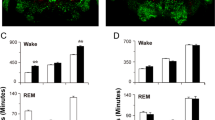
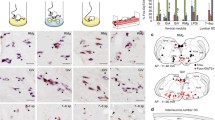
Abstract
REM sleep is characterized by rapid eye movements, desynchronized electroencephalographic activity, dreams and muscle paralysis that preclude the individual from acting out the action of dreams. REM sleep is generated and modulated by a complex and still poorly understood, neuronal network that involves multiple nuclei and neurotransmission systems. The key structures that generate REM sleep muscle paralysis are the subcoeruleus nucleus in the mesopontine tegmentum and the reticular formation of the ventral medial medulla. Using glutamatergic, GABAergic and glycinergic inputs, direct and indirect projections from these two areas inhibit the motoneurons of the spinal cord resulting in skeletal paralysis in REM sleep. Experimental studies in cats and rodents where the subcoeruleus nucleus and ventral medial medulla were impaired by electrolytic, pharmacological and genetic manipulations have repeatedly produced increased electromyography activity during REM sleep associated with abnormal motor behaviors (e.g., prominent twitching, attack-like behaviors). These animal models represent the pathophysiological substrate of REM sleep behavior disorder, a parasomnia in humans characterized by nightmares and abnormal vigorous behaviors (e.g., prominent jerking, shouting, kicking) linked to excessive phasic and/or tonic electromyographic activity in REM sleep. The extraordinary observation that a sleep disorder is often the first manifestation of a devastating neurodegenerative disease such as Parkinson disease carries important diagnostic implications and opens a window for neuroprotection. This review addresses the neuronal substrates of REM sleep generation and modulation and how its impairment may lead to REM sleep behavior disorder.










Similar content being viewed by others
References
Aguirre-Mardones C, Iranzo A, Vilas D, Serradell M, Gaig C, Santamaría J, Tolosa E (2015) Prevalence and timeline of nonmotor symptoms in idiopathic rapid eye movement sleep behavior disorder. J Neurol 262:1568–1578
American Academy of Sleep Medicine (2014) International classification of sleep disorders, 3rd edn. American Academy of Sleep Medicine, Darien
Anaclet C, Pedersen NP, Fuller PM, Lu J (2010) Brain circuitry regulating phasic activity in the trigeminal motor nucleus (Mo5) during REM sleep. PLoS One 5:e878
Antelmi E, Donadio V, Incensi A, Plazzi G, Liguori R (2017) Skin nerve phosphorylated α-synuclein deposits in idiopathic REM sleep behavior disorder. Neurology 88:2128–2131
Arrigoni E, Chen MC, Fuller PM (2016) The anatomical, cellular and synaptic basis of motor atonia during rapid eye movement sleep. J Physiol 594:5391–5414
Aserinsky E, Kleitman N (1953) Regularly occurring periods of eye motility and concomitant phenomena during sleep. Science 118:273–274
Aurora RN, Zak RS, Maganti RK, Auerbach SH, Casey KR, Chowdhuri S, Karippot A, Ramar K, Kristo DA, Morgenthaler TI, Standards of Practice Committee; American Academy of Sleep Medicine (2010) Best practice guide for the treatment of REM sleep behavior disorder (RBD). J Clin Sleep Med 6:85–95
Bischof M, Bassetti CL (2004) Total dream loss: a distinct neuropsychological dysfunction after bilateral PCA stroke. Ann Neurol 56:583–586
Blumberg MS, Plumeau AM (2016) A new view of “dream enactment” in REM sleep behavior disorder. Sleep Med Rev 30:34–42
