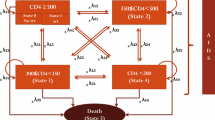
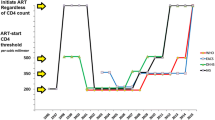
Abstract
HIV infection is one of the most difficult infections to control and manage. The most recent recommendations to control this infection vary according to the guidelines used (US, European, WHO) and are not patient-specific. Unfortunately, no two individuals respond to infection and treatment quite the same way. The purpose of this paper is to make use of the uncertainty and sensitivity analysis to investigate possible short-term treatment options that are patient-specific. We are able to identify the most significant parameters that are responsible for ART outcome and to formulate some insights into the ART success.









Similar content being viewed by others
References
Adams BM, Banks HT, Davidian M, Kwon H-D, Tran HT, Wynne SN, Rosenberg ES (2005) HIV dynamics: modeling, data analysis, and optimal treatment protocols. J Comput Appl Math 184:10–49
Bortz DM, Nelson PW (2006) Model selection and mixed-effects modeling of HIV infection dynamics. Bull Math Biol 68(8):2005–2025
Croicu A-M (2015) Short- and long-term optimal control of a mathematical model for HIV infection of \(CD4^{+} T\) cells. Bull Math Biol 77(11):2035–2071
Cukier RI, Fortuin CM, Shuler KE, Petschek AG, Schaibly JH (1973) Study of sensitivity of coupled reaction systems to uncertainties in rate coefficients. 1. Theory. J Chem Phys 59:3873–3878
Culshaw R, Ruan S, Spiteri R (2004) Optimal HIV treatment by maximizing immune response. J Math Biol 48(5):545–562
DiMascio M, Ribeiro RM, Markowitz M, Ho DD, Perelson AS (2004) Modeling the long-term control of viremia in HIV-1 infected patients treated with antiretroviral therapy. Math Biosci 188(1–2):47–62
Gulik RM, Mellors JW, Havlir D, Eron JJ, Gonzalez C, McMahon D et al (1997) Treatment with indinavir, zidovudine, and lamivudine in adults with human immunodeficiency virus infection and prior antiretroviral therapy. N Engl J Med 337:734–739
Gunthard HF et al (2014) Antiretroviral treatment of adult HIV infection: 2014 recommendations of the International Antiviral Society—USA Panel. Clin Rev Educ 312(4):410–425
Hadjiandreou MM, Conejeros R, Wilson DI (2009a) Long-term HIV dynamics subject to continuous therapy and structured treatment. Chem Eng Sci 64:1600–1617
Hadjiandreou MM, Conejeros R, Wilson DI (2009b) Planning of patient-specific drug-specific optimal HIV treatment strategies. Chem Eng Sci 64:4024–4039
Jang T, Kwon H-D, Lee J (2011) Free terminal time optimal control problem of an HIV model based on a conjugate gradient method. Bull Math Biol 73:2408–2429
Joshi HR (2002) Optimal control of an HIV immunology model. Optim Control Appl Methods 23:199–213
Karrakchou J, Rachik M, Gourari S (2006) Optimal control and infectiology: application to an HIV/AIDS model. Appl Math Comput 177:806–818
Kirschner D, Lenhart S, Serbin S (1997) Optimal control of the chemotherapy of HIV. J Math Biol 35:775–792
Kramer I (1999) Modeling the dynamical impact of HIV on the immune system: viral clearance, infection, and AIDS. Math Comput Modell 29:95–112
Kucherenko S, Feil B, Shah N, Mauntz W (2011) The identification of model effective dimensions using global sensitivity analysis. Reliab Eng Syst Saf 96:440–449
Kwon H-D, Lee J, Yang S-D (2012) Optimal control of an age-structured model of HIV infection. Appl Math Comput 219:2766–2779
Levy JA (2015) Dispelling myths and focusing on notable concepts in HIV pathogenesis. Trends Mol Med 21:341–353
Liu Y (2013) Non-intrusive methods for probabilistic uncertainty quantification and global sensitivity analysis in nonlinear stochastic phenomena. Florida State University, Tallahassee
Liu RX, Owen AB (2006) Estimating mean dimensionality of analysis of variance decompositions. J Am Stat Assoc 101:712–721
Louie M et al (2003) Determining the antiviral activity of tenofovir disoproxil fumarate in treatment-naive chronically HIV-1-infected individuals. AIDS 17:1151–1156
