Abstract
Purpose
In clinical research involving acromegalic patients naïve to somatostatin-receptor ligands (SRLs), 19 and 31% of those receiving the SRLs octreotide LAR and pasireotide LAR, respectively, achieved GH < 2.5 ng/mL + normalized IGF-1 concentrations. The proportions achieving control appeared higher in the post-surgery compared with the de-novo setting with pasireotide, but more similar with octreotide. Using pooled data from multicenter clinical trials, we examined the biochemical efficacy of lanreotide depot/Autogel in similar settings.
Methods
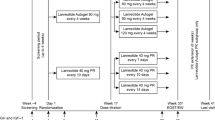
Inclusion criteria: Ipsen-sponsored, 48–52-week trials in SRL-naïve acromegalic populations receiving lanreotide depot (60–120 mg); patients were included if de novo (no prior acromegaly treatment) or post-surgery (no medical treatment; radiotherapy allowed unless within previous 3 years). Efficacy endpoints included normalized IGF-1 levels and GH < 2.5 ng/mL + normalized IGF-1 at study end/last value available. Analyses: all patients (analysis #1) and subset with baseline GH > 5 ng/mL (analysis #2).
Results
Three studies were included. Analysis #1: normalized IGF-1 was achieved by 42% (71/171) of patients overall (post-surgery, 46% [21/46]; de-novo, 40% [50/125]); GH < 2.5 ng/mL + normalized IGF-1 was achieved by 35% (59/171) (39% [18/46] and 33% [41/125], respectively). Analysis #2: normalized IGF-1 levels, 39% (46/118) (post-surgery, 40% [10/25]; de-novo, 39% [36/93]); GH < 2.5 ng/mL + normalized IGF-1, 31% (36/118) (28% [7/25] and 31% [29/93], respectively).
Conclusion
In these pooled analyses of SRL-naïve patients receiving lanreotide depot, 39–42% achieved IGF-1 control and 31–35% achieved GH and IGF-1 control. Control rates within post-surgery cohorts did not differ markedly from those in corresponding de-novo cohorts.
Similar content being viewed by others
References
Holdaway IM, Bolland MJ, Gamble GD (2008) A meta-analysis of the effect of lowering serum levels of GH and IGF-I on mortality in acromegaly. Eur J Endocrinol 159:89–95
Katznelson L, Laws ER Jr, Melmed S, Molitch ME, Murad MH, Utz A, Wass JA (2014) Acromegaly: an endocrine society clinical practice guideline. Endocr Soc J Clin Endocrinol Metab 99:3933–3951
Colao A, Auriemma RS, Pivonello R, Kasuki L, Gadelha MR (2016) Interpreting biochemical control response rates with first-generation somatostatin analogues in acromegaly. Pituitary 19:235–247
Colao A, Bronstein MD, Freda P, Gu F, Shen CC, Gadelha M, Fleseriu M, van der Lely AJ, Farrall AJ, Hermosillo Reséndiz K, Ruffin M, Chen Y, Sheppard M (2014) Pasireotide C2305 Study Group.: Pasireotide versus octreotide in acromegaly: a head- to-head superiority study. J Clin Endocrinol Metab 99:791–799
Bevan JS, Atkin SL, Atkinson AB, Bouloux P-M, Hanna F, Harris PE, James RA, McConnell M, Scanlon MF, Stewart PM, Teasdale E, Turner HE, Wass JAH, Wardlaw JM (2002) Primary medical therapy for acromegaly: an open, prospective, multicenter trial of the effects of subcutaneous and intramuscular slow-release octreotide on growth hormone, insulin-like growth factor—I, and tumor size. J Clin Endocrinol Metab 87:4554–4563
Caron PJ, Bevan JS, Petersenn S, Flanagan D, Tabarin A, Prévost G, Maisonobe P, Clermont A, PRIMARYS Investigators (2014) Tumor shrinkage with lanreotide Autogel 120 mg as primary therapy in acromegaly: results of a prospective multicenter clinical trial. J Clin Endocrinol Metab 99:1282–1290
Chanson P, Borson-Chazot F, Kuhn JM, Blumberg J, Maisonobe P, Delemer B, Lanreotide Acromegaly Study Group (2008) Control of IGF-I levels with titrated dosing of lanreotide Autogel over 48 weeks in patients with acromegaly. Clin Endocrinol (Oxf) 69, 299–305
