Abstract
Objectives
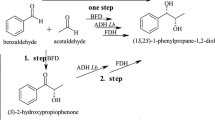
To investigate the efficiency of a new cascade biocatalysis system for the conversion of R, S-β-amino alcohols to enantiopure vicinal diol and β-amino alcohol.
Results
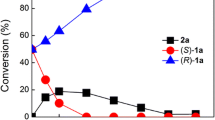
An efficient cascade biocatalysis was achieved by combination of a transaminase, a carbonyl reductase and a cofactor regeneration system. An ee value of > 99% for 2-amino-2-phenylethanol and 1-phenyl-1, 2-ethanediol were simultaneously obtained with 50% conversion from R, S-2-amino-2-phenylethanol. The generality of the cascade biocatalysis was further demonstrated with the whole-cell approaches to convert 10–60 mM R, S-β-amino alcohol to (R)- and (S)-diol and (R)- and (S)-β-amino alcohol in 90–99% ee with 50–52% conversion. Preparative biotransformation was demonstrated at a 50 ml scale with mixed recombinant cells to give both (R)- and (S)-2-amino-2-phenylethanol and (R)- and (S)-1-phenyl-1, 2-ethanediol in > 99% ee and 40–42% isolated yield from racemic 2-amino-2-phenylethanol.
Conclusions
This cascade biocatalysis system provides a new practical method for the simultaneous synthesis of optically pure vicinal diol and an β-amino alcohol.


Similar content being viewed by others
References
Bendjeddou LZ, Loaëc N, Villiers B, Prina E, Späth G, Galons H, Meijer L, Oumata N (2017) Exploration of the imidazo [1, 2-b] pyridazine scaffold as a protein kinase inhibitor. Eur J Med Chem 125:696–709
Birrell JA, Jacobsen EN (2013) A practical method for the synthesis of highly enantioenriched trans-1, 2-amino alcohols. Org Lett 15:2895–2897
Bosetti A, Bianchi D, Cesti P, Golini P, Spezia S (1992) Enzymatic resolution of 1, 2-diols comparison between hydrolysis and transesterification reactions. J Chem Soc Perkin Trans 1:2395–2398
Cao L, Lee JT, Chen W, Wood TK (2006) Enantioconvergent production of (R)-1-phenyl-1, 2-ethanediol from styrene oxide by combining the Solanum tuberosum and an evolved Agrobacterium radiobacter AD1 epoxide hydrolases. Biotechnol Bioeng 94:522–529
Chen FF, Liu YY, Zheng GW, Xu JH (2015) Asymmetric amination of secondary alcohols by using a redox-neutral two-enzyme cascade. ChemCatChem 7:3838–3841
Cui ZM, Zhang JD, Fan XJ, Zheng GW, Chang HH, Wei WL (2017) Highly efficient bioreduction of 2-hydroxyacetophenone to (S)- and (R)-1-phenyl-1, 2-ethanediol by two substrate tolerance carbonyl reductases with cofactor regeneration. J Biotechnol 243:1–9
Dobrikov GM, Valcheva V, Stoilova-Disheva M, Momekov G, Tzvetkova P, Chimov A, Dimitrov V (2012) Synthesis and in vitro antimycobacterial activity of compounds derived from (R)- and (S)-2-amino-1-butanol—the crucial role of the configuration. Eur J Med Chem 48:45–56
Höhne M, Schätzle S, Jochens H, Robins K, Bornscheuer UT (2010) Rational assignment of key motifs for function guides in silico enzyme identification. Nat Chem Biol 6:807–813
Iwasaki F, Maki T, Onomura O, Nakashima W, Matsumura Y (2000) Chemo- and stereoselective monobenzoylation of 1, 2-diols catalyzed by organotin compounds. J Org Chem 65:996–1002
Kaulmann U, Smithies K, Smith MEB, Hailes HC, Ward JM (2007) Substrate spectrum of ω-transaminase from Chromobacterium violaceum DSM30191 and its potential for biocatalysis. Enzym Microb Technol 41:628–637
Kumar P, Upadhyay RK, Pandey RK (2004) Asymmetric dihydroxylation route to (R)-isoprenaline, (R)-norfluoxetine and (R)-fluoxetine. Tetrahedron Asymmetry 15:3955–3959
McKennon MJ, Meyers AI (1993) A convenient reduction of amino acids and their derivatives. J Org Chem 58:3568–3571
Mutti FG, Fuche CS, Pressnitz D, Turrini NG, Sattler JH, Lerchner A, Skerra A, Kroutil W (2012) Amination of ketones by employing two new (S)-selective ω-transaminases and the His-tagged ω-TA from Vibrio fluvialis. Eur J Org Chem 2012:1003–1007
Panek JS, Masse CE (1998) An improved synthesis of (4S, 5S)-2-phenyl-4-(methoxycarbonyl)-5-isopropyloxazoline from (S)-phenylglycinol. J Org Chem 63:2382–2384
