Abstract
Background
Emerging evidence has linked mitochondrial dysfunction to the pathogenesis of many renal disorders, including acute kidney injury, sepsis and even chronic kidney disease. Proteomics is a powerful tool in elucidating the role of mitochondria in renal pathologies. Since the pig is increasingly recognized as a major mammalian model for translational research, the lack of physiological proteome data of large mammals prompted us to examine renal mitochondrial proteome in porcine kidney cortex and medulla
Methods
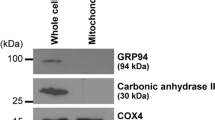
Kidneys were obtained from six healthy pigs. Mitochondria from cortex and medulla were isolated using differential centrifugation and proteome maps of cortical and medullar mitochondria were constructed using two-dimensional gel electrophoresis (2DE). Protein spots with significant difference between mitochondrial fraction of renal cortex and medulla were identified by mass spectrometry.
Results
Proteomic analysis identified 81 protein spots. Of these spots, 41 mitochondrial proteins were statistically different between renal cortex and medulla (p < 0.05). Protein spots containing enzymes of beta oxidation, amino acid metabolism, and gluconeogenesis were predominant in kidney cortex mitochondria. Spots containing tricarboxylic acid cycle enzymes and electron transport system proteins, proteins maintaining metabolite transport and mitochondrial translation were more abundant in medullar mitochondria.
Conclusion
This study provides the first proteomic profile of porcine kidney cortex and medullar mitochondrial proteome. Different protein expression pattern reflects divergent functional metabolic role of mitochondria in various kidney compartments. Our study could serve as a useful reference for further porcine experiments investigating renal mitochondrial physiology under various pathological states.




Similar content being viewed by others
Abbreviations
- 2DE:
-
Two-dimensional electrophoresis
- KM:
-
Kidney medulla
- KC:
-
Kidney cortex
- TCA:
-
Tricarboxylic acid cycle
- ETS:
-
Electron transport system
- ROS:
-
Reactive oxygen species
References
Balaban RS, Mandel LJ, Soltoff SP, Storey JM. Coupling of active ion transport and aerobic respiratory rate in isolated renal tubules. Proc Natl Acad Sci U S A. 1980;77(1):447–51.
Guder WG, Ross BD. Enzyme distribution along the nephron. Kidney Int. 1984;26(2):101–11.
Hall AM, Unwin RJ. The not so ‘mighty chondrion’: emergence of renal diseases due to mitochondrial dysfunction. Nephron Physiol. 2007;105(1):p1–10.
Brooks C, Wei Q, Cho SG, Dong Z. Regulation of mitochondrial dynamics in acute kidney injury in cell culture and rodent models. J Clin Investig. 2009;119(5):1275–85.
Funk JA, Schnellmann RG. Persistent disruption of mitochondrial homeostasis after acute kidney injury. Am J Physiol Renal Physiol. 2012;302(7):F853–64.
Dare AJ, Phillips AR, Hickey AJ, Mittal A, Loveday B, Thompson N, et al. A systematic review of experimental treatments for mitochondrial dysfunction in sepsis and multiple organ dysfunction syndrome. Free Radic Biol Med. 2009;47(11):1517–25.
Parikh SM. Therapeutic targeting of the mitochondrial dysfunction in septic acute kidney injury. Current opinion in critical care. 2013;19(6):554–9.
Thongboonkerd V. Current status of renal and urinary proteomics: ready for routine clinical application? Nephrol Dial Transplant. 2010;25(1):11–6.
Arthur JM, Thongboonkerd V, Scherzer JA, Cai J, Pierce WM, Klein JB. Differential expression of proteins in renal cortex and medulla: a proteomic approach. Kidney Int. 2002;62(4):1314–21.
Xu B, Yoshida Y, Zhang Y, Yaoita E, Osawa T, Yamamoto T. Two-dimensional electrophoretic profiling of normal human kidney: differential protein expression in glomerulus, cortex and medulla. J Electrophor. 2005;49(1):5–13.
Fountoulakis M, Berndt P, Langen H, Suter L. The rat liver mitochondrial proteins. Electrophoresis. 2002;23(2):311–28.
Bugger H, Chen D, Riehle C, Soto J, Theobald HA, Hu XX, et al. Tissue-specific remodeling of the mitochondrial proteome in type 1 diabetic akita mice. Diabetes. 2009;58(9):1986–97.
Freund DM, Prenni JE, Curthoys NP. Response of the mitochondrial proteome of rat renal proximal convoluted tubules to chronic metabolic acidosis. Am J Physiol Renal Physiol. 2013;304(2):F145–55.
