Summary
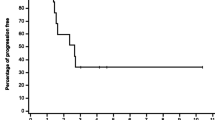
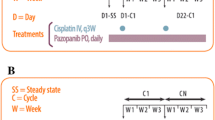
Purpose This study intended to determine the maximum tolerated dose (MTD) and the recommended phase II dose (RD) of trabectedin combined with carboplatin in patients with advanced solid tumors. Patients and methods Carboplatin-pretreated patients received carboplatin AUC 4 (Group 1), whereas carboplatin-naïve patients received carboplatin AUC 5 (Group 2) as a 1-h i.v. infusion followed by trabectedin at dose range from 0.5–1.2 mg/m2 in the schedule of 3-h/every-3-weeks. Pharmacokinetic (PK) sampling was performed in the first 2 cycles. Results Forty-four patients were treated and evaluable for safety and dose-limiting toxicities (DLTs). In Group 1, at trabectedin 1.0 mg/m2, cumulative hematological toxicity was found in all patients and 1/10 patients had DLTs. The RD was considered trabectedin 0.8 mg/m2 combined with carboplatin AUC 4. Although no DLT occurred at this dose level, frequent dose delays (28.6%) and the 4-week cycle re-scheduling (66.7%) were required. In Group 2, DLTs occurred at trabectedin 0.8 mg/m2 (3/8 patients), 1.0 mg/m2 (3/10 patients) and 1.2 mg/m2 (2/2 patients) with cumulative hematological toxicity associated with an important number of transfusions. In this group, neither the MTD nor the RD were established. Promising antitumor activity was found for this carboplatin/trabectedin combination; especially in patients with advanced ovarian cancer and soft tissue sarcoma. No significant PK drug–drug interaction occurred. Conclusions This study established a trabectedin dose of 0.8 mg/m2 combined with carboplatin AUC 4 and given every 4 weeks as the most feasible schedule in carboplatin-pretreated patients. Dose and cycle recommendations for carboplatin-naïve patients warrant further evaluation.

Similar content being viewed by others
References
D’Incalci M, Erba E, Damia G, Galliera E, Carrassa L, Marchini S, Mantovani R, Tognon G, Fruscio R, Jimeno J, Faircloth GT (2002) Unique features of the mode of action of ET-743. Oncologist 7:210–216
Pommier Y, Kohlhagen G, Bailly C, Waring M, Mazumder A, Kohn KW (1996) DNA sequence- and structure-selective alkylation of guanine N2 in the DNA minor groove by ecteinascidin 743, a potent antitumor compound from the Caribbean tunicate Ecteinascidia turbinata. Biochemistry 35:13303–13309
Zewail-Foote M, Hurley LH (1999) Ecteinascidin 743: a minor groove alkylator that bends DNA toward the major groove. J Med Chem 42:2493–2497
Martinez EJ, Corey EJ, Owa T (2001) Antitumor activity- and gene expression-based profiling of ecteinascidin Et 743 and phthalascidin Pt 650. Chem Biol 8:1151–1160
Erba E, Muratore I, Tiozzo G (2001) ET-743 and cisplatin (DDP) show in vitro and in vivo synergy against human sarcoma and ovarian carcicoma cell lines. In: AACR-NCI-EORTC Conference on “Molecular targets and cancer therapeutics”; October 29–November 1; Miami
Damia G, Silvestri S, Carrassa L, Filiberti L, Faircloth GT, Liberi G, Foiani M, D’Incalci M (2001) Unique pattern of ET-743 activity in different cellular systems with defined deficiencies in DNA-repair pathways. Int J Cancer 92:583–588
Demetri GD, Chawla SP, von Mehren M, Ritch P, Baker LH, Blay JY, Hande KR, Keohan ML, Samuels BL, Schuetze S, Lebedinsky C, Elsayed YA, Izquierdo MA, Gomez J, Park YC, Le Cesne A (2009) Efficacy and safety of trabectedin in patients with advanced or metastatic liposarcoma or leiomyosarcoma after failure of prior anthracyclines and ifosfamide: results of a randomized phase II study of two different schedules. J Clin Oncol 27:4188–4196
Monk BJ, Herzog T, Kaye S, Krasner CN, Vermorken JB, Muggia F, Pujade-Lauraine E, Renshaw FG, Lebedinsky C, Poveda A (2008) A Randomized phase III study of trabectedin with pegylated liposomal doxorubicin (PLD) vs PLD in relapsed, recurrent ovarian cancer. Ann Oncol 19 (Supl 8):33rd ESMO Congress, Stockholm, Sweden 12–16 September 2008
