Abstract
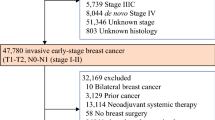
Young women with breast cancer (BC) have a worse survival partly due to more aggressive tumor characteristics; however, their response to chemotherapy seems better. We investigated to what extent the prognostic factor pathological complete remission (pCR) after neoadjuvant chemotherapy is applicable to young women. 8949 patients with primary BC and follow-up from eight German neoadjuvant trials were included. A subgroup of 1453 patients <40 years was compared with women aged 40–49 and ≥50 years regarding pCR (ypT0 ypN0), as well as disease free survival (DFS), local recurrence free survival (LRFS), distant disease free survival (DDFS), and overall survival (OS) overall, according to pCR status and subtypes defined by hormone-receptor (HR) status and HER2. pCR was strongly associated with age without a clear age cut-off. The pCR rate was significantly higher in the young compared with other age groups (20.9 vs. 17.7 vs. 13.7 %; p < 0.001). This difference was confined to triple-negative breast cancer (TNBC) and HR +/HER2−. DFS, DDFS, LRFS, and OS were significantly worse for young women. Age was independently prognostic for survival in HR +/HER2−, with women <40 years without pCR having a worse DFS compared to their counterparts with pCR. Young women are more likely to achieve pCR after neoadjuvant chemotherapy, especially in HR +/HER2− and TNBC. Age is not an important prognostic factor in TNBC and HR−/HER2 + but is in HR +/HER2−. Young women with a luminal-like BC seem to benefit more from neoadjuvant chemotherapy than older women, which needs to be taken into account.




Similar content being viewed by others
References
Eisemann N, Waldmann A, Katalinic A (2013) Epidemiology of breast cancer, current figures and trends. Geburtsh Frauenheilk 73:130–135
DeSantis C, Siegel R, Bandi P, Jemal A (2011) Breast cancer statistics 2011. CA Cancer J Clin 61:409–418
Adami HO, Malker B, Holmberg L, Persson I, Stone B (1986) The relation between survival and age at diagnosis in breast cancer. N Engl J Med 315:559–563
Anders CK, Hsu DS, Broadwater G, Acharya CR, Foekens JA, Zhang Y et al (2008) Young age at diagnosis correlates with worse prognosis and defines a subset of breast cancers with shared patterns of gene expression. J Clin Oncol 26:3324–3330
Fredholm H, Eaker S, Frisell J, Holmberg L, Fredriksson I, Lindman H (2009) Breast cancer in young women: poor survival despite intensive treatment. PLoS One 4:e7695
Colleoni M, Rotmensz N, Robertson C, Orlando L, Viale G, Renne G et al (2002) Very young women (<35 years) with operable breast cancer: features of disease at presentation. Ann Oncol 13:273–279
Maggard MA, O’Connell JB, Lane KE, Liu JH, Etzioni DA, Ko CY (2003) Do young breast cancer patients have worse outcomes? J Surg Res 113:109–113
Dubsky PC, Gnant MF, Taucher S, Roka S, Kandioler D, Pichler-Gebhard B et al (2002) Young age as an independent adverse prognostic factor in premenopausal patients with breast cancer. Clin Breast Cancer 3:65–72
Huober J, von Minckwitz G, Denkert C, Tesch H, Weiss E, Zahm DM et al (2010) Effect of neoadjuvant anthracycline-taxane-based chemotherapy in different biological breast cancer phenotypes: overall results from the GeparTrio study. Breast Cancer Res Treat 124:133–140
von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA et al (2012) Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol 30:1796–1804
Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N et al (2014) Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet 384:164–172
Cardoso F, Loibl S, Pagani O, Graziottin A, Panizza P, Martincich L et al (2012) The european society of breast cancer specialists recommendations for the management of young women with breast cancer. Eur J Cancer 48:3355–3377
Cardoso F, Harbeck N (2012) Breast cancer in young women–a clinical challenge to be addressed in a multidisciplinary setting. Breast Care 7:193–194
