage, 112, 232–243. doi:10.1016/j.neuroimage.2015.02.037.
Church, J. A., et al. (2009). Control networks in paediatric Tourette syndrome show immature and anomalous patterns of functional connectivity. Brain, 132(Pt 1), 225–238. doi:10.1093/brain/awn223.
Cohen, J. (1960). A coefficient of agreement for nominal scales. Educational and Psychological Measurement, 20(1), 37–46.
Correa, D. D., et al. (2013). A prospective evaluation of changes in brain structure and cognitive functions in adult stem cell transplant recipients. Brain Imaging and Behavior. doi:10.1007/s11682-013-9221-8.
Damoiseaux, J. S. (2012). Resting-state fMRI as a biomarker for Alzheimer's disease? Alzheimer's Research & Therapy, 4(2), 8. doi:10.1186/alzrt106.
Damoiseaux, J. S., et al. (2006). Consistent resting-state networks across healthy subjects. Proceedings of the National Academy of Sciences of the United States of America, 103(37), 13848–13853. doi:10.1073/pnas.0601417103.
De Luca, M., et al. (2006). fMRI resting state networks define distinct modes of long-distance interactions in the human brain. NeuroImage, 29(4), 1359–1367. doi:10.1016/j.neuroimage.2005.08.035.
Dickerson, B. C., et al. (2004). Medial temporal lobe function and structure in mild cognitive impairment. Annals of Neurology, 56(1), 27–35. doi:10.1002/ana.20163.
Dietrich, J., et al. (2006). CNS progenitor cells and oligodendrocytes are targets of chemotherapeutic agents in vitro and in vivo. Journal of Biology, 5(7), 22. doi:10.1186/jbiol50.
Dumas, J. A., et al. (2013). Chemotherapy altered brain functional connectivity in women with breast cancer: A pilot study. Brain Imaging and Behavior, 7(4), 524–532. doi:10.1007/s11682-013-9244-1.
Duzel, E., et al. (2009). Functional imaging of the human dopaminergic midbrain. Trends in Neurosciences, 32(6), 321–328. doi:10.1016/j.tins.2009.02.005.
Eichenbaum, H., et al. (2007). The medial temporal lobe and recognition memory. Annual Review of Neuroscience, 30, 123–152. doi:10.1146/annurev.neuro.30.051606.094328.
Fawcett, T. (2006). An introduction to ROC analysis. Pattern Recognition Letters, 27(8), 861–874.
Franco, A. R., et al. (2009). Interrater and intermethod reliability of default mode network selection. Human Brain Mapping, 30(7), 2293–2303. doi:10.1002/hbm.20668.
Gour, N., et al. (2011). Basal functional connectivity within the anterior temporal network is associated with performance on declarative memory tasks. NeuroImage, 58(2), 687–697. doi:10.1016/j.neuroimage.2011.05.090.
Gour, N., et al. (2014). Functional connectivity changes differ in early and late-onset Alzheimer's disease. Hum Brain Mapp, 35(7), 2978–2994. doi: 10.1002/hbm.22379.
Grosshans, D. R., et al. (2008). Neurocognitive function in patients with small cell lung cancer : Effect of prophylactic cranial irradiation. Cancer, 112(3), 589–595. doi:10.1002/cncr.23222.
Hampson, J. P., et al. (2015). Altered resting brain connectivity in persistent cancer related fatigue. Neuroimage Clinic, 8, 305–313. doi:10.1016/j.nicl.2015.04.022.
Hillary, F. G., et al. (2015). Hyperconnectivity is a fundamental response to neurological disruption. Neuropsychology, 29(1), 59–75. doi: 10.1037/neu0000110.
Himberg, J., et al. (2004). Validating the independent components of neuroimaging time series via clustering and visualization. NeuroImage, 22(3), 1214–1222. doi:10.1016/j.neuroimage.2004.03.027.
Horky, L. L., et al. (2014). Systemic chemotherapy decreases brain glucose metabolism. Annals of Clinical Translational Neurology, 1(10), 788–798. doi:10.1002/acn3.121.
Jim, H. S., et al. (2012). Meta-analysis of cognitive functioning in breast cancer survivors previously treated with standard-dose chemotherapy. Journal of Clinical Oncology, 30(29), 3578–3587. doi:10.1200/JCO.2011.39.5640.