Martin LP, Hamilton TC, Schilder RJ (2008) Platinum resistance: the role of DNA repair pathways. Clin Cancer Res 14:1291–1295
Shah N, Dizon DS (2009) New-generation platinum agents for solid tumors. Future Oncol 5:33–42
Lebwohl D, Canetta R (1998) Clinical development of platinum complexes in cancer therapy: an historical perspective and an update. Eur J Cancer 34:1522–1534
Damia G, D’Incalci M (2007) Targeting DNA repair as a promising approach in cancer therapy. Eur J Cancer 43:1791–1801
D’Incalci M, Colombo T, Ubezio P, Nicoletti I, Giavazzi R, Erba E, Ferrarese L, Meco D, Riccardi R, Sessa C, Cavallini E, Jimeno J, Faircloth GT (2003) The combination of yondelis and cisplatin is synergistic against human tumor xenografts. Eur J Cancer 39:1920–1926
Sessa C, Cresta S, Noberasco C, Capri G, Gallerani E, De Braud F, Zucchetti M, D’Incalci M, Locatelli A, Marsoni S, Corradino I, Minoia C, Zintl P, Gianni L (2009) Phase I clinical and pharmacokinetic study of trabectedin and cisplatin in solid tumours. Eur J Cancer 45:2116–2122
van Kesteren C, Cvitkovic E, Taamma A, Lopez-Lazaro L, Jimeno JM, Guzman C, Math RA, Schellens JH, Misset JL, Brain E, Hillebrand MJ, Rosing H, Beijnen JH (2000) Pharmacokinetics and pharmacodynamics of the novel marine-derived anticancer agent ecteinascidin 743 in a phase I dose-finding study. Clin Cancer Res 6:4725–4732
Twelves C, Hoekman K, Bowman A, Vermorken JB, Anthoney A, Smyth J, van Kesteren C, Beijnen JH, Uiters J, Wanders J, Gomez J, Guzman C, Jimeno J, Hanauske A (2003) Phase I and pharmacokinetic study of Yondelis (Ecteinascidin-743; ET-743) administered as an infusion over 1 h or 3 h every 21 days in patients with solid tumours. Eur J Cancer 39:1842–1851
Calvert AH, Newell DR, Gumbrell LA, O’Reilly S, Burnell M, Boxall FE, Siddik ZH, Judson IR, Gore ME, Wiltshaw E (1989) Carboplatin dosage: prospective evaluation of a simple formula based on renal function. J Clin Oncol 7:1748–1756
Gralla R, Osoba D, Kris M, Kirkbride P, Hesketh PJ, Chinnery LW, Clark-Snow R, Gill DP, Groshen S, Grunberg S, Koeller JM, Morrow GR, Perez EA, Silber JH, Pfister DG (1999) Recommendations for the use of antiemetics: evidence-based, clinical practice guidelines. American Society of Clinical Oncology. J Clin Oncol 17:2971–2994
Ozer H, Armitage JO, Bennett CL, Crawford J, Demetri GD, Pizzo PA, Schiffer CA, Smith TJ, Somlo G, Wade JC, Wade JL III, Winn RJ, Wozniak AJ, Somerfield MR, American Society of Clinical Oncology (2000) Update of recommendations for the use of hematopoietic colony-stimulating factors: evidence-based, clinical practice guidelines. J Clin Oncol 18:3558–3585
National Cancer Institute (1999) Guidelines for the reporting of adverse drug reactions. MD, Division of Cancer Treatment, National Cancer Institute 1–80
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Rosing H, Hillebrand MJ, Jimeno JM, Gomez A, Floriano P, Faircloth G, Henrar RE, Vermorken JB, Cvitkovic E, Bult A, Beijnen JH (1998) Quantitative determination of Ecteinascidin 743 in human plasma by miniaturized high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry. J Mass Spectrom 33:1134–1140
van Warmerdam LJ, van Tellingen O, Maes RA, Bijnen JH (1995) Validated method for the determination of carboplatin in biological-fluids by Zeeman atomic-absoption spectrometry. Fresnius J Anal Chem 351:777–781
Rosing H, Cvitkovic E, Jimeno JM, Hillebrand MJ, Twelves C, Villalona M, Beijnen JH (1999) Pharmacokinetics of ecteinaiscidin-743 (ET-743) in three different phase I trials. American Association for Cancer Research (AACR), 90th Annual Meeting, Philadelphia Abstract #542
van Acker BA, Prummel MF, Weber JA, Wiersinga WM, Arisz L (1993) Effect of prednisone on renal function in man. Nephron 65:254–259
