Abstract
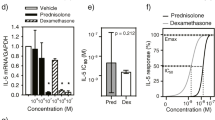
There is a significant fluctuation in clinical symptoms of asthmatic females during their life course, suggesting that the reproductive status and the level of sex hormones may affect the development of asthma and its exacerbation. In this study, we aimed to assess the biological effects of 17β-estradiol (E2) and progesterone (P4), alone or in combination form, on the transcription factors and production of cytokines in peripheral blood mononuclear cells (PBMCs). PBMCs of the mild-to-moderate asthmatic patients and healthy controls (HCs) were treated with equivalent serum levels of E2 or P4 maintained during hormone replacement therapy (HRT). The expression levels of T-bet, GATA-3, RORγt, PU.1, and Foxp3 were assessed by quantitative PCR. We also measured the concentration of IL-4, IL-9, IL-10, IFN-γ, and TGF-β in cell culture supernatants using ELISA. IL-4 production and GATA-3 expression levels slightly increased when asthmatic PBMCs were treated with E2 (p < 0.01), P4 (p < 0.01), or E2 + P4 (p < 0.001) compared to the untreated cells. IL-9 secretion (p < 0.001) and PU.1 gene expression levels (p < 0.05) were slightly higher in asthmatic patients’ PBMCs before treatment but hormone therapy did not affect the level of them. Although the untreated asthmatic PBMCs produced a lower amount of IFN-γ compared to HCs (p < 0.01), hormone treatment did not affect the levels of IFN-γ secretion in patient groups. Moreover, we did not observe any significant changes in IL-10 and TGF-β secretion in the supernatant of hormone treated cells. We found that the common applied HRT may faintly increase GATA-3 expression and IL-4 production levels in PBMCs of asthmatic patients and can slightly increase asthma severity.



Similar content being viewed by others
Data availability
All data are available and may be delivered on reasonable demand.
Abbreviations
- E2:
-
17β-Estradiol
- ER:
-
Estrogen receptor
- ELISA:
-
Enzyme‐linked immunosorbent assay
- ELISpot:
-
Enzyme-linked immune absorbent spot
- GINA:
-
Global initiative for asthma
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- HC:
-
Healthy control
- HRT:
-
Hormone replacement therapy
- IL:
-
Interleukin
- ILC:
-
Innate lymphoid cell
- IFN-γ:
-
Interferon gamma
- ICAM-1:
-
Intracellular cell adhesion molecule-1
- IQR:
-
Interquartile range
- P4:
-
Progesterone
- PBMCs:
-
Peripheral blood mononuclear cells
- PIBF:
-
Progesterone-induced blocking factor
- RT:
-
Room temperature
- RQ:
-
Relative quantity
- TH :
-
T helper
- TMB:
-
Tetramethylbenzidine
- TGF-β:
-
Transforming growth factor beta
- Treg :
-
Regulatory T helper cell
References
Hamilton D, Lehman H (2020) Asthma phenotypes as a guide for current and future biologic therapies. Clin Rev Allergy Immunol 59(2):160–174
Kuruvilla ME, Lee FE-H, Lee GB (2019) Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin Rev Allergy Immunol 56(2):219–233
Fazlollahi MR, Najmi M, Fallahnezhad M, Sabetkish N, Kazemnejad A, Bidad K et al (2018) The prevalence of asthma in Iranian adults: The first national survey and the most recent updates. Clin Respir J 12(5):1872–1881
Moorman JE, Zahran H, Truman BI, Molla MT (2011) Control Cfd, Prevention. Current asthma prevalence-United States, 2006–2008. MMWR Surveill Summ 60(Suppl):84–86
Wegmann M (2009) Th2 cells as targets for therapeutic intervention in allergic bronchial asthma. Expert Rev Mol Diagn 9(1):85–100
