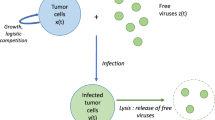
Abstract
We formulate a mathematical model of functional partial differential equations for oncolytic virotherapy which incorporates virus diffusivity, tumor cell diffusion, and the viral lytic cycle based on a basic oncolytic virus dynamics model. We conduct a detailed analysis for the dynamics of the model and carry out numerical simulations to demonstrate our analytic results. Particularly, we establish the positive invariant domain for the \(\omega \) limit set of the system and show that the model has three spatially homogenous equilibriums solutions. We prove that the spatially uniform virus-free steady state is globally asymptotically stable for any viral lytic period delay and diffusion coefficients of tumor cells and viruses when the viral burst size is smaller than a critical value. We obtain the conditions, for example the ratio of virus diffusion coefficient to that of tumor cells is greater than a value and the viral lytic cycle, is greater than a critical value, under which the spatially uniform positive steady state is locally asymptotically stable. We also obtain conditions under which the system undergoes Hopf bifurcations, and stable periodic solutions occur. We point out medical implications of our results which are difficult to obtain from models without combining diffusive properties of viruses and tumor cells with viral lytic cycles.




Similar content being viewed by others
References
Bajzer Ž, Carr T, Josić K et al (2008) Modeling of cancer virotherapy with recombinant measles viruses. J Theor Biol 252(1):109–122
Barish S, Ochs MF, Sontag EO, Gevertz J (2017) Evaluation optimal therapy robustness by virtual expansion of a sample population, with a case study in cancer immunotherapy. In: PNAS E6277–E6286
Boeuf FL, Batenchuk C, Koskela MV, Breton S, Roy D, Lemay C et al (1974) Model-based rational design of an oncolytic virus with improved therapeutic potential. Nat Commun 2013:4
Chiocca EA (2002) Oncolytic viruses. Nat Rev Cancer 2(12):938
Chiocca EA, Rabkin SD (2014) Oncolytic viruses and their application to cancer immunotherapy. Cancer Immunol Res 2(4):295–300
Choudhury B, Nasipuri B (2014) Efficient virotherapy of cancer in the presence of immune response. Int J Dyn Control 2:314–325
Faria T (2000) Normal forms and Hopf bifurcation for partial differential equations with delays. Trans Am Math Soc 352(5):2217–2238
Friedman A, Lai X (2018) Combination therapy for cancer with oncolytic virus and checkpoint inhibitor: a mathematical model. PloS ONE 13(2):e0192449
Friedman A, Tian JP, Fulci G et al (2006) Glioma virotherapy: effects of innate immune suppression and increased viral replication capacity. Cancer Res 66(4):2314–2319
Harpold HL, Alvord EC Jr, Swanson KR (2007) The evolution of mathematical modeling of glioma proliferation and invasion. J Neuropathol Exp Neurol 66(1):1–9
Hassard BD, Kazarinoff ND, Wan YH (1981) Theory and applications of Hopf bifurcation. Cambridge University Press, Cambridge
Jenner AL, Coster ACF, Kim PS, Frascoli F (2018) Treating cancerous cells with viruses: insights from a minimal model for oncolytic virotherapy. Lett Biomath 5:S117–S136. https://doi.org/10.1080/23737867.2018.1440977
Kaplan JM (2005) Adenovirus-based cancer gene therapy. Curr Gene Ther 5(6):595–605
Karev GP, Novozhilov AS, Koonin EV (2006) Mathematical modeling of tumor therapy with oncolytic viruses: effects of parametric heterogeneity on cell dynamics. Biol Direct 1(1):30
Kirn DH, McCormick F (1996) Replicating viruses as selective cancer therapeutics. Mol Med Today 2(12):519–527
Lawler SE, Speranza MC, Cho CF et al (2017) Oncolytic viruses in cancer treatment: a review. JAMA Oncol 3(6):841–849
Lin X, So JWH, Wu J (1992) Centre manifolds for partial differential equations with delays. Proc R Soc Edinb Sect A Math 122(3–4):237–254
Mahasa KJ, Eladdadi A, Pillis L d, Ouifki R (2017) Oncolytic potency and reduced vuris tumor-specificity in oncolytic virotherapy, a mathematical modeling approach. PloS ONE 12(9):e0184347
Maroun J, Muñoz-Alía M, Ammayappan A, Schulze A, Peng KW, Russell S (2017) Designing and building oncolytic viruses. Future Virol 12(4):193–213
Martuza RL, Malick A, Markert JM et al (1991) Experimental therapy of human glioma by means of a genetically engineered virus mutant. Science 252(5007):854–856
Massey SC, Rockne RC, Hawkins-Daarud A, Gallaher J, Anderson ARA, Canoll P, Swanson KR (2018) Simulating PDGF-driven glioma growth and invasion in an anatomically accurate brain domain. Bull Math Biol 80:1292–12309
