Abstract
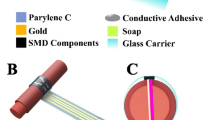
Personalized mobile medicine will continue to advance through the development of wearable sensors that can wirelessly provide pertinent health information while remaining unobtrusive, comfortable, low cost, and easy to operate and interpret. It is the intention that the sensor presented hereafter can contribute to such innovation. By applying a combination of emerging microfluidic and electronic technologies, a miniature, flexible, transparent, highly sensitive and wearable pressure sensor with microfluidic elements has been implemented, referred to as a microflotronic device. High sensitivity of 0.1 kPa−1 and fast response time on the order of tens of milliseconds has been achieved on the microflotronic sensor design. Its sensitivity is among the highest in impedance-based flexible pressure sensors. Once configured into an array, the transparent device can be easily aligned over the target artery to measure blood pressure noninvasively and continuously. In addition, the ultraflexible and thin plastic construct of the microflotronic sensor (of 270 µm in height) can be worn comfortably for extended periods of time. Importantly, the proposed microflotronic sensor has been utilized to perform arterial tonometry with the capability of noninvasive monitoring of arterial blood pressure waveforms in a real-time and continuous fashion.







Similar content being viewed by others
References
Bergmann, J. H. M., and A. H. McGregor. Body-worn sensor design: what do patients and clinicians want? Ann. Biomed. Eng. 39:2299–2312, 2011.
Bhowmik, A. K., Z. Li, and P. J. Bos. Mobile Displays: Technology and Applications. Chichester: Wiley, 2008.
World Health Organization. Cardiovascular diseases (CVDs). Fact sheet No. 317, 2012. http://www.who.int/mediacentre/factsheets/fs317/en/.
Chew, M. S., and A. Åneman. Hemodynamic monitoring using arterial waveform analysis. Curr. Opin. Crit. Care. 19:234–241, 2013.
Currie, K. D., M. Hubli, and A. V. Krassioukov. Applanation tonometry: a reliable technique to assess aortic pulse wave velocity in spinal cord injury. Spinal Cord 52(4):272–275, 2014.
Di Giovanni, M. Flat and Corrugated Diaphram Design Handbook. New York: Marcel Dekker Inc, 1982.
Ding, Y., S. Garland, M. Howland, A. Revzin, and T. Pan. Universal nanopatternable interfacial bonding. Adv Mater. 23(46):5551–5556, 2011.
Fraile, J. A., B. Javier, J. M. Corchado, and A. Abraham. Applying wearable solutions in dependent environments. IEEE Trans. Inf. Technol. Biomed. 14(6):1459–1467, 2010.
Hu, C. S., Y. F. Chung, C. C. Yeh, and C. H. Luo. Temporal and spatial properties of arterial pulsation measurement using pressure sensor array. Evid. Based Complement. Alternat. Med. 745127:1–9, 2012.
Keir, P. J., and R. P. Wells. Changes in geometry of the finger flexor tendons in the carpal tunnel with wrist posture and tendon load: an MRI study on normal wrists. Clin Biomech. 14:635–645, 1999.
Li, R., B, Nie, P. Digiglio and T. Pan. Microflotronics: a flexible, transparent, pressure-sensitive microfluidic film. Adv. Funct. Mater., submitted, 2014.
Lin, H. K., S. M. Chiu, T. P. Cho, and J. C. Huang. Improved bending fatigue behavior of flexible PET/ITO film with thin metallic glass interlayer. Mater. Lett. 113:182–185, 2013.
Lin, W. H., H. Zhang, and Y. T. Zhang. Investigation on cardiovascular risk prediction using physiological parameters. Comput. Math. Methods Med. 2013:272691, 2013.
Lueder, E. Liquid Crystal Displays (2nd ed.). United Kindom: A John Wiley and Sons, 2010.
Mark, J. B. Atlas of Cardiovascular Monitoring. New York: Churchill Livingstone, 1998.
Mateu Campos, M. L., A. Ferrándiz Sellés, G. Gruartmoner de Vera, J. Mesquida Febrer, C. Sabatier Cloarec, Y. Poveda Hernández, and X. García Nogales. Techniques available for hemodynamic monitoring. advantages and limitations. Med. Intensiva 36(6):434–444, 2012.
Mceleavy, O. D., R. W. Mccallum, J. R. Petrie, M. Small, J. M. C. Connell, N. Sattar, and S. J. Cleland. Higher carotid-radial pulse wave velocity in healthy offspring of patients with type 2 diabetes.”. Diabet. Med. 21(3):262–266, 2004.
Mitchell, G. F., S.-J. Hwang, R. S. Vasan, M. G. Larson, M. J. Pencina, N. M. Hamburg, J. A. Vita, D. Levy, and E. J. Benjamin. Arterial stiffness and cardiovascular events: the framingham heart study. Circulation. 121(4):505–511, 2010.