Maskey S, Guinot V, Price RK (2004) Treatment of precipitation uncertainty in rainfall-runoff modelling: a fuzzy set approach. Adv Water Resour 27(9):889–898
Merdan M, Gokdogan A, Yildirim A (2011) On the numerical solution of the model for HIV infection of \(CD4^{+}T\) cells. Comput Math Appl 62:118–123
Montaner JS, Reiss P, Cooper D, Vella S, Harris M, Conway B et al (1998) A randomized, double-blind trial comparing combinations of nevirapine, didanosine, and zidovudine for HIV-infected patients: the INCAS Trial. Italy, The Netherlands, Canada and Australia Study. J Am Med Assoc 279:930–937
Morgan D, Mahe C, Okongo B, Lubega R, Whitworth JA (2002) HIV-1 infection in rural Africa: Is there a difference in median time to aids and survival compared with that in industrialized countries? AIDS 16:597–632
Oberkampf WL, Roy CJ (2010) Verification and validation in scientific computing. Cambridge University Press, Cambridge. ISBN 978-0-511-90800-2
Orellana JM (2011) Optimal drug scheduling for HIV therapy efficiency improvement. Biomed Signal Process Control 6:379–386
Perelson AS, Nelson PW (1999) Mathematical analysis of HIV-I dynamics in vivo. SIAM Rev 41:3–44
Perelson AS, Kirschner DE, Boer RD (1993) Dynamics of HIV infection of \(CD4^{+}T\) cells. Math Biosci 114:81–125
Perera N (2003) Deterministic and stochastic models of virus dynamics. PhD thesis, Texas Tech University
Pontryagin LS, Boltyanskii VG, Gamkrelidze RV, Mishchenko EF (1962) The mathematical theory of optimal processes. Wiley, Hoboken
Roshanfekr M, Farahi MH, Rahbarian R (2014) A different approach of optimal control on an HIV immunology model. Ain Shams Eng J 5:213–219
Saltelli A (2002) Making best use of model evaluations to compute sensitivity indices. Comput Phys 145:280–297
Saltelli A, Bolando R (1998) An alternative way to compute Fourier amplitude sensitivity test (FAST). Comput Stat Data Anal 26:445–460
Shi S, Nguyen PK, Cabral HJ, Diez-Barroso R, Derry PJ, Kanahara SM, Kumar VA (2016) Development of peptide inhibitors of HIV transmission. Bioact Mater 1–13
Simpson L, Gumel AB (2017) Mathematical assessment of the role of pre-exposure prophylaxis on HIV transmission dynamics. Appl Math Comput 293:168–193
Sobol IM (1993) Sensitivity estimates for non-linear mathematical models. Math Model Comput Exp 1:407–414
Sobol IM (2001) Global sensitivity indices for nonlinear mathematical models and their Monte Carlo estimates. Math Comput Simul 55:271–280
Stengel RF (2008) Mutation and control of the human immunodeficiency virus. Math Biosci 213:93–102
Sued O, Figueroa MI, Cahn P (2016) Clinical challenges in HIV/AIDS: hints for advancing prevention and patient management strategies. Adv Drug Deliv Rev 103:5–19
UNAIDS (2015) How AIDS changed everything
Wang L, Li MY (2006) Mathematical analysis of the global dynamics of a model for HIV infection of \(CD4^{+}T\) cells. Math Biosci 200:44–57
Wodarz D, Hamer DH (2007) Infection dynamics in HIV-specific CD4 T cells: does a CD4 T cell boost benefit the host or the virus? Math Biosci 209:14–29
Wu H, Ding A, DeGruttola V (1999) Why are the decay rates in plasma HIV-1 different for different treatments and in different patient populations? AIDS 13(3):429–430
Yuzbasi S (2012) A numerical approach to solve the model for HIV infection of \(CD4^{+}T\) cells. Appl Math Model 36:5876–5890
Zhou Y, Liang Y, Wu J (2014) An optimal strategy for HIV multitherapy. J Comput Appl Math 263:326–337
Acknowledgements
This work was partially supported by the NSF-CBET #1510743 Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Croicu, AM., Jarrett, A.M., Cogan, N.G. et al. Short-Term Antiretroviral Treatment Recommendations Based on Sensitivity Analysis of a Mathematical Model for HIV Infection of CD4+T Cells. Bull Math Biol 79, 2649–2671 (2017). https://doi.org/10.1007/s11538-017-0345-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0345-7