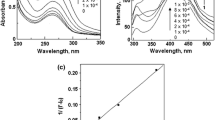
Abstract
Biomolecules that exist in two enantiomeric forms are generally characterized by their physiological action, e.g. l-alanine is a physiological active form of an essential amino acid. The recognition/separation of one of the enantiomers is an important task as they are used as food supplement or pharmacological products where essentially pure enantiomeric forms are required. Therefore, the quantitation of undesirable enantiomers in drug raw material is the challenging task for pharmacists and chemists. Present study demonstrates the differential recognition of l-alanine amino acid by 5,11,17,26-tetrakis-[(N,N-dimethylamino)methyl]-25,26,27,28-tetrahydroxy-calix[4]arene (3). Another characteristic feature of this study is the use of methyl orange as a UV–visible spectrophotometric probe for the determination of stability constant of host–guest inclusion complexes by adopting competitive inclusion method and 1:1 complexation ratio was confirmed by Benesi-Hildebrand equation. Thermodynamics of the recognition have been evaluated that provided the significant distinction for both isomers, i.e. d and l-alanine and it has been deduced that compound 3 may be employed in chromatographic columns for their separation. Thus, the study provides a broad spectrum of its applications in varying fields of analytical and pharmaceutical science.











Similar content being viewed by others

References
Nagata, Y., Tanaka, K., Iida, T.: Occurrence of d-amino acids in a few archaea and dehydrogenase activities in hyperthermophile Pyrobaculum islandicum. Biochim. Biophys. Acta. 1435, 160 (1999)
Chiral amino acids website. http:/www.esainc.com/docs/spool/70-8858-chiral amino acids corona.pdf. Accessed 6 July 2011
Erdemir, S., Tabakci, M., Yilmaz, M.: Synthesis and chiral recognition abilities of new calix[6]arenes bearing amino alcohol moieties. Tetrahedron Asymmetry 17, 1258 (2006)
Lin, J.M., Nakagama, T., Uchiyama, K., Hobo, T.: Capillary electro-chromatographic separation of amino acid enantiomers using on-column prepared molecularly imprinted polymer. J. Pharm. Biomed. Ana. 15, 1358 (1997)
Martin, G.S., Gerald, G.: Enantioseparation by chromatographic and electro-migration techniques using ligand-exchange as chiral separation principle. Anal. Bioanal. Chem. 400, 2305 (2011)
Michael, R., Ran, T.V., Marco, F., Nimrod, Y., Itamar, W.: Stereoselective and chiroselective surface plasmon resonance (SPR) analysis of amino acids by molecularly imprinted Au-nanoparticle composites, chemistry. Eur. J. 16, 7114 (2010)
Shen, G.Y., Gao, Y., Dai, D.S., Cui, J., Liu, Y. M., Pei, L. P.: Kinetics study of chiral recognition between protein and amino acid enantiomers by surface plasmon resonance. Chem. J. Chin. Univ. 8 (2011)
Grobuschek, N., Schmid, M.G., Tuscher, C., Ivanova, M., Gubitz, G.: Chiral separation of methyl-amino acids by ligand exchange using capillary electrophoresis and HPLC. J. Pharm. Biomed. Ana. 27, 599 (2002)
Trikka, F.A., Yoshimatsu, Y., Ye, L., Kyriakidis, D.A.: Molecularly imprinted polymers for histamine recognition in aqueous environment. Amino Acids 43, 2113–2124 (2012)
Kato, M., Dulay, M.Y., Bennett, B., Chen, J., Zare, R.N.: Enantiomeric separation of amino acids and non-proteins amino acids using a particle-loaded monolithic column. Electrophoresis 21, 3145 (2000)
Liu, Y., Qi, A.D., Han, B.H., Li, Y., Zhang, Y.M., Chen, R.T.: Molecular recognition study on supramolecular system (VII). Chin. Sci. Bull. 42, 1189 (1997)