Boeve BF (2013) Idiopathic REM sleep behavior disorder in the development of Parkinson’s disease. Lancet Neurol 12:469–482
Boissard R, Gervasoni D, Scmidt MH, Barbagli B, Fort P, Luppi PH (2002) The rat ponto-medullary network responsible for paradoxical sleep onset and maintenance: a combined microinjection and functional neuroanatomical study. Eur J Neurosci 16:1959–1973
Boissard R, Fort P, Gervasoni D, Barbagli B, Luppi PH (2003) Localization of the GABAergic and non-GABAergic neurons projecting to the sublaterodorsal nucleus and potentially gating of paradoxical sleep. Eur J Neurosci 18:1627–1639
Braak H, Del Tredici K (2017) Neuropathological staging of brain pathology in sporadic Parkinson’s disease: separating the wheat from the chaff. J Parkinsons Dis 7(Suppl 1):S73–S87
Braak H, Rub U, Sandmann-Keil D, Gai WP, de Vos RA, Jansen Steur EN, Arai K, Braak E (2000) Parkinson’s disease: affection of brain stem nuclei controlling premotor and motor neurons of the somatomotor system. Acta Neuropathol 99:489–495
Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol Aging 24:197–211
Brooks PL, Peever JH (2008) Glycinercic and GABAA-mediated inhibition of somatic motoneurons does not mediate rapid eye movement sleep motor atonia. J Neurosci 28:3535–3545
Brooks PL, Peever JH (2011) Impaired GABA and glycine transmission triggers cardinal features of rapid eye movement sleep disorder in mice. J Neurosci 31:7111–7121
Brooks PL, Peever JH (2012) Identification of the transmitter and receptor mechanisms responsible for REM sleep paralysis. J Neurosci 32:9785–9795
Chase MH (2008) Confirmation of the consensus that glycinergic postsynaptic inhibition is responsible for the atonia of REM sleep. Sleep 31:1487–1491
Chase MH, Soja PJ, Morales FR (1989) Evidence that glycine mediates the post-synaptic potentials that inhibit lumbar motoneurons during the atonia of active sleep. J Neurosci 9:743–751
Chen MC, Vetrivelan R, Guo CN, Chang C, Fuller PM, Lu J (2017) Ventral medullary control of rapid eye movement sleep and atonia. Exp Neurol 290:53–62
Clement O, Sapin E, Bérod A, Fort P, Luppi PH (2011) Evidence that neurons of the sublaterodorsal tegmental nucleus triggering paradoxical (REM) sleep are glutamatergic. Sleep 34:419–423
Crochet S, Sakai K (2003) Dopaminergic modulation of behavioral states in meso-pontine tegmentum: a reverse microdialysis study in freely moving cats. Sleep 26:801–806
Dang-Vu TT, Schabaus M, Desseilles M, Sterpenich V, Bonjean M, Maquet P (2010) Functional neuroimaging insights into the physiology of human sleep. Sleep 33:1589–1603
Dauvilliers Y, Boudousq V, Lopez R, De Cock VC (2011) Increased perfusion in supplementary area during a REM sleep behaviour episode. Sleep Med 12:531–532
Davis M (2000) The role of the amygdala in conditioned and unconditioned fear and anxiety. In: Aggleton JP (ed) The amygdala. Oxford University Press, New York, pp 213–288
De Carli F, Proserpio P, Morrone E, Sartori I, Ferrara M, Gibbs SA, De Gennaro L, Lo Russo G, Nobili L (2016) Activation of the motor cortex during phasic rapid eye movement sleep. Ann Neurol 79:326–330
De Cock VC, Vidailhet M, Leu S, Texeira A, Apartis E, Elbaz A, Roze E, Willer JC, Derenne JP, Agid Y, Arnulf I (2007) Restoration of motor control in Parkinson’s disease during REM sleep. Brain 130:450–456
De Cock VC, Debs R, Oudiette D, Leu S, Radji F, Tiberge M, Yu H, Bayard S, Roze E, Vidailhet M, Dauvilliers Y, Rascol O, Arnulf I (2011) The improvement of movement and speech during rapid eye movement sleep behaviour disorder in multiple system atrophy. Brain 134:856–862