Munir, S., A. Guilcher, T. Kamalesh, B. Clapp, S. Redwood, M. Marber, and P. Chowienczyk. Peripheral augmentation index defines the relationship between central and peripheral pulse pressure. Hypertension. 51(1):112–118, 2007.
Naghavi, M., P. Libby, E. Falk, et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: part II. Circulation 108(15):1772–1778, 2003.
Nie, B., R. Li, J. Brandt, and T. Pan. Iontronic microdroplet array for flexible ultrasensitive tactile sensing. Lab on a Chip. 14(6):1107–1116, 2014.
Nie, B., S. Xing, J. Brandt, and T. Pan. Droplet-based interfacial capacitive sensing. Lab on a Chip. 12:1110–1118, 2012.
Pan, T., and W. Wei. From cleanroom to desktop: emerging micro-nanofabrication technology for biomedical applications. Ann. Biomed. Eng. 39:600–620, 2010.
Pan, L., et al. An ultra-sensitive resistive pressure sensor based on hollow-sphere microstructure induced elasticity in conducting polymer film. Nat. Commun. 5:3002, 2014.
Pickering, T. G. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans. Circulation 111(5):697–716, 2005.
Pini, R., M. C. Cavallini, V. Palmieri, et al. Central but not brachial blood pressure predicts cardiovascular events in an unselected geriatric population. J. Am. Coll. Cardiol. 51(25):2432–2439, 2008.
Polyethylene terephthalate. TOXNET Toxicology Data Network. Bethesda: National Institutes of Health, 2009. http://toxnet.nlm.nih.gov/cgi-bin/sis/search/a?dbs+hsdb:@term+@DOCNO+7712.
Saugel, B., F. Fassio, A. Hapfelmeier, A. S. Meidert, R. M. Schmid, and W. Huber. The T-Line TL-200 system for continuous non-invasive blood pressure measurement in medical intensive care unit patients. Intensive Care Med. 38(9):1471–1477, 2012.
Sesso, H. D., M. J. Stampfer, B. Rosner, C. H. Hennekens, J. M. Gaziano, J. E. Manson, and R. J. Glynn. Systolic and diastolic blood pressure, pulse pressure, and mean arterial pressure as predictors of cardiovascular disease risk in men. Hypertension. 36(5):801–807, 2000.
Sule, A. A., T. H. Hwang, and T. J. Chin. Very high central aortic systolic pressures in a young hypertensive patient on Telmisartan: is central aortic systolic pressure associated with white coat hypertension? Int. J. Angiol. 19(4):138–140, 2010.
Takei, K., et al. Nanowire active-matrix circuitry for low-voltage macroscale artificial skin. Nat. Mater. 9(10):821–826, 2010.
Torrado, J., D. Bia, Y. Zocalo, G. Valls, S. Lluberas, D. Craiem, et al. Reactive hyperemia-related changes in carotid-radial pulse wave velocity as a potential tool to characterize the endothelial dynamics. Conf. Proc. IEEE Eng. Med. Biol. Soc., pp. 1800–1803, 2009.
Vassent, E., G. Meunier, A. Foggia, and G. Reyne. Simulation of induction of machine operation using a step by step finite-element method coupled with circuits and mechanical equations. IEEE Trans. Magn. 27(62):5232–5234, 1991.
Webster, J. G., and J. W. Clark. Medical Instrumentation: Application and Design. Hoboken, NJ: Wiley, 2010.
Webster, J. G., and J. W. Clark. Blood pressure and sound. In: Medical Instrumentation: Application and Design, edited by J. G. Webster. Hoboken, NJ: Wiley, 2010, pp. 311–312.
Wilkinson, M. B., and M. Outram. Principles of pressure transducers, resonance, damping and frequency response. Anaesthesia Intensive Care Med. 10(2):102–105, 2009.
Yoshioka, N., Y. Fujita, T. Yasukawa, I. Sano, M. Kiso, M. Nakayama, et al. Do radial arterial pressure curves have diagnostic validity for identify severe aortic stenosis? J. Anesth. 24:7–10, 2010.
Acknowledgments
This work is in part supported by the National Science Foundation (ECCS-0846502 and ECCS-1307831) and the University of California Proof-of-Concept Program (PC-269200) to TP. RL acknowledges the fellowship support from China Scholarship Council (CSC). Authors would also like to thank Yijun Zhang for his assistance on the device illustrations.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Yong Xu oversaw the review of this article.
Philip Digiglio and Ruya Li have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Digiglio, P., Li, R., Wang, W. et al. Microflotronic Arterial Tonometry for Continuous Wearable Non-Invasive Hemodynamic Monitoring. Ann Biomed Eng 42, 2278–2288 (2014). https://doi.org/10.1007/s10439-014-1037-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-1037-1