Melmed S, Cook D, Schopohl J, Goth MI, Lam KS, Marek J (2010) Rapid and sustained reduction of serum growth hormone and insulin-like growth factor-1 in patients with acromegaly receiving lanreotide Autogel therapy: a randomized, placebo-controlled, multicenter study with a 52 week open extension. Pituitary 13:18–28
Caron PJ, Petersenn S, Houchard A, Sert C, Bevan JS, PRIMARYS Study Group (2017) Glucose and lipid levels with lanreotide autogel 120 mg in in treatment-naïve patients with acromegaly: data from the PRIMARYS study. Clin Endocrinol (Oxf) 86:541–551
Giustina A, Chanson P, Kleinberg D, Bronstein MD, Clemmons DR, Klibanski A, van der Lely AJ, Strasburger CJ, Lamberts SW, Ho KK, Casanueva FF, Melmed S (2014) Acromegaly consensus group: expert consensus document: a consensus on the medical treatment of acromegaly. Nat Rev Endocrinol 10:243–248
Dimaraki EV, Jaffe CA, Mott-Friberg RDE, Chandler WF, Barkan AL (2002) Acromegaly with apparently normal GH secretion: implications for diagnosis and follow-up. J Clin Endocrinol Metab 87:3537–3542
Butz LB, Sullivan SE, Chandler WF, Barkan AL (2016) “Micromegaly”: an update on the prevalence of acromegaly with apparently normal GH secretion in the modern era. Pituitary 19:547–551
Faje AT, Barkan AL (2010) Basal, but not pulsatile, growth hormone secretion determines the ambient circulating levels of insulin-like growth factor-I. J Clin Endocrinol Metab 95:2486–2491
Jaffe CA, Barkan AL (1994) Acromegaly. Recognition and treatment. Drugs 47:425–445
Neggers SJ, Pronin V, Balcere I, Lee MK, Rozhinskaya L, Bronstein MD, Gadelha MR, Maisonobe P, Sert C, van der Lely AJ (2015) LEAD Study Group.: Lanreotide Autogel 120 mg at extended dosing intervals in patients with acromegaly biochemically controlled with octreotide LAR: the LEAD study. Eur J Endocrinol 173:313–323
Giustina A, Bonadonna S, Bugari G, Colao A, Cozzi R, Cannavo S, de Marinis L, Uberti D, Bogazzi E, Mazziotti F, Minuto G, Montini F, Ghigo M (2009) High-dose intramuscular octreotide in patients with acromegaly inadequately controlled on conventional somatostatin analogue therapy: a randomised controlled trial. Eur J Endocrinol 161:331–338
Colao A, Ferone D, Marzullo P, Di Sarno A, Longobardi S, Merola S, Salvattore M, Lombardi G (1996) Prediction of efficacy of octreotide therapy in patients with acromegaly. J Clin Endocrinol Metab 81:2356–2362
Biermasz NR, Pereira AM, Smit JWA, Romijn JA, Roelfsema F (2005) Intravenous octreotide test predicts the long-term outcome of treatment with ctreotide-long-acting repeatable in active acromegaly. Growth Horm IGF Res 15:200–226
Cuevas-Ramos D, Carmichael JD, Cooper O, Bonert V, Gertych A, Mamelak AN, Melmed S (2014) A structural and functional acromegaly classification. J Clin Endocrinol Metab 100:122–131
Acknowledgements
We thank the patients and investigators who participated in the studies in this analysis. Medical writing for later drafts of the manuscript (funded by Ipsen) was provided by Watermeadow Medical, an Ashfield Company, part of UDG Healthcare plc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
HA reports no conflict of interests; MdPS and BM are employees of Ipsen; AB received research funding from Novartis and served as consultant for Ipsen, Novartis and Pfizer.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was taken from all participants in these studies.
Rights and permissions
About this article
Cite this article
Alquraini, H., del Pilar Schneider, M., Mirakhur, B. et al. Biochemical efficacy of long-acting lanreotide depot/Autogel in patients with acromegaly naïve to somatostatin-receptor ligands: analysis of three multicenter clinical trials. Pituitary 21, 283–289 (2018). https://doi.org/10.1007/s11102-018-0867-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-018-0867-5