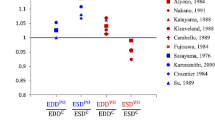
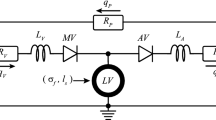
Abstract
Mechanics-based cardiac growth models can now predict changes in mass, chamber size, and wall thickness in response to perturbations such as pressure overload (PO), volume overload, and myocardial infarction with a single set of growth parameters. As these models move toward clinical applications, many of the most interesting applications involve predictions of whether or how a patient’s heart will reverse its growth after an intervention. In the case of PO, significant regression in wall thickness is observed both experimentally and clinically following relief of overload, for example following replacement of a stenotic aortic valve. Therefore, the objective of this work was to evaluate the ability of a published cardiac growth model that captures forward growth in multiple situations to predict growth reversal following relief of PO. Using a finite element model of a beating canine heart coupled to a circuit model of the circulation, we quantitatively matched hemodynamic data from a canine study of aortic banding followed by unbanding. Surprisingly, although the growth model correctly predicted the time course of PO-induced hypertrophy, it predicted only limited growth reversal given the measured unbanding hemodynamics, contradicting experimental and clinical observations. We were able to resolve this discrepancy only by incorporating an evolving homeostatic setpoint for the governing growth equations. Furthermore, our analysis suggests that many strain- and stress-based growth laws using the traditional volumetric growth framework will have similar difficulties capturing regression following the relief of PO unless growth setpoints are allowed to evolve.






Similar content being viewed by others
References
Arts T, Delhaas T, Bovendeerd P et al (2005) Adaptation to mechanical load determines shape and properties of heart and circulation: the CircAdapt model. Am J Physiol Heart Circ Physiol 288:H1943–H1954. https://doi.org/10.1152/ajpheart.00444.2004
Beach JM, Mihaljevic T, Rajeswaran J et al (2014) Ventricular hypertrophy and left atrial dilatation persist and are associated with reduced survival after valve replacement for aortic stenosis. J Thorac Cardiovasc Surg 147:362–369.e8. https://doi.org/10.1016/j.jtcvs.2012.12.016
Cutilletta AF, Dowell RT, Rudnik M et al (1975) Regression of myocardial hypertrophy. 1. Experimental model, changes in heart weight, nucleic-acids and collagen. J Mol Cell Cardiol 7:767–781. https://doi.org/10.1016/0022-2828(75)90042-5
Gao X-M, Kiriazis H, Moore X-L et al (2005) Regression of pressure overload-induced left ventricular hypertrophy in mice. Am J Physiol Heart Circ Physiol 288:H2702–H2707. https://doi.org/10.1152/ajpheart.00836.2004
Göktepe S, Abilez OJ, Parker KK, Kuhl E (2010) A multiscale model for eccentric and concentric cardiac growth through sarcomerogenesis. J Theor Biol 265:433–442. https://doi.org/10.1016/j.jtbi.2010.04.023
Hahn RT, Pibarot P, Stewart WJ et al (2013) Comparison of transcatheter and surgical aortic valve replacement in severe aortic stenosis: a longitudinal study of echocardiography parameters in cohort a of the PARTNER trial (placement of aortic transcatheter valves). J Am Coll Cardiol 61:2514–2521. https://doi.org/10.1016/j.jacc.2013.02.087
Hatani T, Kitai T, Murai R et al (2016) Associations of residual left ventricular and left atrial remodeling with clinical outcomes in patients after aortic valve replacement for severe aortic stenosis. J Cardiol 68:241–247. https://doi.org/10.1016/j.jjcc.2015.09.017
Holmes JW, Lumens J (2018) Clinical applications of patient-specific models: the case for a simple approach. J Cardiovasc Transl Res 11:71–79. https://doi.org/10.1007/s12265-018-9787-z
Humphrey JD, Rajagopal KR (2002) A constrained mixture model for growth and remodeling of soft tissues. Math Models Methods Appl Sci 12:407–430. https://doi.org/10.1142/S0218202502001714
Kerckhoffs RCP, Neal ML, Gu Q et al (2007) Coupling of a 3D finite element model of cardiac ventricular mechanics to lumped systems models of the systemic and pulmonic circulation. Ann Biomed Eng 35:1–18. https://doi.org/10.1007/s10439-006-9212-7
Kerckhoffs RCP, Omens JH, McCulloch AD (2012a) A single strain-based growth law predicts concentric and eccentric cardiac growth during pressure and volume overload. Mech Res Commun 42:40–50. https://doi.org/10.1016/j.mechrescom.2011.11.004
