Abstract
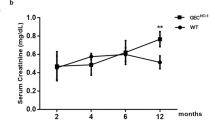
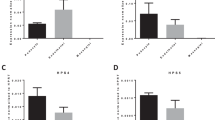
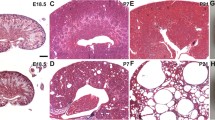
Although the protective role of HO-1 induction in various forms of kidney disease is well established, mechanisms other than heme catabolism to biliverdin, bilirubin and carbon monoxide have recently been identified. Unraveling these mechanisms requires the generation of appropriate animal models. The present study describes the generation of a HO-1 deficient Hmox1 −/− rat model and characterizes its renal and extrarenal phenotype. Hmox1 −/− rats had growth retardation and splenomegaly compared to their Hmox1 +/+ littermates. Focal segmental glomerulosclerosis-type lesions and interstitial inflammatory infiltrates were prominent morphologic findings and were associated with increased blood urea nitrogen, serum creatinine and albuminuria. There was no increase in iron deposition in glomeruli, tubules or interstitium. Iron deposition in spleen and liver was reduced. Electron microscopic examination of glomeruli revealed edematous podocytes with scant areas of foot process effacement but otherwise well preserved processes and slit-diaphragms. Of the filtration barrier proteins examined, β-catenin expression was markedly reduced both in glomeruli and extrarenal tissues. Since the rat is the preferred laboratory animal in experimental physiology and pathophysiology, the rat model of HO-1 deficiency may provide a novel tool for investigation of the role of this enzyme in renal function and disease.







Similar content being viewed by others
References
Agarwal A, Nick HS (2000) Renal response to tissue injury: lessons from heme oxygenase-1 Gene Ablation and expression. J Am Soc Nephrol 11:965–973
Aichele P, Zinke J, Grode L, Schwendener RA, Kaufmann SH, Seiler P (2003) Macrophages of the splenic marginal zone are essential for trapping of blood-borne particulate antigen but dispensable for induction of specific T cell responses. J Immunol 171:1148–1155
Anders H, Schlondorff D (2000) Murine models of renal disease: possibilities and problems in studies using mutant mice. Exp Nephrol 8:181–193
Anyanwu AC et al (2014) Suppression of inflammatory cell trafficking and alveolar simplification by the heme oxygenase-1 product carbon monoxide. Am J Physiol Lung Cell Mol Physiol 306:749–763. doi:10.1152/ajplung.00236.2013
Atsaves V et al (2014) The oncogenic JUNB/CD30 axis contributes to cell cycle deregulation in ALK + anaplastic large cell lymphoma. Br J Haematol. doi:10.1111/bjh.13079
Becker GJ, Hewitson TD (2013) Animal models of chronic kidney disease: useful but not perfect. Nephrol Dial Transplant 28:2432–2438. doi:10.1093/ndt/gft071
Cao J et al (2012) Heme oxygenase gene targeting to adipocytes attenuates adiposity and vascular dysfunction in mice fed a high-fat diet. Hypertension 60:467–475. doi:10.1161/hypertensionaha.112.193805
Cybulsky AV, Quigg RJ, Salant DJ (2005) Experimental membranous nephropathy redux. Am J Physiol Renal Physiol 289:F660–F671. doi:10.1152/ajprenal.00437.2004
Daehn I et al (2014) Endothelial mitochondrial oxidative stress determines podocyte depletion in segmental glomerulosclerosis. J Clin Investig 124:1608–1621. doi:10.1172/jci71195
Darouich S, Goucha R, Jaafoura MH, Zekri S, Ben Maiz H, Kheder A (2011) Clinicopathological characteristics of obesity-associated focal segmental glomerulosclerosis. Ultrastruct Pathol 35:176–182. doi:10.3109/01913123.2011.584657
Detsika MG et al (2016) Effect of heme oxygenase-1 deficiency on glomerular proteomics. Am J Nephrol 43:441–450. doi:10.1159/000446859
Doorschodt BM, Teubner A, Kobayashi E, Tolba RH (2014) Promising future for the transgenic rat in transplantation research. Transplant Rev 28:155–162. doi:10.1016/j.trre.2014.05.002
Fraser ST, Midwinter RG, Berger BS, Stocker R (2011) Heme oxygenase-1: a critical link between iron metabolism, erythropoiesis, and development. Adv Hematol 2011:473709. doi:10.1155/2011/473709
Geurts AM et al (2010) Generation of gene-specific mutated rats using zinc-finger nucleases. Methods Mol Biol 597:211–225. doi:10.1007/978-1-60327-389-3_15
Jacob HJ, Lazar J, Dwinell MR, Moreno C, Geurts AM (2010) Gene targeting in the rat: advances and opportunities. Trends Genet 26:510–518. doi:10.1016/j.tig.2010.08.006