Dean P, Redgrave P, Westby G (1989) Event or emergency? Two response systems in the mammalian superior colliculus. Trends Neurosci 12:137e47
Del Tredici K, Rub U, de Vos RA, Bohl JR, Braak H (2002) Where does Parkinson disease pathology begin in the brain? J Neuropathol Exp Neurol 61:413–426
Doppler K, Jentschke HM, Schulmeyer L, Vadasz D, Janzen A, Luster M, Höffken H, Mayer G, Brumberg J, Booij J, Musacchio T, Klebe S, Sittig-Wiegand E, Volkmann J, Sommer C, Oertel WH (2017) Dermal phospho-alpha-synuclein deposits confirm REM sleep behaviour disorder as prodromal Parkinson's disease. Acta Neuropathol 133:535–545
Ehrminger M, Latimier A, Pyatigorskaya N, Garcia-Lorenzo D, Leu-Semenescu S, Vidailhet M, Lehericy S, Arnulf I (2016) The coeruleus/subcoeruleus complex in idiopathic rapid eye movement sleep behaviour disorder. Brain 139:1180–1188
Evarts EV (1964) Temporal patterns of discharge of pyramidal tract neurons during sleep and waking in the monkey. J Neurophysiol 27:152–171
Fraigne JJ, Torontali ZA, Snow MB, Peever JH (2015) REM sleep at its core—circuits, neurotransmitters, and pathophysiology. Front Neurol 29:123. https://doi.org/10.3389/fneur.2015.00123
Frauscher B, Iranzo A, Högl B, Casanova-Molla J, Salamero M, Gschliesser V, Tolosa E, Poewe W, Santamaria J, SINBAR (Sleep Innsbruck Barcelona group) (2008) Quantification of electromyographic activity during REM sleep in multiple muscles in REM sleep behaviour disorder. Sleep 31:724–731
Fuller PM, Saper CB, Lu J (2007) The pontine REM switch: past and present. J Physiol 584-3:735–741
Fuller P, Sherman D, Pedersen N, Saper CB, Lu J (2011) Reassessment of the structural basis of the ascending arousal system. J Comp Neurol 519:933–956
Fung SJ, Xi M, Zhang J, Torterolo P, Sampogna S, Morales FR, Chase MH (2011) Projection neurons from the central nucleus of the amygdala to the nucleus pontis oralis. J Neurosci Res 89:429–436
Gagnon JF, Bédard MA, Fantini ML, Petit D, Panisset M, Rompré S, Carrier J, Montplaisir J (2002) REM sleep behavior disorder and REM sleep without atonia in Parkinson’s disease. Neurology 59:585–589
Gagnon JF, Postuma RB, Montplaisir J (2006) Update on the pharmacology of REM sleep behavior disorder. Neurology 67:742–747
Gelpi E, Höftberger R, Graus F et al (2016) Neuropathological criteria of anti-IgLON5-related tauopathy. Acta Neuropathol 132:531–543
Gerashchenko D, Blanco-Centurion CA, Miller JD, Shiromani PJ (2006) Insomnia following hypocretin2-saporin lesions of the substantia nigra. Neuroscience 137:29–36
Hendricks JC, Morrison AR (1981) Normal and abnormal sleep in animals. JAVMA 178:121–126
Hendricks JC, Morrison AR, Mann GL (1982) Different behaviors during paradoxical sleep without atonia depend on pontine lesion site. Brain Res 239:81–105
Högl B, Stefani A, Videnovic A (2018) Idiopathic REM sleep behaviour disorder and neurodegeneration—an update. Nat Rev Neurol 14:40–55
Holmes CJ, Jones BE (1994) Importance of cholinergic, GABAergic, serotonergic and other neurons in the medial medullary reticular formation for sleep-wake states studied by cytotoxic lesions in the cat. Neuroscience 62:1179–1200
Hsieh KC, Nguyen D, Siegel JM, Lai YY (2013) New pathways and data on rapid eye movement sleep behaviour disorder in a rat model. Sleep Med 14:719–728
