Abstract
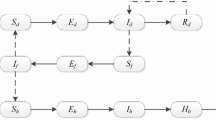
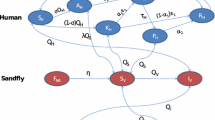
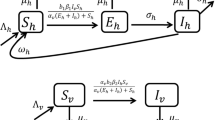
Deterministic (ordinary differential equation) models for the transmission dynamics of vector-borne diseases that incorporate disease-induced death in the host(s) population(s) are generally known to exhibit the phenomenon of backward bifurcation (where a stable disease-free equilibrium of the model coexists with a stable endemic equilibrium when the associated reproduction number of the model is less than unity). Further, it is well known that, in these models, the phenomenon of backward bifurcation does not occur when the disease-induced death rate is negligible (e.g., if the disease-induced death rate is set to zero). In a recent paper on the transmission dynamics of visceral leishmaniasis (a disease vectored by sandflies), titled “A Mathematical Study to Control Visceral Leishmaniasis: An Application to South Sudan,” published in Bulletin of Mathematical Biology, Vol. 79, Pages 1110–1134, 2017, Ghosh et al. (2017) stated that their deterministic model undergoes a backward bifurcation even when the disease-induced mortality in the host population is set to zero. This result is contrary to the well-established theory on the dynamics of vector-borne diseases. In this short note, we illustrate some of the key errors in the Ghosh et al. (2017) study.




Similar content being viewed by others
References
Blayneh K, Gumel AB, Lenhart S, Clayton T (2010) Backward bifurcation analysis and optimal control of West Nile virus. Bull Math Biol 72(4):1006–1028
Bowman C, Gumel AB, van den Driessche P, Wu J, Zhu H (2005) A mathematical model for assessing control strategies against West Nile virus. Bull Math Biol 67:1107–1133
Castillo-Chavez CC, Song B (2004) Dynamical models of tuberculosis and their applications. Math Biosci Energy 1:361–404
Farinaz F, Gumel AB (2013) Analysis of an age-structured model for malaria transmission dynamics. Math Biosci 247:80–94
Feng X, Ruan S, Teng Z, Wang K (2015) Stability and backward bifurcation in a malaria transmission model with applications to the control of malaria in China. Math Biosci 266:52–64
Garba SM, Gumel AB (2010) Effect of cross-immunity on the transmission dynamics of two strains of dengue. Int J Comp Math 87(10):2361–2384
Garba SM, Gumel AB, Abu Bakar MR (2008) Backward bifurcations in dengue transmission dynamics. Math Biosci 215(1):11–25
Ghosh I, Sardar T, Chattopadhyay J (2017) A mathematical study to control visceral leishmaniasis: an application to South Sudan. Bull Math Biol 79:1100–1134
Hussaini N, Lubuma JM-S, Barley K, Gumel AB (2016) Mathematical analysis of a model for AVL-HIV co-endemicity. Math Biosci 271:80–95
Jiang J, Qiu Z, Wu J, Zhu H (2009) Threshold conditions for West Nile virus outbreaks. Bull Math Biol 71(3):627–647
LaSalle J, Lefschetz S (1976) The stability of dynamical systems. SIAM, Philadelphia
Niger AM, Gumel AB (2008) Mathematical analysis of the role of repeated exposure on malaria transmission dynamics. Dyn Syst Differ Equ 16(3):251–287
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Proof of Theorem 3.1
Proof
Consider the model (1) in the absence of disease-induced mortality in the host population (i.e., \(\delta =0\)). Setting \(\delta =0\) in the model (1) shows that \(N_{H}(t) \rightarrow N^*_{H}\) as \({t\rightarrow \infty }\) (hence, the limiting value of \(N_H(t)\), given by \(N_H^{*}\), will be used in the analysis for this setting). The proof of Theorem 3.1 is based on using the center manifold theorem (see, for instance, Blayneh et al. (2010), Castillo-Chavez and Song (2004) and Hussaini et al. (2016) for details on how the method is applied). After transforming the system (model equations) onto a center manifold (where the resulting linearized system has a zero eigenvalue and all other eigenvalues having negative real part), it can then be shown that the Jacobian of the linearized system (with \(\delta = 0\)) has left (\(\mathbf v \)) and right (\(\mathbf w \)) eigenvectors, given, respectively, by