von Minckwitz G, Raab G, Caputo A, Schütte M, Hilfrich J, Blohmer JU et al (2005) Doxorubicin with cyclophosphamide followed by docetaxel every 21 days compared with doxorubicin and docetaxel every 14 days as preoperative treatment in operable breast cancer: the GEPARDUO study of the German Breast Group. J Clin Oncol 23:2676–2685
von Minckwitz G, Blohmer JU, Raab G, Löhr A, Gerber B, Heinrich G et al (2005) In vivo chemosensitivity-adapted preoperative chemotherapy in patients with early-stage breast cancer: the GEPARTRIO pilot study. Ann Oncol 16:56–63
von Minckwitz G, Kümmel S, Vogel P, Hanusch C, Eidtmann H, Hilfrich J et al (2008) Intensified neoadjuvant chemotherapy in early-responding breast cancer: phase III randomized GeparTrio study. J Natl Cancer Inst 100:552–562
von Minckwitz G, Kümmel S, Vogel P, Hanusch C, Eidtmann H, Hilfrich J et al (2008) Neoadjuvant vinorelbine-capecitabine versus docetaxel-doxorubicin-cyclophosphamide in early nonresponsive breast cancer: phase III randomized GeparTrio trial. J Natl Cancer Inst 100:542–551
Untch M, Rezai M, Loibl S, Fasching PA, Huober J, Tesch H et al (2010) Neoadjuvant treatment with trastuzumab in HER2-positive breast cancer: results from GeparQuattro study. J Clin Oncol 28:2024–2031
Untch M, Möbus V, Kuhn W, Muck BR, Thomssen C, Bauerfeind I et al (2009) Intensive dose-dense compared with conventionally scheduled preoperative chemotherapy for high-risk primary breast cancer. J Clin Oncol 27:2938–2945
Untch M, Fasching PA, Konecny GE, von Koch F, Conrad U, Fett W et al (2011) PREPARE trial: a randomized phase III trial comparing preoperative, dose-dense, dose-intensified chemotherapy with epirubicin, paclitaxel and CMF versus a standard-dosed epirubicin/cyclophosphamide followed by paclitaxel ± darbepoetin alfa in primary breast cancer–results at the time of surgery. Ann Oncol 22:1988–1998
Untch M, von Minckwitz G, Konecny GE, Conrad U, Fett W, Kurzeder C et al (2011) PREPARE trial: a randomized phase III trial comparing preoperative, dose-dense, dose-intensified chemotherapy with epirubicin, paclitaxel, and CMF versus a standard-dosed epirubicin-cyclophosphamide followed by paclitaxel with or without darbepoetin alfa in primary breast cancer–outcome on prognosis. Ann Oncol 22:1999–2006
Untch M, Fasching PA, Konecny GE, Hasmüller S, Lebeau A, Kreienberg R et al (2011) Pathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: results from the TECHNO trial of the AGO and GBG study groups. J Clin Oncol 29:3351–3357
von Minckwitz G, Untch M, Nüesch E, Loibl S, Kaufmann M, Kümmel S et al (2011) Impact of treatment characteristics on response of different breast cancer phenotypes: pooled analysis of the German neo-adjuvant chemotherapy trials. Breast Cancer Res Treat 125:145–156
von Minckwitz G, Eidtmann H, Rezai M, Fasching PA, Tesch H, Eggemann H et al (2012) Neoadjuvant chemotherapy and bevacizumab for HER2-negative breast cancer. N Engl J Med 366:299–309
Untch M, Loibl S, Bischoff J, Eidtmann H, Kaufmann M, Blohmer JU et al (2012) Lapatinib versus trastuzumab in combination with neoadjuvant anthracycline-taxane-based chemotherapy (GeparQuinto, GBG 44): a randomised phase 3 trial. Lancet Oncol 13:135–144
von Minckwitz G, Costa SD, Raab G, Blohmer JU, Eidtmann H, Hilfrich J et al (2001) Dose-dense doxorubicin, docetaxel, and granulocyte colony-stimulating factor support with or without tamoxifen as preoperative therapy in patients with operable carcinoma of the breast: a randomized, controlled, open phase IIb study. J Clin Oncol 19:3506–3515
http://www.ago-online.de/fileadmin/downloads/leitlinien/mamma/march2012/14_2012E_Breast_Cancer_Specific_Situations.pdf Accessed 02 Feb 2015
http://www.ago-online.de/fileadmin/downloads/leitlinien/mamma/march2012/13_2012E_Adjuvant_Radiotherapy.pdf Accessed 02 Feb 2015
Bonetti M, Gelber RD (2004) Patterns of treatment effects in subsets of patients in clinical trials. Biostatistics 5:465–481