Fleck C (1999) Determination of the glomerular filtration rate (GFR): methodological problems, age-dependence, consequences of various surgical interventions, and the influence of different drugs and toxic substances. Physiol Res 48:267–279
Obasaju CK, Johnson SW, Rogatko A, Kilpatrick D, Brennan JM, Hamilton TC, Ozols RF, O’Dwyer PJ, Gallo JM (1996) Evaluation of carboplatin pharmacokinetics in the absence and presence of paclitaxel. Clin Cancer Res 2:549–552
Sessa C, De Braud F, Perotti A, Bauer J, Curigliano G, Noberasco C, Zanaboni F, Gianni L, Marsoni S, Jimeno J, D’Incalci M, Dall’o E, Colombo N (2005) Trabectedin for women with ovarian carcinoma after treatment with platinum and taxanes fails. J Clin Oncol 23:1867–1874
Paz-Ares L, Rivera-Herreros F, Diaz-Rubio E, Garcia M, Casado E, Cubedo R, Gravalos C, Alfaro V, Gomez J, Izquierdo MA, Tabernero J (2007) Phase II study of trabectedin in pretreated patients with advanced colorectal cancer. Clin Colorectal Cancer 6:522–528
Twelves C, Hoekman K, Bowman A, Vermorken JB, Anthoney A, Smyth J, Van Kesteren C, Beijnen JH, Uiters J, Wanders J, Gomez J, Guzmán C, Jimeno J, Hanauske A (2003) Phase I and pharmacokinetic study of Yondelis TM (Ecteinascidin-743; ET-743) administered as an infusion over 1 h or 3 h every 21 days in patients with solid tumours. Eur J Cancer 39:1842–1851
Le Cesne A, Domont J, Cioffi A, Bonvalot S, Terrier P, Ray-Coquard I, Alfaro V, Lebedinsky C, Santabarbara P, Blay JY (2009) Mapping the literature: role of trabectedin as a new chemotherapy option in advanced pretreated soft tissue sarcoma. Drugs Today (Barc) 45:403–421
von Mehren M, Schilder RJ, Cheng JD, Temmer E, Cardoso TM, Renshaw FG, Bayever E, Zannikos P, Yuan Z, Cohen RB (2008) A phase I study of the safety and pharmacokinetics of trabectedin in combination with pegylated liposomal doxorubicin in patients with advanced malignancies. Ann Oncol 19:1802–1809
Blay JY, von Mehren M, Samuels BL, Fanucchi MP, Ray-Coquard I, Buckley B, Gilles L, Lebedinsky C, Elsayed YA, Le Cesne A (2008) Phase I combination study of trabectedin and doxorubicin in patients with soft-tissue sarcoma. Clin Cancer Res 14:6656–6662
Jodrell DI, Egorin MJ, Canetta RM, Langenberg P, Goldbloom EP, Burroughs JN, Goodlow JL, Tan S, Wiltshaw E (1992) Relationships between carboplatin exposure and tumor response and toxicity in patients with ovarian cancer. J Clin Oncol 10:520–528
Sederholm C, Hillerdal G, Lamberg K, Kolbeck K, Dufmats M, Westberg R, Gawande SR (2005) Phase III trial of gemcitabine plus carboplatin versus single-agent gemcitabine in the treatment of locally advanced or metastatic non-small-cell lung cancer: the Swedish Lung Cancer Study Group. J Clin Oncol 23:8380–8388
Kose MF, Sufliarsky J, Beslija S, Saip P, Tulunay G, Krejcy K, Minarik T, Fitzthum E, Hayden A, Melemed A (2005) A phase II study of gemcitabine plus carboplatin in platinum-sensitive, recurrent ovarian carcinoma. Gynecol Oncol 96:374–380
George S, Maki RG, Harmon D, Supko J, Morgan J, Seiden M, Buonanno M, Griffin J, Moutsiolis A, Lopez T, Guzman C, Jimeno J, Demetri GD (2002) Phase II study of ecteinascidin-743 (ET-743) given by 3-hour i.v. infusion in patients with soft tissue sarcomas (STS) failing prior chemotherapies. Proc Am Soc Clin Oncol 21:408a (Abstr)
Dileo P, Casali PG, Bacci G, Casanova M, Aglietta M, Ferrari S, Grignani G, Sessa C, Jimeno J, Dall’O’ E, Marsoni S, Gianni L, Ruiz A (2002) Phase II evaluation of 3-hr infusion ET-743 in patients with recurrent sarcomas. Proc Am Soc Clin Oncol 21:408 (abstract 1628)
Acknowledgments
The authors would like to acknowledge the work done by Adnan Tanović (medical writer, Pharma Mar) in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vidal, L., Magem, M., Barlow, C. et al. Phase I clinical and pharmacokinetic study of trabectedin and carboplatin in patients with advanced solid tumors. Invest New Drugs 30, 616–628 (2012). https://doi.org/10.1007/s10637-010-9559-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-010-9559-3