Abstract
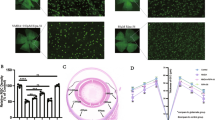
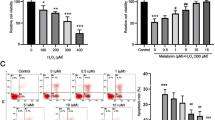
Glutathione (GSH) plays a critical role in cellular defense against unregulated oxidative stress in mammalian cells including neurons. We previously demonstrated that GSH decrease using [D, L]-buthionine sulphoximine (BSO) induces retinal cell death, but the underlying mechanisms of this are still unclear. Here, we demonstrated that retinal GSH level is closely related to retinal cell death as well as expression of an anti-apoptotic molecule, Bcl-2, in the retina. We induced differential expression of retinal GSH by single and multiple administrations of BSO, and examined retinal GSH levels and retinal cell death in vivo. Single BSO administration showed a transient decrease in the retinal GSH level, whereas multiple BSO administration showed a persistent decrease in the retinal GSH level. Retinal cell death also showed similar patterns: transient increases of retinal cell death were observed after single BSO administration, whereas persistent increases of retinal cell death were observed after multiple BSO administration. Changes in the retinal GSH level affected Bcl-2 expression in the retina. Immunoblot and immunohistochemical analyses showed that single and multiple administration of BSO induced differential expressions of Bcl-2 in the retina. Taken together, the results of our study suggest that the retinal GSH is important for the survival of retinal cells, and retinal GSH appears to be deeply related to Bcl-2 expression in the retina. Thus, alteration of Bcl-2 expression may provide a therapeutic tool for retinal degenerative diseases caused by retinal oxidative stress such as glaucoma or retinopathy.
Similar content being viewed by others
References
Allsopp, T.E., Robinson, M., Wyatt, S., and Davies, A.M. (1993). Ectopic trkA expression mediates a NGF survival response in NGF-independent sensory neurons but not in parasympathetic neurons. J. Cell Biol. 123, 1555–1566.
Anderson, M.E. (1998). Glutathione: an overview of biosynthesis and modulation. Chem. Biol. Int. 111–112, 1–14.
Armstrong, J.S., and Jones, D.P. (2002). Glutathione depletion enforces the mitochondrial permeability transition and causes cell death in Bcl-2 overexpressing HL60 cells. FASEB J. 16, 1263–1265.
Chen, J.Z., and Kadlubar, F.F. (2003). A new clue to glaucoma pathogenesis. Am. J. Med. 114, 697–698.
Circu, M.L., and Aw, T.Y. (2012). Glutathione and modulation of cell apoptosis. Biochim. Biophys. Acta 1823, 1767–1777.
Dalle-Donne, I., Rossi, R., Giustarini, D., Colombo, R., and Milzani, A. (2007). S-glutathionylation in protein redox regulation. Free Rad. Biol. Med. 43, 883–898.
Dalton, T.P., Chen, Y., Schneider, S.N., Nebert, D.W., and Shertzer, H.G. (2004). Genetically altered mice to evaluate glutathione homeostasis in health and disease. Free Radic. Biol. Med. 37, 1511–1526.
de Bernardo, S., Canals, S., Casarejos, M.J., Solano, R.M., Menendez, J., and Mena, M.A. (2004). Role of extracellular signal-regulated protein kinase in neuronal cell death induced by glutathione depletion in neuron/glia mesencephalic cultures. J. Neurochem. 91, 667–682.
Ellerby, L.M., Ellerby, H.M., Park, S.M., Holleran, A.L., Murphy, A.N., Fiskum, G., Kane, D.J., Testa, M.P., Kayalar, C., and Bredesen, D.E. (1996). Shift of the cellular oxidation-reduction potential in neural cells expressing Bcl-2. J. Neurochem. 67, 1259–1267.
Ferreira, S.M., Lerner, S.F., Brunzini, R., Evelson, P.A., and Llesuy, S.F. (2004). Oxidative stress markers in aqueous humor of glaucoma patients. Am. J. Ophthalmol. 137, 62–69.
Franco, R., and Cidlowski, J.A. (2009). Apoptosis and glutathione: beyond an antioxidant. Cell Death Differ. 16, 1303–1314.
Ganea, E., and Harding, J.J. (2006). Glutathione-related enzymes and the eye. Curr. Eye Res. 31, 1–11.
Gherghel, D., Griffiths, H.R., Hilton, E.J., Cunliffe, I.A., and Hosking, S.L. (2005). Systemic reduction in glutathione levels occurs in patients with primary open-angle glaucoma. Invest. Ophthalmol. Vis. Sci. 46, 877–883.
Hochman, A., Sternin, H., Gorodin, S., Korsmeyer, S., Ziv, I., Melamed, E., and Offen, D. (1998). Enhanced oxidative stress and altered antioxidants in brains of Bcl-2-deficient mice. J. Neurochem. 71, 741–748.
Hockenbery, D., Nunez, G., Milliman, C., Schreiber, R.D., and Korsmeyer, S.J. (1990). Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 348, 334–336.
Holm, K., and Isacson, O. (1999). Factors intrinsic to the neuron can induce and maintain its ability to promote axonal outgrowth: a role for BCL2? Trends Neurosci. 22, 269–273.