Mok W, Stylianopoulos T, Boucher Y, Jain RK (2009) Mathematical modeling of herpes simplex virus distribution in solid tumors: implications for cancer gene therapy. Clin Cancer Res 15(7):2352–2360
Novozhilov AS, Berezovskaya FS, Koonin EV et al (2006) Mathematical modeling of tumor therapy with oncolytic viruses: regimes with complete tumor elimination within the framework of deterministic models. Biol Direct 1(1):6
Phan TA, Tian JP (2017) The role of the innate immune system in oncolytic virotherapy. Comput Math Methods Med 6587258
Ratajczyk E, Ledzewicz U, Leszczynski M, Schattler H (2018) Treatment of glioma with virotherapy and TNF-a inhibitors: analysis as a dynamical system. Discrete Contin Dyn Syst B 23(1):425–441
Roberts MS, Lorence RM, Groene WS et al (2006) Naturally oncolytic viruses. Curr Opin Mol Ther 8(4):314–321
Swanson KR, Alvord EC, Murray JD (2002) Virtual brain tumours (gliomas) enhance the reality of medical imaging and highlight inadequacies of current therapy. Br J Cancer 86:14–18
Tian JP (2011) The replicability of oncolytic virus: defining conditions in tumor virotherapy. Math Biosci Eng 8(3):841–860
Timalsina A, Tian JP, Wang J (2017) Mathematical and computational modeling for tumor virotherapy with mediated immunity. Bull Math Biol 79(8):1736–1758
Vasiliu D, Tian JP (2011) Periodic solutions of a model for tumor virotherapy. Discrete Contin Dyn Syst S 4(6):1587–1597
Wang Y, Tian JP, Wei J (2013) Lytic cycle: a defining process in oncolytic virotherapy. Appl Math Model 37(8):5962–5978
Wang Z, Guo Z, Peng H (2017) Dynamical behavior of a new oncolytic virotherapy model based on gene variation. Discrete Contin Dyn Syst S 10(5):1079–1093
Wares J, Crivelli J, Yun CO, Choi IK, Gevertz J, Kim P (2015) Treatment strategies for combining immunostimulatory oncolytic virus therapeutics with dendritic cell injections. Math Biosci Eng 2(6):1237–1256
Wein LM, Wu JT, Kirn DH (2003) Validation and analysis of a mathematical model of a replication-competent oncolytic virus for cancer treatment: implications for virus design and delivery. Cancer Res 63(6):1317–1324
Wodarz D (2001) Viruses as antitumor weapons: defining conditions for tumor remission. Cancer Res 61(8):3501–3507
Wodarz D, Komarova N (2009) Towards predictive computational models of oncolytic virus therapy: basis for experimental validation and model selection. PloS ONE 4(1):e4271
Wodarz D, Hofacre A, Lau JW, Sun Z, Fan H, Komarova N (2012) Complex spatial dynamics of oncolytic viruses in vitro: mathematical and experimental approaches. PloS Comput Biol 8(6):e1002547
Wu J (2012) Theory and applications of partial functional differential equations. Springer, Berlin
Wu JT, Byrne HM, Kirn DH et al (2001) Modeling and analysis of a virus that replicates selectively in tumor cells. Bull Math Biol 63(4):731
Acknowledgements
JZ and JPT would like to acknowledge the support from U54CA132383 of NIH (awarded to JPT), Fundamental Research Funds for the Universities in Heilongjiang Province (No: RCCX201718, awarded to JZ) and Fundamental Research Funds of Education Department of Heilongjiang Province (No: 135109228, awarded to JZ). The authors would like to acknowledge the suggestion of model formulation from Philip K. Maini. The authors would like to thank two anonymous reviewers for their insightful comments and constructive suggestions which greatly help in improving the presentation of our results.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix: Stability and Direction of the Hopf Bifurcations
Appendix: Stability and Direction of the Hopf Bifurcations
Theory of functional reaction–diffusion systems says that a family of spatially homogeneous or inhomogeneous periodic solutions may bifurcate from the positive homogeneous equilibrium state \(E^{*}\) of the system (5) when \(\tau \) crosses through the critical value \(\tau ^{*}\). In Appendix, we investigate the stability and direction of Hopf bifurcations by using the center manifold theorem and the normal formal theory of partial functional differential equation (Faria 2000; Wu 2012). Basically, the system (5) firstly is represented as an abstract ODE system. Secondly, at the center manifold of the ODE system corresponding to \(E^{*}\), the normal form or Taylor expansion of the ODE system is computed. Then, the coefficients of the first four terms of the normal form will reveal all the properties of the periodical solutions (Hassard et al. 1981). At the end, we briefly describe the numerical method we use to solve our system.