Iranzo A, Aparicio J (2009) A lesson from anatomy: focal brain lesions causing REM sleep behavior disorder. Sleep Med 10:9–12
Iranzo A, Valldeoriola F, Santamaría J, Rumiá J, Tolosa E (2002) Sleep symptoms and polysomnographic architecture in advanced Parkinson’s disease after chronic bilateral subthalamic stimulation. J Neurol Neurosurg Psychiatry 72:661–664
Iranzo A, Santamaria J, Rye DB, Valldeoriola F, Marti MJ, Muñoz E, Vilaseca I, Tolosa E (2005) Characteristics of idiopathic REM sleep behavior disorder and that associated with MSA and PD. Neurology 65:247–252
Iranzo A, Graus F, Clover L, Morera J, Bruna J, Vilar C, Martínez-Rodriguez JE, Vincent A, Santamaría J (2006) Rapid eye movement sleep behavior disorder and potassium channel antibody-associated limbic encephalitis. Ann Neurol 59:178–182
Iranzo A, Frauscher B, Santos H, Gschliesser V, Ratti L, Falkenstetter T, Stürner C, Salamero M, Tolosa E, Poewe W, Santamaria J, Högl B, SINBAR (Sleep Innsbruck Barcelona) Group (2011) Usefulness of the SINBAR electromyographic montage to detect the motor and vocal manifestations occurring in REM sleep behaviour disorder. Sleep Med 12:284–288
Iranzo A, Tolosa E, Gelpi E, Molinuevo JL, Valldeoriola F, Serradell M, Sanchez-Valle R, Vilaseca I, Lomeña F, Vilas D, Lladó A, Gaig C, Santamaria J (2013) Neurodegenerative status and postmortem pathology in idiopathic rapid-eye-movement behavior disorder: an observational cohort study. Lancet Neurol 12:443–453
Iranzo A, Gelpi E, Tolosa E, Molinuevo JL, Serradell M, Gaig C, Santamaria J (2014) Neuropathology of prodromal Lewy body disease. Mov Disord 29:410–415
Iranzo A, Santamaria J, Tolosa E (2016) Idiopathic rapid eye movement sleep behaviour disorder: diagnosis, management, and the need for neuroprotective interventions. Lancet Neurol 15:405–419
Iranzo A, Santamaría J, Valldeoriola F, Monica S, Salamero M, Carles G, Niñerola-Baizán A, Sánchez-Valle R, Lladó A, De Marzi R, Stefani A, Seppi K, Pavia J, Högl B, Poewe W, Tolosa E, Lomeña F (2017) Dopamine transporter imaging deficit predicts early transition to synucleinopathy in idiopathic rapid eye movement sleep behavior disorder. Ann Neurol 82:419–428
Irwin MR (2015) Why sleep is important for health: a psychoneuroimmunology perspective. Annu Rev Psychol 66:143–172
Jouvet M, Delrome F (1965) Locus coeruleus et sommeil paradoxal. C R Soc Biol 159:895–899
Jürgens U (2002) Neural pathways underlying vocal control. Neurosci Biobehav Reviews 26:235–258
Jürgens U (2009) The neural control of vocalization in mammals: a review. J Voice 23:1–10
Krenzer M, Anaclet C, Vetrivelan R, Wang N, Vong L, Lowell BB, Fulller PM, Lu J (2011) Brainstem and spinal cord circuitry regulating REM sleep and muscle atonia. PLoS One 6(10):e24998
Krenzer M, Lu J, Mayer G, Oertel W (2013) From bench to bed: putative animal models of REM sleep behavior disorder (RBD). J Neural Transm 120:683–688
Kumru H, Iranzo A, Carrasco E, Valldeoriola F, Marti MJ, Santamaria J, Tolosa E (2008) Lack of effects of pramipexole on REM sleep behavior disorder in Parkinson’s disease. Sleep 31:1418–1421
Kunz D, Bes F (1999) Melatonin as a therapy in REM sleep behavior disorder patients: an open-labeled pilot study on the possible influence of melatonin on REM-sleep regulation. Mov Disord 14:507–511
Lai YY, Hsieh KC, Nguyen D, Peever J, Siegel JM (2008) Neurotoxic lesions at the ventral mesopontine junction change sleep time and muscle activity during sleep: an animal model of motor disorders in sleep. Neuroscience 154:431–443