Wenzel S, Wilbraham D, Fuller R, Getz EB, Longphre M (2007) Effect of an interleukin-4 variant on late phase asthmatic response to allergen challenge in asthmatic patients: results of two phase 2a studies. Lancet 370(9596):1422–1431
Hayashi N, Yoshimoto T, Izuhara K, Matsui K, Tanaka T, Nakanishi K (2007) T helper 1 cells stimulated with ovalbumin and IL-18 induce airway hyperresponsiveness and lung fibrosis by IFN-γ and IL-13 production. Proc Natl Acad Sci 104(37):14765–14770
Sugimoto T, Ishikawa Y, Yoshimoto T, Hayashi N, Fujimoto J, Nakanishi K (2004) Interleukin 18 acts on memory T helper cells type 1 to induce airway inflammation and hyperresponsiveness in a naive host mouse. J Exp Med 199(4):535–545
Bettelli E, Korn T, Oukka M, Kuchroo VK (2008) Induction and effector functions of T H 17 cells. Nature 453(7198):1051
Nakae S, Komiyama Y, Nambu A, Sudo K, Iwase M, Homma I et al (2002) Antigen-specific T cell sensitization is impaired in IL-17-deficient mice, causing suppression of allergic cellular and humoral responses. Immunity 17(3):375–387
Real FG, Svanes C, Björnsson EH, Franklin K, Gislason D, Gislason T et al (2006) Hormone replacement therapy, body mass index and asthma in perimenopausal women: a cross sectional survey. Thorax 61(1):34–40
Barr RG, Wentowski CC, Grodstein F, Somers SC, Stampfer MJ, Schwartz J et al (2004) Prospective study of postmenopausal hormone use and newly diagnosed asthma and chronic obstructive pulmonary disease. Arch Intern Med 164(4):379–386
Hepburn MJ, Dooley DP, Morris MJ (2001) The effects of estrogen replacement therapy on airway function in postmenopausal, asthmatic women. Arch Intern Med 161(22):2717–2720
Townsend EA, Meuchel LW, Thompson MA, Pabelick CM, Prakash Y (2011) Estrogen increases nitric-oxide production in human bronchial epithelium. J Pharmacol Exp Ther 339(3):815–824
Global Initiative for Asthma (GINA) Global strategy for asthma management and prevention, 2017. https://www.ginasthma.org
Endrikat J, Blode H, Gerlinger C, Rosenbaum P, Kuhnz W (2002) A pharmacokinetic study with a low-dose oral contraceptive containing 20 μg ethinylestradiol plus 100 μg levonorgestrel. Eur J Contracept Reprod Health Care 7(2):79–90
Marks MA, Gravitt PE, Burk RD, Studentsov Y, Farzadegan H, Klein SL (2010) Progesterone and 17β-estradiol enhance regulatory responses to human papillomavirus type 16 virus-like particles in peripheral blood mononuclear cells from healthy women. Clin Vaccine Immunol 17(4):609–617
Rao CK, Moore CG, Bleecker E, Busse WW, Calhoun W, Castro M et al (2013) Characteristics of perimenstrual asthma and its relation to asthma severity and control: data from the Severe Asthma Research Program. Chest 143(4):984–992
Kos-Kudła B, Ostrowska Z, Marek B, Ciesielska-Kopacz N, Sieminska L, Kajdaniuk D et al (2000) Hormone replacement therapy in postmenopausal asthmatic women. J Clin Pharm Ther 25(6):461–466
Engelmann F, Rivera A, Park B, Messerle-Forbes M, Jensen JT, Messaoudi I (2016) Impact of estrogen therapy on lymphocyte homeostasis and the response to seasonal influenza vaccine in post-menopausal women. PLoS ONE 11(2):e0149045
Jenkins M, Dharmage S, Flander L, Douglass J, Ugoni A, Carlin J et al (2006) Parity and decreased use of oral contraceptives as predictors of asthma in young women. Clin Exp Allergy 36(5):609–613
Nwaru BI, Sheikh A (2015) Hormonal contraceptives and asthma in women of reproductive age: analysis of data from serial national Scottish Health Surveys. J R Soc Med 108(9):358–371
