Abstract
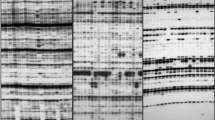
Efficacy of two dominant molecular markers, namely, amplified fragment length polymorphism (AFLP) and three endonuclease (TE)-AFLP, were assessed in 20 individuals of the biodiesel species Pongamia pinnata. Four primer combinations generated a total of 254 and 194 bands in AFLP and TE-AFLP, respectively. Both techniques could unequivocally identify each accession used in this study. The Jaccard’s similarity coefficient ranged from 0.30 to 0.90 for AFLP and from 0.25 to 0.85 for TE-AFLP. The correlation coefficient between AFLP and TE-AFLP dendrogram was 0.56 which was low but significant (P < 0.001). Values of effective multiplex ratio, marker index, and resolving power were markedly higher in AFLP than in TE-AFLP. However, the band intensities across different lanes were uniform in TE-AFLP leading to easy and accurate scoring of gels which resulted in slightly higher bootstrap values with TE-AFLP data as compared to AFLP data. Inferences based on TE-AFLP data had similar level of biological relevance as compared to AFLP data when location and diameter of trees were taken in to consideration. However, the easy scorability of TE-AFLP profiles is extremely important and especially desirable in studies requiring genotyping of large number of individuals distributed across many gels.


Similar content being viewed by others
References
Feng C, Hou A, Chen P, Cornelious B, Shi A, Zhang B (2008) Genetic diversity among popular historical Southern U.S. soybean cultivars using AFLP markers. J Crop Improvement 22:31–46
Garcia AAF, Benchimol LL, Barbosa AMM, Geraldi IO, Souza CLJ, de Souza AP (2004) Comparison of RAPD, RFLP, AFLP and SSR markers for diversity studies in tropical maize inbred lines. Genet Mol Biol 27:579–588
Han TH, van Eck HJ, De Jeu MJ, Jacobsen E (1999) Optimization of AFLP fingerprinting of organisms with a large-sized genome: a study on Alstroemeria spp. Theor Appl Genet 98:465–471
Jaccard P (1908) Nouvelles rescherches sur la distribution florale. Bull Soc Vaud Sci Nat 44:223–270
Kaushik N, Kumar S, Kumar K, Beniwal R, Kaushik N, Roy S (2007) Genetic variability and association studies in pod and seed traits of Pongamia pinnata (L.) Pierre in Haryana, India. Genet Res Crop Evol 54:1827–1832
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Perrier X, Jacquemoud-Collet JP (2006) DARwin software, Version 5.0.158, http://darwin.cirad.fr/darwin
Powell W, Morgante M, Andre C, Hanafey M, Vogel J, Tingey S, Rafalski A (1996) The comparison of RFLP, RAPD, AFLP and SSR (microsatellite) markers for germplasm analysis. Mol Breed 2:225–238
Prevost A, Wilkinson MJ (1999) A new system of comparing PCR primers applied to ISSR fingerprinting of potato cultivars. Theor Appl Genet 98:107–112
Rohlf FJ (2000) NTSYS-pc: Numerical Taxonomy and Multivariate Analysis System, Version 2.2, Exeter Software, Setauket, NY
Roldan-Ruiz I, Dendauw J, VanBockstaele E, Depicker A, Loose MD (2000) AFLP markers reveal high polymorphic rates in ryegrasses (Lolium spp.). Mol Breed 6:125–134
Singh A, Negi MS, Rajagopal J, Bhatia S, Tomar UK, Srivastava PS, Lakshmikumaran M (1999) Assessment of genetic diversity in Azadirachta indica using AFLP markers. Theor Appl Genet 99:272–279
Tewari DN (2003) Report of the committee on development of Bio-fuel, Planning Commission, Government of India
van der Wurff AWG, Chan YL, van Straalen NM, Schouten J (2000) TE-AFLP: combining rapidity and robustness in DNA fingerprinting. Nucl Acids Res 28:e105
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Friters A, Pot J, Paleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23:4407–4414
Williams JG, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl Acids Res 18:6531–6535
Yue G, Young (2001) Selective AFLP primers for reduction of complexity of marker genotyping and DNA markers for loblolly pine identified using AFLP primers USPTO No. 6,306, 593
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183
Acknowledgments
The authors thank Department of Science and Technology, Government of India, for its gracious funding for the project (grant number SR/SO/PS-08/2006). We thank Mr. Bidya Bhushan Gupta and Mr. Bharatendu Gupta for their assistance during field surveys. We thank the anonymous reviewer for valuable suggestions. The authors are also thankful to Dr. R. K. Pachauri, Director General, TERI, for his kind support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure S1
Mantel matrix plot between pairwise cophenetic distances obtained from AFLP and TE-AFLP data. R is the matrix correlation coefficient between the two matrices (PPT 31 kb)
Rights and permissions
About this article
Cite this article
Sharma, S.S., Negi, M.S., Sinha, P. et al. Assessment of Genetic Diversity of Biodiesel Species Pongamia pinnata Accessions using AFLP and Three Endonuclease-AFLP. Plant Mol Biol Rep 29, 12–18 (2011). https://doi.org/10.1007/s11105-010-0204-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-010-0204-2