Lapierre O, Montplaisir J (1992) Polysomnographic features of REM sleep behavior disorder: development of a scoring method. Neurology 42:1371–1374
Li D, Peever J (2015) Pharmacogenetic stimulation of the red nucleus influences muscle tone during rapid eye movement (REM) sleep in mice. Sleep 37:A21
Lu J, Sherman D, Devor M, Saper CB (2006) A putative flip-flop switch for control of REM sleep. Nature 441:589–594
Luppi PH, Gervasono D, Verret L et al (2007) Paradoxical (REM) sleep genesis: the switch from an aminergic-cholinergic to a GABAergic-glutamatergic hypothesis. J Phsiol 100:271–283
Luppi PH, Clement O, Sapin E et al (2011) The neuronal network responsible for paradoxical sleep and its dysfunctions causing narcolepsy and rapid eye movement (REM) behavior disorder. Sleep Med Rev 15:153–163
Luppi PH, Clement O, Valencia Garcia S, Brischoux F, Fort P (2013) New aspects in the pathophysiology of rapid eye movement behavior disorder: the potential role of glutamate, gamma-aminobutyric acid, and glycine. Sleep Med 14:714–718
Luppi PH, Billwiller F, Fort P (2017) Selective activation of a few limbic structures during paradoxical (REM) sleep by the claustrum and the suprammamilary nucleus: evidence and function. Curr Opin Neurobiol 44:59–64
Mander BA, Winer JR, Walker MP (2017) Sleep and human aging. Neuron 94:19–36
Manni R, Terzaghi M, Glorioso M (2009) Motor-behavioral episodes in REM sleep behavior disorder and phasic events during sleep. Sleep 32:241–245
Mayer G, Bitterlich M, Kuwert T, Ritt P, Stefan H (2015) Ictal SPECT in patients with rapid eye movement sleep behaviour disorder. Brain 138:1263–1270
McKenna D, Peever J (2017) Degeneration of rapid eye movement sleep circuitry underlies rapid eye movement sleep behavior disorder. Mov Disord 32:636–644
Morrison AR, Mann GL, Hendrijks JC (1981) The relationship of excessive exploratory behavior in wakefulness to paradoxical sleep without atonia. Sleep 4:247–257
Oudiette D, De Cock VC, Lavault S, Leu S, Vidailhet M, Arnulf I (2009) Nonviolent elaborate behaviors may also occur in REM sleep behavior disorder. Neurology 72:551–557
Peever J, Fuller PM (2017) The biology of REM sleep. Curr Biol 27:R1237–R1248
Peever L, Luppi H, Montplaisir J (2014) Breakdown in REM sleep circuitry underlies REM sleep behavior disorder. Trends Neurosci 37:279–288
Postuma RB, Gagnon JF, Bertrand JA, Génier Marchand D, Montplaisir JY (2015) Parkinson risk in idiopathic REM sleep behavior disorder: preparing for neuroprotective trials. Neurology 84:1104–1113
Ramaligam V, Chen MC, Saper CB, Lu J (2013) Perspectives on the rapid eye movement sleep switch in rapid eye movement sleep behavior disorder. Sleep Med 14:707–7013
Recasens A, Dehay B, Bove J, Carballo-Carbajal I, Dovero S, Perez-Villalba A, Fernagut PO, Blesa J, Parent A, Perier C, Fariñas I, Obeso JA, Bezard E, Vila M (2014) Lewy body extracts from Parkinson disease brains trigger a-synuclein pathology and neurodegeneration in mice and monkeys. Ann Neurol 75:351–362
Reinoso-Suárez F, de Andrés I, Rodrigo-Angulo ML, Garzón M (2001) Brain structures and mechanisms involved in the generation of REM sleep. Sleep Med Rev 5:67–77
Rye DB (1997) Contributions of the pedunculopontine region to normal and altered REM sleep. Sleep 20:855–890
Rye DB, Bliwise DL (2004) Movement disorders specific to sleep and the nocturnal manifestations of waking movement disorders. In: Watts RL, Koller WC (eds) Movement disorders: neurologic principles and practice, 2nd edn. McGraw Hill, New York, pp 855–890