Macsali F, Real FG, Omenaas ER, Bjorge L, Janson C, Franklin K et al (2009) Oral contraception, body mass index, and asthma: a cross-sectional Nordic-Baltic population survey. J Allergy Clin Immunol 123(2):391–397
Salam MT, Wenten M, Gilliland FD (2006) Endogenous and exogenous sex steroid hormones and asthma and wheeze in young women. J Allergy Clin Immunol 117(5):1001–1007
Burleson MH, Malarkey WB, Cacioppo JT, Poehlmann KM, Kiecolt-Glaser JK, Berntson GG et al (1998) Postmenopausal hormone replacement: effects on autonomic, neuroendocrine, and immune reactivity to brief psychological stressors. Psychosom Med 60(1):17–25
Yang J-H, Chen C-D, Wu M-Y, Chao K-H, Yang Y-S, Ho H-N (2000) Hormone replacement therapy reverses the decrease in natural killer cytotoxicity but does not reverse the decreases in the T-cell subpopulation or interferon-gamma production in postmenopausal women. Fertil Steril 74(2):261–267
Bonds RS, Midoro-Horiuti T (2013) Estrogen effects in allergy and asthma. Curr Opin Allergy Clin Immunol 13(1):92
Cai Y, Zhou J, Webb DC (2012) Estrogen stimulates Th2 cytokine production and regulates the compartmentalisation of eosinophils during allergen challenge in a mouse model of asthma. Int Arch Allergy Immunol 158(3):252–260
Broide DH (2008) Immunologic and inflammatory mechanisms that drive asthma progression to remodeling. J Allergy Clin Immunol 121(3):560–570
Baldaçara RPDC, Silva I (2017) Association between asthma and female sex hormones. Sao Paulo Med J 135(1):4–14
Haghmorad D, Salehipour Z, Nosratabadi R, Rastin M, Kokhaei P, Mahmoudi MB et al (2016) Medium-dose estrogen ameliorates experimental autoimmune encephalomyelitis in ovariectomized mice. J Immunotoxicol 13(6):885–896
Kanda N, Hoashi T, Saeki H (2019) The roles of sex hormones in the course of atopic dermatitis. Int J Mol Sci 20(19):4660
Vock C, Hauber H-P, Wegmann M (2010) The other T helper cells in asthma pathogenesis. J Allergy 2010:519298
Wang W, Cheng ZS, Chen YF, Lin YH (2016) Increased circulating IL-9-producing CD8+ T cells are associated with eosinophilia and high FeNO in allergic asthmatics. Exp Ther Med 12(6):4055–4060
Sehra S, Yao W, Nguyen ET, Glosson-Byers NL, Akhtar N, Zhou B et al (2015) TH9 cells are required for tissue mast cell accumulation during allergic inflammation. J Allergy Clin Immunol 136(2):433–440
Turner H, Kinet J-P (1999) Signalling through the high-affinity IgE receptor FcεRI. Nature 402(suppl 6760):24
Petit-Frere C, Dugas B, Braquet P, Mencia-Huerta J (1993) Interleukin-9 potentiates the interleukin-4-induced IgE and IgG1 release from murine B lymphocytes. Immunology 79(1):146
Koch S, Sopel N, Finotto S (2017) Th9 and other IL-9-producing cells in allergic asthma. Seminars in immunopathology. Springer, Berlin
Maggi E, Parronchi P, Manetti R, Simonelli C, Piccinni M, Rugiu FS et al (1992) Reciprocal regulatory effects of IFN-gamma and IL-4 on the in vitro development of human Th1 and Th2 clones. J Immunol 148(7):2142–2147
Look DC, Rapp SR, Keller BT, Holtzman MJ (1992) Selective induction of intercellular adhesion molecule-1 by interferon-gamma in human airway epithelial cells. Am J Physiol Lung Cell Mol Physiol 263(1):L79–L87
Campbell D, Fryga A, Bol S, Kemp A (1999) Intracellular interferon-gamma (IFN-γ) production in normal children and children with atopic dermatitis. Clin Exp Immunol 115(3):377
Kimura M, Yamaide A, Tsuruta S, Okafuji I, Yoshida T (2002) Development of the capacity of peripheral blood mononuclear cells to produce IL-4, IL-5 and IFN-γ upon stimulation with house dust mite in children with atopic dermatitis. Int Arch Allergy Immunol 127(3):191–197