Kerckhoffs RCP, Omens JH, McCulloch AD (2012b) Mechanical discoordination increases continuously after the onset of left bundle branch block despite constant electrical dyssynchrony in a computational model of cardiac electromechanics and growth. Europace 14:v65–v72. https://doi.org/10.1093/europace/eus274
Kimata S, Morkin E (1971) Comparison of myosin synthesis in heart and red and white skeletal muscles. Am J Physiol 221:1706–1713. https://doi.org/10.1152/ajplegacy.1971.221.6.1706
Krayenbuehl HP, Hess OM, Monrad ES et al (1989) Left ventricular myocardial structure in aortic valve disease before, intermediate, and late after aortic valve replacement. Circulation 79:744–755. https://doi.org/10.1161/01.cir.79.4.744
Kroon W, Delhaas T, Arts T, Bovendeerd P (2009) Computational modeling of volumetric soft tissue growth: application to the cardiac left ventricle. Biomech Model Mechanobiol 8:301–309. https://doi.org/10.1007/s10237-008-0136-z
Lee LC, Genet M, Acevedo-Bolton G et al (2015) A computational model that predicts reverse growth in response to mechanical unloading. Biomech Model Mechanobiol 14:217–229. https://doi.org/10.1007/s10237-014-0598-0
Lin IE, Taber LA (1995) A model for stress-induced growth in the developing heart. J Biomech Eng Trans ASME 117:343–349. https://doi.org/10.1115/1.2794190
Matsumura Y, Gillinov AM, Toyono M et al (2008) Usefulness of left ventricular shape to predict the early recovery of left ventricular function after isolated aortic valve replacement for aortic valve stenosis. Am J Cardiol 102:1530–1534. https://doi.org/10.1016/j.amjcard.2008.07.044
Monrad ES, Hess OM, Murakami T et al (1988) Time course of regression of left ventricular hypertrophy after aortic valve replacement. Circulation 77:1345–1355. https://doi.org/10.1161/01.cir.77.6.1345
Rausch MK, Dam A, Göktepe S et al (2011) Computational modeling of growth: systemic and pulmonary hypertension in the heart. Biomech Model Mechanobiol 10:799–811. https://doi.org/10.1007/s10237-010-0275-x
Rodriguez EK, Hoger A, McCulloch AD (1994) Stress-dependent finite growth in soft elastic tissues. J Biomech 27:455–467. https://doi.org/10.1016/0021-9290(94)90021-3
Santamore WP, Burkhoff D (1991) Hemodynamic consequences of ventricular interaction as assessed by model analysis. Am J Physiol Heart Circ Physiol 260:H146–H157. https://doi.org/10.1152/ajpheart.1991.260.1.H146
Sasayama S, Ross J, Franklin D et al (1976) Adaptations of the left ventricle to chronic pressure overload. Circ Res 38:172–178. https://doi.org/10.1161/01.RES.38.3.172
Stansfield WE, Rojas M, Corn D et al (2007) Characterization of a model to independently study regression of ventricular hypertrophy. J Surg Res 142:387–393. https://doi.org/10.1016/j.jss.2007.01.037
Taber LA (1998) Biomechanical growth laws for muscle tissue. J Theor Biol 193:201–213. https://doi.org/10.1006/jtbi.1997.0618
Treibel TA, Kozor R, Schofield R et al (2018) Reverse myocardial remodeling following valve replacement in patients with aortic stenosis. J Am Coll Cardiol 71:860–871. https://doi.org/10.1016/j.jacc.2017.12.035
Villari B, Vassalli G, Monrad ES et al (1995) Normalization of diastolic dysfunction in aortic stenosis late after valve replacement. Circulation 91:2353–2358. https://doi.org/10.1161/01.cir.91.9.2353
Witzenburg CM, Holmes JW (2017) A comparison of phenomenologic growth laws for myocardial hypertrophy. J Elast 129:257–281. https://doi.org/10.1007/s10659-017-9631-8
Witzenburg CM, Holmes JW (2018) Predicting the time course of ventricular dilation and thickening using a rapid compartmental model. J Cardiovasc Transl Res. https://doi.org/10.1007/s12265-018-9793-1
Yarbrough WM, Mukherjee R, Ikonomidis JS et al (2012) Myocardial remodeling with aortic stenosis and after aortic valve replacement: mechanisms and future prognostic implications. J Thorac Cardiovasc Surg 143:656–664. https://doi.org/10.1016/j.jtcvs.2011.04.044
Zhao Z, Chen L, Xiao Y-B et al (2013) A rabbit model to study regression of ventricular hypertrophy. Heart Lung Circ 22:373–382. https://doi.org/10.1016/j.hlc.2012.11.021
Acknowledgements
This study was funded by the National Institutes of Health (U01 HL127654).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoshida, K., McCulloch, A.D., Omens, J.H. et al. Predictions of hypertrophy and its regression in response to pressure overload. Biomech Model Mechanobiol 19, 1079–1089 (2020). https://doi.org/10.1007/s10237-019-01271-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-019-01271-w