Lakkakula, J., Krause, R.W.M., Ndinteh, D.T., Vijaylakshmi, S.P., Raichur, A.M.: Detailed investigation of a c-cyclodextrin inclusion complex with l-thyroxine for improved pharmaceutical formulations. J. Incl. Phenom. Macrocycl. Chem. 74, 397–405 (2012)
Seyhan, S., Turgut, Yl., Merdivan, M., Hoaygaren, H.: Chiral separation of amino acids using a chiral crown ether by impregnation on a polymeric support and monoamine modified silica gel. Tetrahedron Asymmetry 17, 1700 (2006)
Qureshi, I., Memon, S., Yilmaz, M.: Extraction and binding efficiency of calix[8]arene derivative toward selected transition metals. Pak. J. Anal. Environ. Chem. 9, 96 (2008)
Gutsche, C.D.: Calixarenes revisited. In: Stoddart, J.F. (ed.) Royal society of chemistry, Cambridge (1998)
Yilmaz, M., Memon, S., Tabakci, M., Bartsch, R.A.: Design of polymer appended calixarenes as ion carriers. New frontiers in polymer research, pp. 125–172. Nova Science Publishers, Hauppauge (2006)
Asfari, Z., Bohmer, V., Harrowfield, J., Vicens, J.: Calixarenes 2001. Kluwer Academic Publishers, Dordrecht (2001)
Memon, S., Yilmaz, M., Roundhill, D.M.: Remediation and liquid–liquid phase transfer extraction of chromium(vi). A review. Collect. Czech. Chem. Commun. 69, 1231–1250 (2004)
Dickert, F.L., Haunschild, A.: Sensor materials for solvent vapor detection donor–acceptor and host–guest interactions. Adv. Mater. 5, 887–895 (1993)
Sahin, O., Memon, S., Yilmaz, M.: Synthesis of chiral calix[4]arene derivative and evaluation of its recognition properties. J. Macromol. Sci. 47, 20–25 (2010)
Mutihac, L., Buschmann, H.J., Mutihac, R.C., Schollmeyer, E.: Complexation and separation of amines, amino acids, and peptides by functionalized calix[n]arenes. J. Incl. Phenom. Macrocycl. Chem. 51, 1–10 (2005)
Liu, Y., Han, B.H., Qi, A.D., Chen, R.T.: Molecular recognition study of a supramolecular system XI. Chiral recognition of aliphatic amino acids by natural and modified alpha-cyclodextrins in acidic aqueous solution. Bioorg. Chem. 25, 155–162 (1997)
Gutsche, C.D., Iqbal, M., Stewart, D.: Calixarenes. 18. Synthesis procedures for p-tert-butylcalix[4]arene. J. Org. Chem. 51, 742 (1986)
Gutsche, C.D., Lin, L.G.: Calixarenes. 12. The synthesis of functionalized calixarenes. Tetrahedron 42, 1633–1644 (1986)
Gutsche, C.D., Nam, K.C.: Calixarenes. 22. Synthesis, properties, and metal complexation of aminocalixarenes. J. Am. Chem. Soc. 110, 6153–6162 (1988)
Benesi, H.A., Hildebrand, J.H.: A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 71, 2703 (1949)
Matsui, Y., Fujie, M., Sakate, H.: 1H NMR study of the regioisomers of primary o-di- and trisubstituted α-cyclodextrins. Bull. Chem. Soc. Jpn. 61, 3409–3415 (1988)
Liu, Y., Zhang, Y.M., Qi, A.D., Chen, R.T., Yamamoto, K., Wada, T., Inoue, Y.: Molecular recognition study on a supramolecular system. 10. Inclusion complexation of modified α-cyclodextrins with amino acids: enhanced enantioselectivity for l/d-leucine. J. Org. Chem. 62, 1826–1830 (1997)
Liu, Y., Han, B.H., Li, B., Zhang, Y.M., Zhao, P., Chen, Y.T., Wada, T., Inoue, Y.: Molecular recognition study on supramolecular system. 14. Synthesis of modified cyclodextrins and their inclusion complexation thermodynamics with l-tryptophan and some naphthalene derivatives. J. Org. Chem. 63, 1444–1454 (1998)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Memon, F.N., Memon, S. Differential recognition of d and l-alanine by calix[4]arene amino derivative. J Incl Phenom Macrocycl Chem 77, 413–420 (2013). https://doi.org/10.1007/s10847-012-0261-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-012-0261-2