El-Mezzein R, Matsumoto T, Nomiyama H, Miike T (2001) Increased secretion of IL-18 in vitro by peripheral blood mononuclear cells of patients with bronchial asthma and atopic dermatitis. Clin Exp Immunol 126(2):193–198
Jung T, Witzak K, Dieckhoff K, Zachmann K, Heidrich S, Aversa G et al (1999) IFN-gamma is only partially restored by co-stimulation with IL-12, IL-2, IL-15, IL-18 or engagement of CD28. Clin Exp Allergy 29(2):207–216
Yao Y, Li H, Ding J, Xia Y, Wang L (2017) Progesterone impairs antigen-non-specific immune protection by CD8 T memory cells via interferon-γ gene hypermethylation. PLoS Pathog 13(11):e1006736
Matalka KZ (2003) Riginal AR. Neuroendocrinol Lett 24(3/4):185–191
Xiang Y, Jin Q, Li L, Yang Y, Zhang H, Liu M et al (2018) Physiological low-dose oestrogen promotes the development of experimental autoimmune thyroiditis through the up-regulation of Th1/Th17 responses. J Reprod Immunol 126:23–31
Al-Ramli W, Préfontaine D, Chouiali F, Martin JG, Olivenstein R, Lemière C et al (2009) TH17-associated cytokines (IL-17A and IL-17F) in severe asthma. J Allergy Clin Immunol 123(5):1185–1187
Newcomb DC, Cephus JY, Boswell MG, Fahrenholz JM, Langley EW, Feldman AS et al (2015) Estrogen and progesterone decrease let-7f microRNA expression and increase IL-23/IL-23 receptor signaling and IL-17A production in patients with severe asthma. J Allergy Clin Immunol 136(4):1025–1034
Vélez-Ortega AC, Temprano J, Reneer MC, Ellis GI, McCool A, Gardner T et al (2013) Enhanced generation of suppressor T cells in patients with asthma taking oral contraceptives. J Asthma 50(3):223–230
Adurthi S, Kumar MM, Vinodkumar H, Mukherjee G, Krishnamurthy H, Acharya KK et al (2017) Oestrogen receptor-α binds the FOXP3 promoter and modulates regulatory T-cell function in human cervical cancer. Sci Rep 7(1):17289
Iannello A, Rolla S, Maglione A, Ferrero G, Bardina V, Inaudi I et al (2018) Pregnancy epigenetic signature in T helper 17 and T regulatory cells in multiple sclerosis. Front Immunol 9:3075
Acknowledgements
We would like to express our sincere thanks to all volunteers who participated in this study and donated blood samples.
Funding
This study was supported by Grants # 26562 and 26572 from Iran University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
RB and PB introduced the initial concept, LNS, SAJM and RF designed the project; MF and SAJM introduced the participants including the patients and controls from the clinic; LNS, MK and RF performed the initial experiments and optimized the methods; LNS fulfilled the main experiments; LNS, MF, MK collected data and performed statistical analysis, LNS, RF, SAJM and MK interpreted the findings; LNS wrote the manuscript draft; RF and MK critically reviewed and revised the final draft. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest.
Ethical approval
All subjects gave written consent and the study was approved by the Ethics Committee of Iran University of Medical Sciences (IRIUMS. REC 1398. 9311127007). All experiments were performed in compliance with the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nejatbakhsh Samimi, L., Fallahpour, M., Khoshmirsafa, M. et al. The impact of 17β-estradiol and progesterone therapy on peripheral blood mononuclear cells of asthmatic patients. Mol Biol Rep 48, 297–306 (2021). https://doi.org/10.1007/s11033-020-06046-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-06046-6