Kane, D.J., Sarafian, T.A., Anton, R., Hahn, H., Gralla, E.B., Valentine, J.S., Ord, T., and Bredesen, D.E. (1993). Bcl-2 inhibition of neural death: decreased generation of reactive oxygen species. Science 262, 1274–1277.
Kotulska, K., Lewin-Kowalik, J., Jaroslaw-Jerzy, B., Larysz-Brysz, M., Marcol, W., and Fus, Z. (2003). Bcl-2 deficiency deprives peripheral nerves of neurotrophic activity against injured optic nerve. J. Neurosci. Res. 73, 846–852.
Lim, E.J., Kim, I.B., Oh, S.J., and Chun, M.H. (2007). Identification and characterization of SMI32-immunoreactive amacrine cells in the mouse retina. Neurosci. Lett. 424, 199–202.
Martinou, J.C., Dubois-Dauphin, M., Staple, J.K., Rodriguez, I., Frankowski, H., Missotten, M., Albertini, P., Talabot, D., Catsicas, S., Pietra, C., et al. (1994). Overexpression of BCL-2 in transgenic mice protects neurons from naturally occurring cell death and experimental ischemia. Neuron 13, 1017–1030.
Moon, C., Jaberi, P., Otto-Bruc, A., Baehr, W., Palczewski, K., and Ronnett, G.V. (1998). Calcium-sensitive particulate guanylyl cyclase as a modulator of cAMP in olfactory receptor neurons. J. Neurosci. 18, 3195–3205.
Moreno, M.C., Campanelli, J., Sande, P., Sáenz, D.A., Keller Sarmiento, M.I., and Rosenstein, R.E. (2004). Retinal oxidative stress induced by high intraocular pressure. Free Radic. Biol. Med. 37, 803–812.
Reed, J.C., Doctor, K.S., and Godzik, A. (2004). The domains of apoptosis: a genomics perspective. Sci. STKE 2004, re9.
Roh, Y.J., Moon, C., Kim, S.Y., Park, M.H., Bae, Y.C., Chun, M.H., and Moon, J.I. (2007). Glutathione depletion induces differential apoptosis in cells of mouse retina, in vivo. Neurosci. Lett. 417, 266–270.
Schutte, M., and Werner, P. (1998). Redistribution of glutathione in the ischemic rat retina. Neurosci. Lett. 246, 53–56.
Song, H.G., Moon, C., Park, M.H., Moon, J.I., and Moon, C. (2008). Decrease in intracellular glutathione level alters expressions of B-cell CLL/Lymphoma 2 family members in the mouse retina. J. Health Sci. 54, 464–470.
Sternberg, P., Jr., Davidson, P.C., Jones, D.P., Hagen, T.M., Reed, R.L., and Drews-Botsch, C. (1993). Protection of retinal pigment epithelium from oxidative injury by glutathione and precursors. Invest. Ophthalmol. Vis. Sci. 34, 3661–3668.
Unal, M., Guven, M., Devranoglu, K., Ozaydin, A., Batar, B., Tamcelik, N., Gorgun, E.E., Ucar, D., and Sarici, A. (2007). Glutathione S transferase M1 and T1 genetic polymorphisms are related to the risk of primary open-angle glaucoma: a study in a Turkish population. Br. J. Ophthalmol. 91, 527–530.
Voehringer, D.W. (1999). BCL-2 and glutathione: alterations in cellular redox state that regulate apoptosis sensitivity. Free Radic. Biol. Med. 27, 945–950.
Voehringer, D.W., and Meyn, R.E. (2000). Redox aspects of Bcl-2 function. Antioxid. Redox Signal. 2, 537–550.
Wilkins, H.M., Marquardt, K., Lash, L.H., and Linseman, D.A. (2012). Bcl-2 is a novel interacting partner for the 2-oxoglutarate carrier and a key regulator of mitochondrial glutathione. Free Radic. Biol. Med. 52, 410–419.
Yang, J., Tezel, G., Patil, R.V., Romano, C., and Wax, M.B. (2001). Serum autoantibody against glutathione S-transferase in patients with glaucoma. Invest. Ophthalmol. Vis. Sci. 42, 1273–1276.
Yildirim, O., Ates, N.A., Tamer, L., Muslu, N., Ercan, B., Atik, U., and Kanik, A. (2004). Changes in antioxidant enzyme activity and malondialdehyde level in patients with age-related macular degeneration. Ophthalmologica 218, 202–206.
Zhong, L.T., Sarafian, T., Kane, D.J., Charles, A.C., Mah, S.P., Edwards, R.H., and Bredesen, D.E. (1993). bcl-2 inhibits death of central neural cells induced by multiple agents. Proc. Natl. Acad. Sci. USA 90, 4533–4537.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Park, M.H., Kim, S.Y., Moon, C. et al. Differential cell death and Bcl-2 expression in the mouse retina after glutathione decrease by systemic D,L-buthionine sulphoximine administration. Mol Cells 35, 235–242 (2013). https://doi.org/10.1007/s10059-013-2276-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-2276-y