Kato H et al (2011) Wnt/beta-catenin pathway in podocytes integrates cell adhesion, differentiation, and survival. J Biol Chem 286:26003–26015. doi:10.1074/jbc.M111.223164
Kawashima A, Oda Y, Yachie A, Koizumi S, Nakanishi I (2002) Heme oxygenase-1 deficiency: the first autopsy case. Hum Pathol 33:125–130
Konrad FM, Braun S, Ngamsri KC, Vollmer I, Reutershan J (2014) Heme oxygenase-1 attenuates acute pulmonary inflammation by decreasing the release of segmented neutrophils from the bone marrow. Am J Physiol Lung Cell Mol Physiol 307:L707–L717. doi:10.1152/ajplung.00145.2014
Kovtunovych G, Eckhaus MA, Ghosh MC, Ollivierre-Wilson H, Rouault TA (2010) Dysfunction of the heme recycling system in heme oxygenase 1-deficient mice: effects on macrophage viability and tissue iron distribution. Blood 116:6054–6062. doi:10.1182/blood-2010-03-272138
Kovtunovych G et al (2014) Wild-type macrophages reverse disease in heme oxygenase 1-deficient mice. Blood 124:1522–1530. doi:10.1182/blood-2014-02-554162
Lever JM, Boddu R, George JF, Agarwal A (2016) Heme oxygenase-1 in kidney health and disease. Antioxid Redox Signal. doi:10.1089/ars.2016.6659
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Maines MD, Gibbs PE (2005) 30 some years of heme oxygenase: from a “molecular wrecking ball” to a “mesmerizing” trigger of cellular events. Biochem Biophys Res Commun 338:568–577. doi:10.1016/j.bbrc.2005.08.121
Misra RP (1972) Isolation of glomeruli from mammalian kidneys by graded sieving. Am J Clin Pathol 58:135–139
Nath KA, Haggard JJ, Croatt AJ, Grande JP, Poss KD, Alam J (2000) The indispensability of heme oxygenase-1 in protecting against acute heme protein-induced toxicity in vivo. Am J Pathol 156:1527–1535. doi:10.1016/S0002-9440(10)65024-9
Ndisang JF, Tiwari S (2014) Mechanisms by which heme oxygenase rescue renal dysfunction in obesity. Redox Biol 2:1029–1037. doi:10.1016/j.redox.2014.09.001
Nishino T, Sasaki N, Nagasaki K, Ahmad Z, Agui T (2010) Genetic background strongly influences the severity of glomerulosclerosis in mice. J Vet Med Sci/Jpn Soc Vet Sci 72:1313–1318
Patrakka J, Tryggvason K (2010) Molecular make-up of the glomerular filtration barrier. Biochem Biophys Res Commun 396:164–169. doi:10.1016/j.bbrc.2010.04.069
Poss KD, Tonegawa S (1997) Heme oxygenase 1 is required for mammalian iron reutilization. Proc Natl Acad Sci USA 94:10919–10924
Radhakrishnan N et al (2011) Human heme oxygenase-1 deficiency presenting with hemolysis, nephritis, and asplenia. J Pediatr Hematol/Oncol 33:74–78. doi:10.1097/MPH.0b013e3181fd2aae
Soares MP et al (1998) Expression of heme oxygenase-1 can determine cardiac xenograft survival. Nat Med 4:1073–1077. doi:10.1038/2063
Vanella L et al (2013) Increased heme-oxygenase 1 expression in mesenchymal stem cell-derived adipocytes decreases differentiation and lipid accumulation via upregulation of the canonical Wnt signaling cascade. Stem Cell Res Ther 4:28. doi:10.1186/scrt176
Voskarides K, Deltas C (2009) Screening for mutations in kidney-related genes using SURVEYOR nuclease for cleavage at heteroduplex mismatches. J Mol Diagn 11:311–318. doi:10.2353/jmoldx.2009.080144
Xia ZW et al (2002) Analysis of heme oxygenase isomers in rat. World J Gastroenterol 8:1123–1128
Yachie A et al (1999) Oxidative stress causes enhanced endothelial cell injury in human heme oxygenase-1 deficiency. J Clin Investig 103:129–135. doi:10.1172/jci4165
Yang G et al (2013) Heme oxygenase-1 regulates postnatal lung repair after hyperoxia: role of beta-catenin/hnRNPK signaling. Redox Biol 1:234–243. doi:10.1016/j.redox.2013.01.013
Acknowledgments
We would like to thank Zoe Kolia for technical assistance on animal work.
Funding
This study was co-financed by a European Union (European Social Fund, ESF) and by Greek National Funds through the Operational Program ‘‘Education and Lifelong Learning’’ of the National Strategic Reference Framework (NSRF) Research Funding Program: “Aristeia- I” to E. A. L.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Atsaves, V., Detsika, M.G., Poulaki, E. et al. Phenotypic characterization of a novel HO-1 depletion model in the rat. Transgenic Res 26, 51–64 (2017). https://doi.org/10.1007/s11248-016-9986-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-016-9986-9