Arvold ND, Taghian AG, Niemierko A, Abi Raad RF, Sreedhara M, Nguyen PL et al (2011) Age, breast cancer subtype approximation, and local recurrence after breast-conserving therapy. J Clin Oncol 29:3885–3891
Azim HA Jr, Michiels S, Bedard PL, Singhal SK, Criscitiello C, Ignatiadis M et al (2012) Elucidating prognosis and biology of breast cancer arising in young women using gene expression profiling. Clin Cancer Res 18:1341–1351
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG, Peto R, Davies C, Godwin J, Gray R, Pan HC, Clarke M et al (2012) Comparisons between different polychemotherapy regimens for early breast cancer: meta-analyses of long-term outcome among 100,000 women in 123 randomised trials. Lancet 379:432–444
Partridge AH, Gelber S, Piccart-Gebhart MJ, Focant F, Scullion M, Holmes E et al (2013) Effect of age on breast cancer outcomes in women with human epidermal growth factor receptor 2-positive breast cancer: results from a herceptin adjuvant trial. J Clin Oncol 31:2692–2698
Ahn SH, Son BH, Kim SW, Kim SI, Jeong J, Ko SS et al (2007) Poor outcome of hormone receptor-positive breast cancer at very young age is due to tamoxifen resistance: nationwide survival data in Korea–a report from the Korean Breast Cancer Society. J Clin Oncol 25:2360–2368
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), Godwin J, Gray R, Clarke M, Cutter D, Darby S et al (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378:771–784
Gnant M, Mlineritsch B, Schippinger W, Luschin-Ebengreuth G, Pöstlberger S, Menzel C et al (2009) Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med 360:679–691
Francis PA, Regan MM, Fleming GF, Láng I, Ciruelos E, Bellet M et al (2015) Adjuvant ovarian suppression in premenopausal breast cancer. N Engl J Med 372:436–446
Kroman N, Jensen MB, Wohlfahrt J, Mouridsen HT, Andersen PK, Melbye M (2000) Factors influencing the effect of age on prognosis in breast cancer: population based study. BMJ 320:474–478
Brouckaert O, Salihi R, Laenen A, Vanderhaegen J, Amant F, Leunen K et al (2012) Independent prognostic value of age depends on breast cancer subtype. Cancer Res 72(24):7–29
Liedtke C, Hess KR, Karn T, Rody A, Kiesel L, Hortobagyi GN et al (2013) The prognostic impact of age in patients with triple-negative breast cancer. Breast Cancer Res Treat 138:591–599
Acknowledgments
We would like to thank all patients, investigators, and study personnel who supported the trials and the German Breast Group for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Following authors declare that they have no conflict of interest: Sibylle Loibl, Bianca Lederer, Michael Untch, Stefan Paepke, Andreas Schneeweiss, Jörn Hilfrich, Bernd Gerber, Holger Eidtmann, Carsten Denkert, Serban Dan Costa, Jens-Uwe Blohmer, Valentina Nekljudova, Keyur Mehta, Gunter von Minckwitz. Following authors declare a conflict of interest: Sherko Kümmel, consultant/advisory role: Roche AG Jens Huober, consultant/advisory role: Sanofi Aventis, Roche, GSK, Novartis Claus Hanusch, consultant/advisory role: Novartis, Pfizer, Amgen, Roche Carsten Denkert, stock ownership: Sividon Diagnostics Germany.
Informed consent
All patients provided written informed consent for study participation and data collection.
Research involving human participants
All trials were approved by the relevant ethics committees.
Additional information
On behalf of the German Breast Group (GBG) and the Arbeitsgemeinschaft Gynäkologische Onkologie-Breast (AGO-B) study groups.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Loibl, S., Jackisch, C., Lederer, B. et al. Outcome after neoadjuvant chemotherapy in young breast cancer patients: a pooled analysis of individual patient data from eight prospectively randomized controlled trials. Breast Cancer Res Treat 152, 377–387 (2015). https://doi.org/10.1007/s10549-015-3479-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3479-z