Let \(u_1(\cdot ,t)=u(\cdot ,\tau t)-u^{*},~u_2(\cdot ,t)=w(\cdot ,\tau t)-w^{*},~u_3(\cdot ,t)=v(\cdot ,\tau t)-v^{*}\) and \(U(t)=(u_1(\cdot ,t),u_2(\cdot ,t),u_3(\cdot ,t))^T\). Then, the system (5) can be written as an equation in the function space \(\mathscr {C}=C([-1,0],X):\)
where \(D=\text {diag}(d_1,d_1,d_2)\), \(L(\tau )(\cdot ):\mathscr {C}\rightarrow X\) and \(f:\mathscr {C}\times \mathbb {R}\mathscr {c}\rightarrow X\) are given, respectively, by
with
for \(\varphi =(\varphi _1,\varphi _2,\varphi _3)^\text {T} \in \mathscr {C}\).
Let \(\tau =\tau ^{*}+\sigma \), then (19) can be rewritten as
where
for \(\varphi \in \mathscr {C}\).
From the previous subsection, when \(\sigma =0\) (i.e. \(\tau =\tau ^{*}\)) the system (20) undergoes Hopf bifurcation at the equilibrium (0, 0, 0), it is also clear that \(\pm \text {i}\omega ^{*}\tau ^{*}\) are simply purely imaginary eigenvalues of the linearized system of (20) at the origin
with \(\sigma =0\) and all other eigenvalues of (21) at \(\sigma =0\) have negative real parts.
The eigenvalues of \(\tau D \Delta \) on X are \(-\tau d_1\mu _n\) (the number of multiples is two) and \(-\tau d_2\mu _n,~n\in \mathbb {N}_0\), with corresponding eigenfunctions \(\beta _n^1(x)=(\gamma _n(x),0,0)^T, ~\beta _n^2(x)=(0,\gamma _n(x),0)^T\) and \(\beta _n^3(x)=(0,0, \gamma _n(x))^T\), where \(\gamma _n(x)=\frac{\phi _n(x)}{(\int _\omega \phi _n(x)\text {d}x)^{\frac{1}{2}}}.\)
We have that the solution operator of (21) is a \(C_0\) semigroup, and the infinitesimal generator \(A_{\sigma }\) is given by
and the domain \(\text {dom}(A_{\sigma })\) of \(A_{\sigma }\) is
Hence, Eq. (20) can be rewritten as the abstract ODE in \(\mathscr {C}\):
where
We denote
For \(\phi =(\phi ^{^{(1)}},\phi ^{^{(2)}})^{T}\in \mathscr {C}\), we denote
Define \(A_{\sigma , n}\) as
Furthermore, we have
and
where
Define \(\mathscr {C}^{*}=C([0,1];X)\) and a bilinear form \((\cdot ,\cdot )\) on \(\mathscr {C}^{*}\times \mathscr {C}\)
where \((\cdot ,\cdot )_c\) is the bilinear form defined on \(C^{*}\times C\)
and
with
Notice that
we have
We define the adjoint operator \(A^{*}\) of \(A_0\)
Let
be the eigenfunctions of \(A_0,\) and \(A^{*}\) corresponds to \(i\omega ^{*}\tau ^{*}\) and \(-i\omega ^{*}\tau ^{*}\), respectively. By direct calculations, we chose
where
and
Obviously, \((q^{*},q)_{c}=1\). Then, we decompose \(\mathscr {C}\) by
\(\mathscr {C}=P\oplus Q\), where
Thus, system (23) could be rewritten as
where
As in Hassard et al. (1981), we have
Then, it follows that
where
With the center manifold theorem (Lin et al. 1992), there exists a center manifold \(\mathcal {C}_0\) and on \(\mathcal {C}_0\), we have