Pavlidis IV, Weib MS, Genz M, Spurr P, Hanlon SP, Wirz B, Iding H, Bornscheuer UT (2016) Identification of (S)-selective transaminases for the asymmetric synthesis of bulky chiral amines. Nat Chem 8:1076–1082
Rouf A, Gupta P, Aga MA, Kumar B, Parshad R, Taneja SC (2011) Cyclic trans-β-amino alcohols: preparation and enzymatic kinetic resolution. Tetrahedron Asymmetry 22:2134–2143
Schrittwieser JH, Velikogne S, Hall M, Kroutil W (2017) Artificial biocatalytic linear cascades for preparation of organic molecules. Chem Rev. https://doi.org/10.1021/acs.chemrev.7b00033
Shimada T, Mukaide K, Shinohara A, Han JW, Hayashi T (2002) Asymmetric synthesis of 1-aryl-1, 2-ethanediols from arylacetylenes by palladium-catalyzed asymmetric hydrosilylation as a key step. J Am Chem Soc 124:1584–1585
Shin G, Mathew S, Yun H (2015) Kinetic resolution of amines by (R)-selective omega-transaminase from Mycobacterium vanbaalenii. J Ind Eng Chem 23:128–133
Tsujigami T, Sugai T, Ohta H (2001) Microbial asymmetric reduction of α-hydroxy ketones in the anti-Prelog selectivity. Tetrahedron Asymmetry 12:2543–2549
Zhang JD, Xu TT, Li Z (2013) Enantioselective biooxidation of racemic trans-cyclic vicinal diols: one-pot synthesis of both enantiopure (S, S)-cyclic vicinal diols and (R)-α-hydroxy ketones. Adv Synth Catal 16:3147–3153
Zhang JD, Wu HL, Meng T, Zhang CF, Fan XJ, Chang HH, Wei WL (2017) A high-throughput microtiter plate assay for the discovery of active and enantioselective amino alcohol-specific transaminases. Anal Biochem 518:94–101
Acknowledgements
This study was financially supported by the National Natural Science Foundation of China (Grant No. 21772141), the Shanxi Province Science Foundation for Youths (Grant No. 201701D221042) and the Open Funding Project of the State Key Laboratory of Bioreactor Engineering.
Supporting information
Supplementary Table 1—Primers used for TAm cloning
Supplementary Fig. 1—The effect of 2-amino-2-phenylethanol on ωTAm activity
Supplementary Fig. 2—The effect of pyruvate on ωTAm activity
Supplementary Fig. 3—The effect of Ala on ωTAm activity
Supplementary Fig. 4—The effect of 2-hydroxyacetophenone on TAm activity
Supplementary Fig. 5—The effect of 2-amino-2-phenylethanol on ADH activity
Supplementary Fig. 6—Chiral GC analysis of 2-amino-2-phenylethanol (1a) standard
Supplementary Fig. 7—Chiral GC analysis of 2-amino-2-phenylethanol (1a) obtained via cascade biocatalysis with the resting cells of E. coli (TAm) and E. coli (CR–GDH)
Supplementary Fig. 8—Chiral GC analysis of 2-amino-1-butanol (1b)
Supplementary Fig. 9—Chiral GC analysis of 2-amino-1-butanol (1b) obtained via cascade biocatalysis with the resting cells of E. coli (TAm) and E. coli (CR–GDH)
Supplementary Fig. 10—Chiral GC analysis of 1-phenyl-1, 2-ethanediol (3a) standard
Supplementary Fig. 11—Chiral GC analysis of 1-phenyl-1, 2-ethanediol (3a) obtained via cascade biocatalysis with the resting cells of E. coli (TAm) and E. coli (CR–GDH)
Supplementary Fig. 12—Chiral GC analysis of 1, 2-butanediol (3b) standard
Supplementary Fig. 13—Chiral GC analysis of 1, 2-butanediol (3b) obtained via cascade biocatalysis with the resting cells of E. coli (TAm) and E. coli (CR–GDH)
Supplementary Fig. 14—Chiral GC analysis of 3b obtained via 2b asymmetric reduction with the resting cells of E. coli (GoSCR–GDH)
Supplementary Fig. 15—The purity analysis of 1a and 3a
Supplementary Fig. 16—1H NMR spectra analysis of chiral 2-amino-2-phenylethanol
Supplementary Fig. 17—1H NMR spectra analysis of chiral 1-phenyl-1, 2-ethanediol
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, JW., Wu, HL., Zhang, JD. et al. One pot simultaneous preparation of both enantiomer of β-amino alcohol and vicinal diol via cascade biocatalysis. Biotechnol Lett 40, 349–358 (2018). https://doi.org/10.1007/s10529-017-2471-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-017-2471-6