Kaasa, S., et al. (1988a). Neuropsychological evaluation of patients with inoperable non-small cell lung cancer treated with combination chemotherapy or radiotherapy. Acta Oncologica, 27(3), 241–246.
Kaasa, S., et al. (1988b). Reduced short-term neuropsychological performance in patients with nonsmall-cell lung cancer treated with cisplatin and etoposide. Antibiotics and Chemotherapy, 41, 226–231.
Kesler, S. R. (2014). Default mode network as a potential biomarker of chemotherapy-related brain injury. Neurobiology of Aging, 35(Suppl 2), S11–S19. doi:10.1016/j.neurobiolaging.2014.03.036.
Kesler, S. R., et al. (2013). Default mode network connectivity distinguishes chemotherapy-treated breast cancer survivors from controls. Proceedings of the National Academy of Sciences of the United States of America, 110(28), 11600–11605. doi:10.1073/pnas.1214551110.
Kiviniemi, V., et al. (2009). Functional segmentation of the brain cortex using high model order group PICA. Human Brain Mapping, 30(12), 3865–3886. doi:10.1002/hbm.20813.
Komaki, R., et al. (1995). Evaluation of cognitive function in patients with limited small cell lung cancer prior to and shortly following prophylactic cranial irradiation. International Journal of Radiation Oncology, Biology, Physics, 33(1), 179–182. doi:10.1016/0360-3016(95)00026-U.
LaViolette, P., Collier, W., Schmainda, K. M., Piacentine, L., Douville, K. L., Chitambar, C. R., Tran, A., Claesges, S. A., Durgerian, S. J., & Bloom, A.S. (2009). International Society for Magnetic Resonance Medicine (ISMRM), Honolulu, HI, USA. In http://cds.ismrm.org/. Functional Connectivity and Arterial Spin Labeling in Chemotherapy Induced Cognitive Impairment (“Chemobrain”). International Society for Magnetic Resonance Medicine (ISMRM).
Lee, M. H., et al. (2013). Resting-state fMRI: A review of methods and clinical applications. AJNR. American Journal of Neuroradiology, 34(10), 1866–1872. doi:10.3174/ajnr.A3263.
Lynall, M. E., et al. (2010). Functional connectivity and brain networks in schizophrenia. The Journal of Neuroscience, 30(28), 9477–9487. doi:10.1523/JNEUROSCI.0333-10.2010.
Marquand, A., et al. (2010). Quantitative prediction of subjective pain intensity from whole-brain fMRI data using Gaussian processes. NeuroImage, 49(3), 2178–2189. doi:10.1016/j.neuroimage.2009.10.072.
Mattis, S. (1988). Dementia rating scale: Professinal manual. Odessa: Psychological Assessment Resources.
Mourao-Miranda, J., et al. (2005). Classifying brain states and determining the discriminating activation patterns: Support vector machine on functional MRI data. NeuroImage, 28(4), 980–995. doi:10.1016/j.neuroimage.2005.06.070.
O'Reilly, J. X., et al. (2010). Distinct and overlapping functional zones in the cerebellum defined by resting state functional connectivity. Cerebral Cortex, 20(4), 953–965. doi:10.1093/cercor/bhp157.
Piccirillo, J. F., et al. (2015). Cognitive impairment after chemotherapy related to atypical network architecture for executive control. Oncology, 88(6), 360–368. doi:10.1159/000370117.
Raichle, M. E. (2011). The restless brain. Brain Connectivity, 1(1), 3–12. doi:10.1089/brain.2011.0019.
Sanz-Arigita, E. J., et al. (2010). Loss of 'small-world' networks in Alzheimer's disease: Graph analysis of FMRI resting-state functional connectivity. PloS One, 5(11), e13788. doi:10.1371/journal.pone.0013788.
Schrouff, J., et al. (2013). PRoNTo: Pattern recognition for neuroimaging toolbox. Neuroinformatics, 11(3), 319–337. doi:10.1007/s12021-013-9178-1.
Sheline, Y. I., et al. (2010). Amyloid plaques disrupt resting state default mode network connectivity in cognitively normal elderly. Biological Psychiatry, 67(6), 584–587. doi:10.1016/j.biopsych.2009.08.024.