where
and,
with \(G = \dfrac{b p^H_v \lambda S^*_V}{S^*_H}, K_1=\gamma _H+\mu _H,\,K_2=\alpha _1+\mu _H,\,K_3=\tau _H+\mu _H, \,K_4=\alpha _2+\mu _H,\,K_5=\beta +\mu _H\) and \(K_6=\eta +\mu _{NA}\). Hence, it follows from Theorem 4.1 in Castillo-Chavez and Song (2004) that the associated bifurcation coefficients of the model (1) are given, respectively, by (noting from above that \(w_1<0\), \(w_{10}<0\), \(w_2>0\), \(w_3>0\), \(w_5>0\), \(w_8>0\), \(v_2>0\) and \(v_3>0\)):
Thus, it follows from Theorem 4.1 in Castillo-Chavez and Song (2004) that the model (1), with \(\delta = 0\), does not undergo a backward bifurcation at \({\mathcal {R}}_{0} = 1\). \(\square \)
Appendix B: Proof of Theorem 3.2
Proof
Consider the model (1) with \(\delta =0\). Let \({\mathcal R}_{01}\le 1\). Furthermore, consider the following linear Lyapunov function:
where (noting that \(K_i (i=1,2,3,4)>0\) are as defined in Sect. 2),
with Lyapunov derivative (where a dot differentiation with respect to time t)
Hence, \(\dot{{\mathcal {L}}}\le 0\) if \({\mathcal {R}}_{01}\le 1\) with \(\dot{{\mathcal {L}}}= 0\) if and only if \(I_V=0\). It follows, by substituting \(I_V(t)=0\) into the model (1), that \((A_H(t),K_H(t),D_H(t),P_H(t),R_H(t),I_A(t),I_V(t))\rightarrow (0,0,0,0,0,0,0),\) as \(t\rightarrow \infty \). Furthermore, substituting \((A_H, K_H, D_H, P_H, R_H,I_A, I_V)(t)=(0,0,0,0,0)\) into the full model shows that \(S_H(t)\rightarrow \frac{\Pi _H}{\mu _H}\), \(S_A(t)\rightarrow \frac{\Pi _A}{\mu _A}\), \(F_{NA}(t)\rightarrow F^*_{NA}\) and \(S_V(t)\rightarrow \frac{\eta F^*_{NA}}{\mu _F}\), as \(t\rightarrow \infty .\) Therefore, \({\mathcal {L}}\) is a Lyapunov function in \(\Omega \backslash {\mathcal {E}}_0\), and it follows from LaSalle’s invariance principle (LaSalle and Lefschetz 1976) that every solution to the model (1) (with \(\delta =0\)), with initial conditions in \(\Omega \backslash {\mathcal {E}}_0\), converges to the non-trivial disease-free equilibrium (\({\mathcal {E}}_{1}\)) as \(t\rightarrow \infty \) whenever \({\mathcal R}_{01}<1.\) Hence, for the case of the model (1) with no disease-induced death in the host population (i.e., \(\delta =0)\), the non-trivial disease-free equilibrium of the model, \({\mathcal {E}}_1\), is globally asymptotically stable in \(\Omega \backslash {\mathcal {E}}_0\) if \({\mathcal {R}}_{01}\le 1\). \(\square \)
Appendix C: Proof of Theorem 3.3
Proof
Consider the model given by (1). It can be shown (as in Appendix A) that the associated right and left eigenvectors of the linearized system associated with the model (1) are given, respectively, by
where (noting that \(\epsilon _2< 1\)),
and,
with,
It follows from Theorem 4.1 in Castillo-Chavez and Song (2004) that the associated bifurcation coefficients, \(a_2\) and \(b_2\), are given, respectively, by
and,
Hence, it follows from (4), the bifurcation coefficient \(a_2\) is positive if the following inequality holds:
where
Since the coefficient \(b_2\) is automatically positive, it follows that the model (1) will undergo a backward bifurcation at \({\mathcal {R}}_0=1\) if Inequality (6) holds. \(\square \)
Rights and permissions
About this article
Cite this article
Iboi, E., Okuneye, K., Sharomi, O. et al. Comments on “A Mathematical Study to Control Visceral Leishmaniasis: An Application to South Sudan”. Bull Math Biol 80, 825–839 (2018). https://doi.org/10.1007/s11538-018-0403-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-018-0403-9