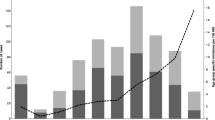
Abstract
A major virulence factor of group A streptococci (GAS) is the M protein. Strains with the M3 type are more often associated with necrotizing fasciitis (NF) and streptococcal toxic shock syndrome, and have a higher case fatality rate than strains of other M types. To better understand the epidemiology of M3 GAS strains in Norway, we analyzed 59 invasive and 69 pharyngeal isolates with respect to prophage content, allelic variation in emm3, mtsR encoding the metal transporter of Streptococcus repressor (mtsR), and sclB coding for streptococcal collagen-like protein B. The Norwegian emm3 strains were very homogeneous, mainly harboring the emm allele 3.1 and prophage profile ΦG3.01. Other prophage profiles were transient. The mutation in mtsR known to truncate the protein and result in decreased capacity to cause NF was not found in our isolates. The sclB gene usually harbored five or eight contiguous repeats of a CAAAA pentanucleotide sequence and a highly modular and variable collagen structural motif (CSM) region with 9 and 12 amino acid M3-specific conserved motif repeats distributed across the entire CSM region. Strains with 5 CAAAA repeats emerged in 1993 and these strains were associated with the increase in invasive M3 cases in the period 1993–2003.



Similar content being viewed by others
References
Akesson P, Rasmussen M, Mascini E, Pawel-Rammingen U, Janulczyk R, Collin M, Olsen A, Mattsson E, Olsson ML, Bjorck L, Christensson B (2004) Low antibody levels against cell wall-attached proteins of Streptococcus pyogenes predispose for severe invasive disease. J Infect Dis 189:797–804
Banks DJ, Beres SB, Musser JM (2002) The fundamental contribution of phages to GAS evolution, genome diversification and strain emergence. Trends Microbiol 10:515–521
Bates CS, Toukoki C, Neely MN, Eichenbaum Z (2005) Characterization of MtsR, a new metal regulator in group A Streptococcus, involved in iron acquisition and virulence. Infect Immun 73:5743–5753
Beres SB, Musser JM (2007) Contribution of exogenous genetic elements to the group A Streptococcus metagenome. PLoS ONE 2:e800
Beres SB, Sylva GL, Sturdevant DE, Granville CN, Liu M, Ricklefs SM, Whitney AR, Parkins LD, Hoe NP, Adams GJ, Low DE, DeLeo FR, McGeer A, Musser JM (2004) Genome-wide molecular dissection of serotype M3 group A Streptococcus strains causing two epidemics of invasive infections. Proc Natl Acad Sci U S A 101:11833–11838
Beres SB, Richter EW, Nagiec MJ, Sumby P, Porcella SF, DeLeo FR, Musser JM (2006) Molecular genetic anatomy of inter- and intraserotype variation in the human bacterial pathogen group A Streptococcus. Proc Natl Acad Sci U S A 103:7059–7064
Bisno AL, Brito MO, Collins CM (2003) Molecular basis of group A streptococcal virulence. Lancet Infect Dis 3:191–200
Caparon MG, Geist RT, Perez-Casal J, Scott JR (1992) Environmental regulation of virulence in group A streptococci: transcription of the gene encoding M protein is stimulated by carbon dioxide. J Bacteriol 174:5693–5701
Caswell CC, Lukomska E, Seo NS, Hook M, Lukomski S (2007) Scl1-dependent internalization of group A Streptococcus via direct interactions with the α2β1 integrin enhances pathogen survival and re-emergence. Mol Microbiol 64:1319–1331
Caswell CC, Barczyk M, Keene DR, Lukomska E, Gullberg DE, Lukomski S (2008) Identification of the first prokaryotic collagen-sequence motif that mediates binding to human collagen receptors, integrins α2β1 and α11β1. J Biol Chem 283:36168–36175
Colman G, Tanna A, Efstratiou A, Gaworzewska ET (1993) The serotypes of Streptococcus pyogenes present in Britain during 1980–1990 and their association with disease. J Med Microbiol 39:165–178
Efstratiou A (2000) Group A streptococci in the 1990s. J Antimicrob Chemother 45 [Suppl]:3–12
Graham MR, Virtaneva K, Porcella SF, Barry WT, Gowen BB, Johnson CR, Wright FA, Musser JM (2005) Group A Streptococcus transcriptome dynamics during growth in human blood reveals bacterial adaptive and survival strategies. Am J Pathol 166:455–465
Green NM, Beres SB, Graviss EA, Allison JE, McGeer AJ, Vuopio-Varkila J, Lefebvre RB, Musser JM (2005) Genetic diversity among type emm28 group A Streptococcus strains causing invasive infections and pharyngitis. J Clin Microbiol 43:4083–4091