Shilling, V., et al. (2005). The effects of adjuvant chemotherapy on cognition in women with breast cancer--preliminary results of an observational longitudinal study. Breast, 14(2), 142–150. doi:10.1016/j.breast.2004.10.004.
Simo, M., et al. (2013). Chemobrain: A systematic review of structural and functional neuroimaging studies. Neuroscience and Biobehavioral Reviews, 37(8), 1311–1321. doi:10.1016/j.neubiorev.2013.04.015.
Simo, M., et al. (2015). Cognitive and brain structural changes in a lung cancer population. Journal of Thoracic Oncology, 10(1), 38–45. doi:10.1097/JTO.0000000000000345.
Simo, M., et al. (2016a). Longitudinal brain changes associated with prophylactic cranial irradiation in lung cancer. Journal of Thoracic Oncology, 11(4), 475–486. doi:10.1016/j.jtho.2015.12.110.
Simo, M., et al. (2016b). Longitudinal brain changes associated with prophylactic cranial irradiation in lung cancer. Journal of Thoracic Oncology. doi:10.1016/j.jtho.2015.12.110.
Smith, S. M., et al. (2009). Correspondence of the brain's functional architecture during activation and rest. Proceedings of the National Academy of Sciences of the United States of America, 106(31), 13040–13045. doi:10.1073/pnas.0905267106.
Sperling, R. (2007). Functional MRI studies of associative encoding in normal aging, mild cognitive impairment, and Alzheimer's disease. Annals of the New York Academy of Sciences, 1097, 146–155. doi:10.1196/annals.1379.009.
Spreng, R. N., & Schacter, D. L. (2012). Default network modulation and large-scale network interactivity in healthy young and old adults. Cerebral Cortex, 22(11), 2610–2621. doi:10.1093/cercor/bhr339.
Tate, R. F. (1954). Correlation between a discrete and a continuous variable point-biserial correlation. The Annals of Mathematical Statistics, 25(3), 603–607.
Wang, J., et al. (2010). Graph-based network analysis of resting-state functional MRI. Frontiers in Systems Neuroscience, 4, 16. doi:10.3389/fnsys.2010.00016.
Wefel, J. S., et al. (2004). The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma: Results of a prospective, randomized, longitudinal trial. Cancer, 100(11), 2292–2299. doi:10.1002/cncr.20272.
Welzel, T., et al. (2008). Diffusion tensor imaging screening of radiation-induced changes in the white matter after prophylactic cranial irradiation of patients with small cell lung cancer: First results of a prospective study. AJNR. American Journal of Neuroradiology, 29(2), 379–383. doi:10.3174/ajnr.A0797.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by la Fundació Marató-TV3 [Acquired Spinal Cord and Brain Injuries Program (2012–2015) awarded to ARF] and Hospital of Bellvitge Research Award 2016. Marta Simó is a recipient of a Juan Rodés contract from the Carlos III National Health Institute (Spanish Government)- European Social Fund (ESF).
Conflicts of interest
All the signing authors have seen and approved the manuscript content and there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
M. Simó and X. Rifà-Ros contributed equally to this work
J. Bruna and A. Rodríguez-Fornells share co-senior authorship
Electronic supplementary material
Supplementary Figure 1
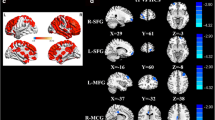
Overlapping functional and structural results between C+ and HC in right temporal regions. Significant differences between groups are reported at a FWE-corrected threshold at voxel level but displayed on a t-map and superimposed on a standardized T1 template using MNI coordinates at an uncorrected p ˂ 0.001 with a cluster extent of 20 voxels. Structural T1-Voxel-Based Morphometry (dark blue color) and Fractional Anisotropy-Voxel-Based Analysis (green color) results (Simo et al. 2015) are overlapped with the present functional connectivity results (red color). Overlap between structural maps occurred in insular regions and overlap between structural and functional occurred in middle temporal gyrus. ILF: inferior longitudinal fasciculus. MTG: middle temporal gyrus. (GIF 89 kb)
Rights and permissions
About this article
Cite this article
Simó, M., Rifà-Ros, X., Vaquero, L. et al. Brain functional connectivity in lung cancer population: an exploratory study. Brain Imaging and Behavior 12, 369–382 (2018). https://doi.org/10.1007/s11682-017-9697-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-017-9697-8