where z and \(\bar{z}\) are local coordinates for center manifold \(\mathcal {C}_0\) in the direction of \(q\gamma _{n_0}\) and \(\bar{q}\gamma _{n_0}\), respectively. For solution \(U_t\in \mathcal {C}_0\), we denote
and
For convenience, we rewrite (26) as
and denote
From direct calculation, we get
and
Here, \(w_{11}\) and \(w_{20}\) are need to be computed. From (25), we have
where
Obviously,
Comparing the coefficients of (28) with the derived function of (27), we obtain
From (22) and (29), for \(\theta \in [-1,0)\), we have
where \(E_1\) and \(E_2\) are both three-dimensional vectors in X and can be determined by setting \(\theta =0\) in H. In fact, set \(\theta =0\) and by (29) and (30), we obtain
The terms \(\tilde{F}''_{zz}\) and \(\tilde{F}''_{z\bar{z}}\) are elements in the space \(\mathscr {C}\), and
We denote
then from (31) we have
Thus, \(E_1^n\) and \(E_2^n\) could be calculated by
where
Hence, \(g_{21}\) could be represented explicitly.
We denote
Then, by the general Hopf bifurcation theory (see Hassard et al. 1981), we know that \(\mu \) determines the directions of the Hopf bifurcation: If \(\mu >0(<0)\), then the direction of the Hopf bifurcation is forward (backward), that is the bifurcating periodic solutions exist when \(a>0(<0)\); \(\beta _2\) determines the stability of the bifurcating periodic solutions: The bifurcating periodic solutions are orbitally stable(unstable) if \(\beta _2<0(>0)\), and \(T_2\) determines the period of the bifurcation periodic solutions: The period increases(decreases) if \(T_2>0(<0)\).
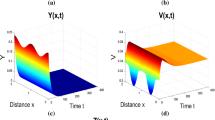
We now describe briefly the numerical method we use to solve the system (5) as follows. For \(\Omega =(0,p\pi )\times (0,q\pi )\), let \(x_i=ih_x, i=1,2,\cdots , l, h_x=\frac{p\pi }{l},y_j=jh_y, j=1,2,\cdots , m, h_y=\frac{q\pi }{m}, ,t_k=kh_t, h_t=\frac{\tau }{N}\) (N is a positive integer), and \(u(i,j,k)=u(x_i,y_j,t_k),v(i,j,k) =v(x_i,y_j,t_k),w(i,j,k)=w(x_i,y_j,t_k)\). We replace \(\frac{\partial u(x_i,y_j,t_k)}{\partial t}\) with a first-order difference \(\frac{u(i,j,k+1)-u(i,j,k)}{h_t}\) and replace \(\frac{\partial ^2 u(x_i,y_j,t_k)}{\partial x^2}+\frac{\partial ^2 u(x_i,y_j,t_k)}{\partial y^2}\) with a second-order difference \(\frac{u(i+1,j,k)-2u(i,j,k)+u(i-1,j,k)}{h_x}+\frac{u(i,j+1,k) -2u(i,j,k)+u(i,j-1,k)}{h_y}\). Similarly, we take differences of the partial derivative of v and w. Then, we get a system of difference equations:
We implement this method in MATLAB and obtain numerical solutions of the system (5).
Rights and permissions
About this article
Cite this article
Zhao, J., Tian, J.P. Spatial Model for Oncolytic Virotherapy with Lytic Cycle Delay. Bull Math Biol 81, 2396–2427 (2019). https://doi.org/10.1007/s11538-019-00611-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-019-00611-2