Chaiyarit S, Thongboonkerd V Changes in mitochondrial proteome of renal tubular cells induced by calcium oxalate monohydrate crystal adhesion and internalization are related to mitochondrial dysfunction. J Proteome Res. 2012
Goldfarb RD, Dellinger RP, Parrillo JE. Porcine models of severe sepsis: emphasis on porcine peritonitis. Shock. 2005;24(Suppl 1):75–81.
Doi K, Leelahavanichkul A, Yuen PS, Star RA. Animal models of sepsis and sepsis-induced kidney injury. J clin investig. 2009;10(119):2868–78.
Baumert H, Faure JP, Zhang K, Petit I, Goujon JM, Dutheil D, et al. Evidence for a mitochondrial impact of trimetazidine during cold ischemia and reperfusion. Pharmacology. 2004;71(1):25–37.
Bendixen E. Animal models for translational proteomics. Proteomics Clin Appl. 2014;8(10):637–9.
de Cavanagh EM, Piotrkowski B, Basso N, Stella I, Inserra F, Ferder L, et al. Enalapril and losartan attenuate mitochondrial dysfunction in aged rats. FASEB j: off publ Fed Am Soc Exp Biol. 2003;17(9):1096–8.
Mares J, Richtrova P, Hricinova A, Tuma Z, Moravec J, Lysak D, et al. Proteomic profiling of blood-dialyzer interactome reveals involvement of lectin complement pathway in hemodialysis-induced inflammatory response. Proteomics Clin Appl. 2010;4(10–11):829–38.
Kiyomiya K, Matsushita N, Matsuo S, Kurebe M. Cephaloridine-induced inhibition of cytochrome c oxidase activity in the mitochondria of cultured renal epithelial cells (LLC-PK(1)) as a possible mechanism of its nephrotoxicity. Toxicol Appl Pharmacol. 2000;167(2):151–6.
Verma N, Rettenmeier AW, Schmitz-Spanke S. Recent advances in the use of Sus scrofa (pig) as a model system for proteomic studies. Proteomics. 2011;11(4):776–93.
Lebiedzinska M, Szabadkai G, Jones AW, Duszynski J, Wieckowski MR. Interactions between the endoplasmic reticulum, mitochondria, plasma membrane and other subcellular organelles. Int J Biochem Cell Biol. 2009;41(10):1805–16.
Eaton S, Bartlett K, Pourfarzam M. Mammalian mitochondrial beta-oxidation. Biochem J. 1996;320(Pt 2):345–57.
Lehir M, Dubach UC. Peroxisomal and mitochondrial beta-oxidation in the rat-kidney: distribution of fatty acyl-coenzyme a oxidase and 3-hydroxyacyl-coenzyme-a dehydrogenase-activities along the nephron. J Histochem Cytochem. 1982;30(5):441–4.
Gerich JE, Meyer C, Woerle HJ, Stumvoll M. Renal gluconeogenesis: its importance in human glucose homeostasis. Diabetes Care. 2001;24(2):382–91.
Yasuda M, Fujita T, Higashio T, Okahara T, Abe Y, Yamamoto K. Effects of 4-pentenoic acid and furosemide on renal functions and renal uptake of individual free fatty acids. Pflug Arch. 1980;385(2):111–6.
Huss JM, Levy FH, Kelly DP. Hypoxia inhibits the peroxisome proliferator-activated receptor alpha/retinoid X receptor gene regulatory pathway in cardiac myocytes: a mechanism for O2-dependent modulation of mitochondrial fatty acid oxidation. J Biol Chem. 2001;276(29):27605–12.
Knight J, Jiang J, Assimos DG, Holmes RP. Hydroxyproline ingestion and urinary oxalate and glycolate excretion. Kidney Int. 2006;70(11):1929–34.
Lowry M, Hall DE, Brosnan JT. Hydroxyproline metabolism by the rat kidney: distribution of renal enzymes of hydroxyproline catabolism and renal conversion of hydroxyproline to glycine and serine. Metab, Clin Exp. 1985;34(10):955–61.
Burgmeier N, Zawislak R, Defeudis FV, Bollack C, Helwig JJ. Glutamic acid decarboxylase in tubules and glomeruli isolated from rat kidney cortex. Eur J Biochem. 1985;151(2):361–4.
Tillakaratne NJ, Medina-Kauwe L, Gibson KM. Gamma-aminobutyric acid (GABA) metabolism in mammalian neural and nonneural tissues. Comp Biochem Physiol A Physiol. 1995;112(2):247–63.
