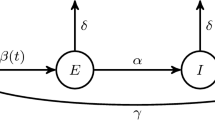
Abstract
A new method is proposed to infer unobserved epidemic subpopulations by exploiting the synchronization properties of multistrain epidemic models. A model for dengue fever is driven by simulated data from secondary infective populations. Primary infective populations in the driven system synchronize to the correct values from the driver system. Most hospital cases of dengue are secondary infections, so this method provides a way to deduce unobserved primary infection levels. We derive center manifold equations that relate the driven system to the driver system and thus motivate the use of synchronization to predict unobserved primary infectives. Synchronization stability between primary and secondary infections is demonstrated through numerical measurements of conditional Lyapunov exponents and through time series simulations.





Similar content being viewed by others
References
Anderson RM, May RM (1991) Infectious diseases of humans. Oxford University Press, Oxford
Bianco S, Shaw LB, Schwartz IB (2009) Epidemics with multistrain interactions: the interplay between cross immunity and antibody-dependent enhancement. Chaos 19:043123
Billings L, Schwartz IB, Shaw LB, McCrary M, Burke DS, Cummings DAT (2007) Instabilities in multiserotype disease models with antibody-dependent enhancement. J Theor Biol 246:18
Bjornstad ON, Finkenstadt BF, Grenfell BT (2002) Dynamics of measles epidemics: estimating scaling of transmission rates using a time series sir model. Ecolo Monogr 72(2):169–184
Blarer A, Doebeli M (1999) Resonance effects and outbreaks in ecological time series. Ecol Lett 2:167–177
Boccaletti S, Kurths J, Osipov G, Valladares DL, Zhou CS (2002) The synchronization of chaotic systems. Phys Rep Rev Sect Phys Lett 366:1–101
Carr J (1981) Applications of centre manifold theory. Springer, Berlin
Chen S, Lü J (2002) Parameters identification and synchronization of chaotic systems based upon adaptive control. Phys Lett A 299:353
Chicone C, Latushkin Y (1997) Center manifolds for infinite dimensional nonautonomous differential equations. J Differ Equ 141:356–399
Cummings DAT, Schwartz IB, Billings L, Shaw LB, Burke DS (2005) Dynamic effects of antibody-dependent enhancement on the fitness of viruses. Proc Nat Acad Sci USA 102(42):15259–15264
Dedieu H, Ogorzalek MJ (1997) Identifiability and identification of chaotic systems based on adaptive synchronization. IEEE Trans Circuits Syst I Fundam Theory Appl 44:948
Duan J, Lu K, Schmalfuss B (2003) Invariant manifolds for stochastic partial differential equations. Ann. Probab. 31(4):2109–2135
Ferguson NM, Donnelly CA, Anderson RM (1999) Transmission dynamics and epidemiology of dengue: insights from age-stratified sero-prevalence surveys. Philos Trans R Soc Lond B Biol Sci 354:757–768
Forgoston E, Billings L, Schwartz IB (2009) Accurate noise projection for reduced stochastic epidemic models. Chaos 19:043110
Forgoston E, Schwartz IB (2013) Predicting unobserved exposures from seasonal epidemic data. Bull Math Biol 75:1450
Gibson GJ, Kleczkowski A, Gilligan CA (2004) Bayesian analysis of botanical epidemics using stochastic compartmental models. PNAS 101:12120
Huang L, Lin L (2013) Parameter identification and synchronization of uncertain chaotic systems based on sliding mode observer. Mathe Probl Eng 2013:859304
Lekone PE, Finkenstädt BF (2006) Statistical inference in a stochastic epidemic seir model with control intervention: ebola as a case study. Biometrics 62:1170
Nagao Y, Koelle K (2008) Decreases in dengue transmission may act to increase the incidence of dengue hemorrhagic fever. Proc Natl Acad Sci USA 105:2238–2243
Nisalak A, Endy TP, Nimmannitya S, Kalayanarooj S, Thisayakorn U, Scott RM, Burke DS, Hoke CH, Innis BL, Vaughn DW (2003) Serotype-specific dengue virus circulation and dengue disease in Bangkok, Thailand from 1973 to 1999. Am J Trop Med Hyg 68:191–202
Parlitz U, Junge L, Kocarev L (1996) Synchronization-based parameter estimation from time series. Phys Rev E 54:6253