Humtsoe JO, Kim JK, Xu Y, Keene DR, Hook M, Lukomski S, Wary KK (2005) A streptococcal collagen-like protein interacts with the α2β1 integrin and induces intracellular signaling. J Biol Chem 280:13848–13857
Keefer CS, Ingelfinger FJ, Spink WW (1986) Significance of hemolytic streptococcic bacteremia: a study of two hundred and forty-six patients. Rev Infect Dis 8:832–837
Lamagni TL, Efstratiou A, Vuopio-Varkila J, Jasir A, Schalen C (2005) The epidemiology of severe Streptococcus pyogenes associated disease in Europe. Euro Surveill 10:179–184
Lukomski S, Nakashima K, Abdi I, Cipriano VJ, Ireland RM, Reid SD, Adams GG, Musser JM (2000) Identification and characterization of the scl gene encoding a group A Streptococcus extracellular protein virulence factor with similarity to human collagen. Infect Immun 68:6542–6553
Lukomski S, Nakashima K, Abdi I, Cipriano VJ, Shelvin BJ, Graviss EA, Musser JM (2001) Identification and characterization of a second extracellular collagen-like protein made by group A Streptococcus: control of production at the level of translation. Infect Immun 69:1729–1738
Martin PR, Hoiby EA (1990) Streptococcal serogroup A epidemic in Norway 1987–1988. Scand J Infect Dis 22:421–429
McShan WM, Savic DJ, Karasawa T, Lin SP, Roe B, Ferretti JJ (2006) The Streptococcus genome era. Int Congr Ser 1289:171–174
Meisal R, Hoiby EA, Aaberge IS, Caugant DA (2008) Sequence type and emm type diversity in Streptococcus pyogenes isolates causing invasive disease in Norway between 1988 and 2003. J Clin Microbiol 46:2102–2105
O’Brien KL, Beall B, Barrett NL, Cieslak PR, Reingold A, Farley MM, Danila R, Zell ER, Facklam R, Schwartz B, Schuchat A (2002) Epidemiology of invasive group a Streptococcus disease in the United States, 1995–1999. Clin Infect Dis 35:268–276
Proft T, Webb PD, Handley V, Fraser JD (2003) Two novel superantigens found in both group A and group C Streptococcus. Infect Immun 71:1361–1369
Rasmussen M, Bjorck L (2001) Unique regulation of SclB—a novel collagen-like surface protein of Streptococcus pyogenes. Mol Microbiol 40:1427–1438
Rasmussen M, Eden A, Bjorck L (2000) SclA, a novel collagen-like surface protein of Streptococcus pyogenes. Infect Immun 68:6370–6377
Rivera A, Rebollo M, Miro E, Mateo M, Navarro F, Gurgui M, Mirelis B, Coll P (2006) Superantigen gene profile, emm type and antibiotic resistance genes among group A streptococcal isolates from Barcelona, Spain. J Med Microbiol 55:1115–1123
Sharkawy A, Low DE, Saginur R, Gregson D, Schwartz B, Jessamine P, Green K, McGeer A (2002) Severe group a streptococcal soft-tissue infections in Ontario: 1992–1996. Clin Infect Dis 34:454–460
Veasy LG, Tani LY, Daly JA, Korgenski K, Miner L, Bale J, Kaplan EL, Musser JM, Hill HR (2004) Temporal association of the appearance of mucoid strains of Streptococcus pyogenes with a continuing high incidence of rheumatic fever in Utah. Pediatrics 113:e168–e172
Vlaminckx BJ, Schuren FH, Montijn RC, Caspers MP, Fluit AC, Wannet WJ, Schouls LM, Verhoef J, Jansen WT (2007) Determination of the relationship between group A streptococcal genome content, M type, and toxic shock syndrome by a mixed genome microarray. Infect Immun 75:2603–2611
Whatmore AM (2001) Streptococcus pyogenes sclB encodes a putative hypervariable surface protein with a collagen-like repetitive structure. Microbiology 147:419–429
Acknowledgements
We thank Stephen B. Beres, Nicole M. Green, Izabela Sitkiewicz, Anne H. Tart, and Paul Sumby at The Methodist Hospital Research Institute for help and technical advice. We also thank Line Eikvil from the Norwegian Computing Center, for help with the principal component analysis. Funding was provided by the Norwegian Research Council grants 166001/v40 and 177397/v40 to D.A.C.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meisal, R., Høiby, E.A., Caugant, D.A. et al. Molecular characteristics of pharyngeal and invasive emm3 Streptococcus pyogenes strains from Norway, 1988–2003. Eur J Clin Microbiol Infect Dis 29, 31–43 (2010). https://doi.org/10.1007/s10096-009-0814-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-009-0814-5