Pircher H, Straganz GD, Ehehalt D, Morrow G, Tanguay RM, Jansen-Durr P. Identification of human fumarylacetoacetate hydrolase domain-containing protein 1 (FAHD1) as a novel mitochondrial acylpyruvase. J Biol Chem. 2011;286(42):36500–8.
Wyss M, Kaddurah-Daouk R. Creatine and creatinine metabolism. Physiol Rev. 2000;80(3):1107–213.
van de Poll MC, Soeters PB, Deutz NE, Fearon KC, Dejong CH. Renal metabolism of amino acids: its role in interorgan amino acid exchange. Am J Clin Nutr. 2004;79(2):185–97.
Monteil C, Fillastre JP, Morin JP. Expression and subcellular distribution of phosphoenolpyruvate carboxykinase in primary cultures of rabbit kidney proximal tubule cells: comparative study with renal and hepatic PEPCK in vivo. Biochim Biophys Acta. 1995;1243(3):437–45.
Watford M, Hod Y, Chiao YB, Utter MF, Hanson RW. The unique role of the kidney in gluconeogenesis in the chicken. The significance of a cytosolic form of phosphoenolpyruvate carboxykinase. J Biol Chem. 1981;256(19):10023–7.
Modaressi S, Brechtel K, Christ B, Jungermann K. Human mitochondrial phosphoenolpyruvate carboxykinase 2 gene. Structure, chromosomal localization and tissue-specific expression. Biochem J. 1998;333(Pt 2):359–66.
Schmidt U, Guder WG. Sites of enzyme activity along the nephron. Kidney Int. 1976;9(3):233–42.
Chinopoulos C. Which way does the citric acid cycle turn during hypoxia? The critical role of alpha-ketoglutarate dehydrogenase complex. J Neurosci Res. 2013;91(8):1030–43.
Dukhande VV, Sharma GC, Lai JC, Farahani R. Chronic hypoxia-induced alterations of key enzymes of glucose oxidative metabolism in developing mouse liver are mTOR dependent. Mol Cell Biochem. 2011;357(1–2):189–97.
Levillain O, Hus-Citharel A, Garvi S, Peyrol S, Reymond I, Mutin M, et al. Ornithine metabolism in male and female rat kidney: mitochondrial expression of ornithine aminotransferase and arginase II. Am J Physiol Renal Physiol. 2004;286(4):F727–38.
Hirst J. Why does mitochondrial complex I have so many subunits? Biochem J. 2011;437(2):e1–3.
Hirst J, Carroll J, Fearnley IM, Shannon RJ, Walker JE. The nuclear encoded subunits of complex I from bovine heart mitochondria. Biochim Biophys Acta. 2003;1604(3):135–50.
Guzy RD, Hoyos B, Robin E, Chen H, Liu LP, Mansfield KD, et al. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 2005;1(6):401–8.
Trueblood CE, Wright RM, Poyton RO. Differential regulation of the two genes encoding Saccharomyces cerevisiae cytochrome c oxidase subunit V by heme and the HAP2 and REO1 genes. Mol Cell Biol. 1988;8(10):4537–40.
Rostovtseva T, Colombini M. ATP flux is controlled by a voltage-gated channel from the mitochondrial outer membrane. J Biol Chem. 1996;271(45):28006–8.
Abu-Hamad S, Sivan S, Shoshan-Barmatz V. The expression level of the voltage-dependent anion channel controls life and death of the cell. Proc Natl Acad Sci U S A. 2006;103(15):5787–92.
Woriax VL, Burkhart W, Spremulli LL. Cloning, sequence analysis and expression of mammalian mitochondrial protein synthesis elongation factor Tu. Biochim Biophys Acta. 1995;1264(3):347–56.
Sasarman F, Antonicka H, Shoubridge EA. The A3243G tRNALeu(UUR) MELAS mutation causes amino acid misincorporation and a combined respiratory chain assembly defect partially suppressed by overexpression of EFTu and EFG2. Hum Mol Genet. 2008;17(23):3697–707.
Acknowledgments
This work was supported by the Research Project No. MSM0021620819 “Replacement of and Support to Some Vital Organs”, by the Charles University Research Fund (project number P36) by the project ED2.1.00/03.0076 by the European Regional Development Fund, and the Specific Student Research Project no. 260175/2015 of the Charles University in Prague.
Conflict of interest
All the authors have declared no competing interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Tuma, Z., Kuncova, J., Mares, J. et al. Mitochondrial proteomes of porcine kidney cortex and medulla: foundation for translational proteomics. Clin Exp Nephrol 20, 39–49 (2016). https://doi.org/10.1007/s10157-015-1135-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-015-1135-x