Rigau-Perez J, Clark G, Gubler D, Reiter P, Sanders E, Vancevorndam A (1998) Dengue and dengue hemmorrhagic fever. Lancet 352:971–977
Roberts AJ (2008) Normal form transforms separate slow and fast modes in stochastic dynamical systems. Phys A 387(1):12–38
Schaffer WM, Kendall BE, Tidd CW, Olsen LF (1993) Transient periodicity and episodic predictability in biological dynamics. IMA J Math Appl Med 10:227–247
Schwartz I, Smith H (1983) Infinite subharmonic bifurcations in an SEIR epidemic model. J Math Biol 18:233–253
Schwartz IB, Shaw LB, Cummings D, Billings L, McCrary M, Burke D (2005) Chaotic desynchronization of multi-strain diseases. Phys Rev E 72:066201
Shaw LB, Billings L, Schwartz IB (2007) Using dimension reduction to improve outbreak predictability of multistrain diseases. J Math Biol 55:1–19
Acknowledgments
EF is supported by Award Number CMMI-1233397 from the National Science Foundation. LBS is supported by Award Number R01GM090204 from the National Institute of General Medical Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute Of General Medical Sciences or the National Institutes of Health. IBS is supported by the NRL Base Research Program contract number N0001414WX00023 and by the Office of Naval Research contract number N0001414WX20610.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Shifted, Rescaled and Augmented System of Equations
The governing equations for the two-serotype multistrain disease model and the subsystem driven by secondary infectious individuals are given by Eqs. (4a)–(5c). We define a new set of variables, \(\bar{s}\), \(\bar{x}_i\), \(\bar{r}_i\), \(\bar{x}_{ij}\), \(\bar{s}_d\) and \(\bar{x}_{id}\) for all i, j as \(\bar{s}(t)=s(t)-s_0\), \(\bar{x}_i(t)=x_i(t)-x_{i,0}\), \(\bar{r}_i(t)=r(t)-r_{i,0}\), \(\bar{x}_{ij}(t)=x_{ij}(t)-x_{ij,0}\), \(\bar{s}_d(t)=s_d(t)-s_{d,0}\), \(\bar{x}_{id}(t)=x_{id}(t)-x_{id,0}\), and these new variables are substituted into Eqs. (4a)–(5c).
Then, treating \(\mu \) as a small parameter, we rescale time by letting \(t=\mu \tau \). We may then introduce the following rescaled parameters: \(\beta =\beta _0/\mu \) and \(\sigma =\sigma _0/\mu \), where \(\beta _0\) and \(\sigma _0\) are \(\mathcal {O}(1)\). The inclusion of the parameter \(\mu \) as a new state variable means that the terms in our rescaled system which contain \(\mu \) are now nonlinear terms. Furthermore, the system is augmented with the auxiliary equation \(\frac{d\mu }{d\tau }=0\). The addition of this auxiliary equation contributes an extra simple zero eigenvalue to the system and adds one new center direction that has trivial dynamics. The shifted, rescaled, and augmented system of equations is given as
where the endemic fixed point is now located at the origin.
Appendix 2: Definition of New Variables
Using the fact that \((\bar{s},\bar{x}_1,\bar{x}_2,\bar{r}_1,\bar{r}_2,\bar{x}_{21},\bar{x}_{12},\bar{s}_d,\bar{x}_{1d},\bar{x}_{2d})^T = \mathbf{P}\cdot \mathbf{W}^T\), where \(\mathbf{P}\) is given by Eq. (7) and \(\mathbf{W}=(W_1,W_2,W_3,W_4,W_5,W_6,W_7,W_8,W_9,W_{10})\), then the transformation matrix leads to the following definition of new variables, \(W_i\), \(i=1\ldots 10\):
Appendix 3: Transformed Evolution Equations
The application of the transformation matrix \(\mathbf{P}\) given by Eqs. (7) to (21a)–(21j) leads to the following set of transformed evolution equations:
Appendix 4: Center Manifold Condition
Substitution of the center manifold functions \(W_i=h_i\) given by Eq. (12) into the transformed evolution equations given in “Appendix 3” leads to the following center manifold condition:
Rights and permissions
About this article
Cite this article
Forgoston, E., Shaw, L.B. & Schwartz, I.B. A Framework for Inferring Unobserved Multistrain Epidemic Subpopulations Using Synchronization Dynamics. Bull Math Biol 77, 1437–1455 (2015). https://doi.org/10.1007/s11538-015-0091-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-015-0091-7