Sabater L, Gaig C, Gelpi E, Bataller L, Lewerenz J, Torres-Vega E, Contreras A, Giometto B, Compta Y, Embid C, Vilaseca I, Iranzo A, Santamaría J, Dalmau J, Graus F (2014) A novel non-rapid-eye movement and rapid-eye-movement parasomnia with sleep breathing disorder associated with antibodies to IgLON5: a case series, characterisation of the antigen, and post-mortem study. Lancet Neurol 13:575–586
Sakai K, Kanamori N, Jouvet M (1979) Neuronal activity specific to paradoxical sleep in the bulbar reticular formation in the unrestrained cat. C R Seances Acad Sci D 289:557–561
Sandford LD, Cheng CS, Sivestri AJ (2001) Sleep and behavior in rats with pontine lesions producing REM without atonia. Sleep Research Online 4:1–5
Scammel TE, Arrigoni E, Lipton JO (2017) Neural circuitry of wakefulness and sleep. Neuron 93:747–765
Schenck CH, Mahowald MW (1992) Motor dyscontrol in narcolepsy: rapid-eye-movement (REM) sleep without atonia and REM sleep behaviour disorder. Ann Neurol 32:3–10
Schenck CH, Mahowald MW (2002) REM sleep behavior disorder: clinical, developmental, and neuroscience perspectives 16 years after its formal identification in SLEEP. Sleep 25:120–138
Schenck CH, Bundlie SR, Ettinger MG, Mahowald MW (1986) Chronic behavioural disorders of human REM sleep: a new category of parasomnia. Sleep 9:293–308
Schenck CH, Bundlie SR, Patterson AL, Mahowald MW (1987) Rapid eye movement sleep behavior disorder. A treatable parasomnia affecting older adults. JAMA 257:1786–1789
Schenck CH, Bundlie SR, Mahowald MW (1996) Delayed emergence of a parkinsonian disorder in 38% of 29 older men initially diagnosed with idiopathic rapid eye movement sleep behavior disorder. Neurology 46:388–392
Schenckel E, Siegel JM (1989) REM sleep without atonia after lesions of the medial medulla. Neursci Lett 98:159–165
Scherfler C, Frauscher B, Schocke M, Iranzo A, Gschliesser V, Seppi K, Santamaria J, Tolosa E, Högl B, Poewe W, SINBAR (Sleep Innsbruck Barcelona) Group (2011) With and gray matter abnormalities in idiopathic rapid eye movement disorder: a diffusion-tensor imaging and voxel-based morphometry study. Ann Neurol 12:284–288
Shouse MN, Siegel JM (1992) Pontine regulation of REM sleep components in cats: integrity of the pedunculopontine tegmentum (PPT) is important for phasic events but unnecessary for atonia during REM sleep. Brain Res 571:50–63
Siegel J (2009) The neurobiology of sleep. Semin Neurol 29:277–296
Silber MH, Krahan LE, Morgenthaler T (2004) Physiological basis of sleep. In: Silber MH, Krahan LE, Morgenthaler T (eds) Sleep medicine in clinical practice. Taylor & Francis, London, pp 3–24
Sprenger FS, Stefanova N, Gelpi E, Seppi K, Navarro-Otano J, Offner F, Vilas D, Valldeoriola F, Pont-Sunyer C, Aldecoa I, Gaig C, Gines A, Cuatrecasas M, Högl B, Frauscher B, Iranzo A, Wenning GK, Vogel W, Tolosa E, Poewe W (2015) Enteric nervous system α-synuclein immunoreactivity in idiopathic REM sleep behavior disorder. Neurology 85:1761–1768
Stefani A, Gabelia D, Mitterling T, Poewe W, Högl B, Frauscher B (2015) A prospective video-polysomnographic analysis of movements during physiological sleep in 100 healthy sleepers. Sleep 38:1479–1487
Tamgüney G, Korczyn AD (2017) A critical review of the prion hypothesis of human synucleinopathies. Cell Tissue Res 8. https://doi.org/10.1007/s00441-017-2712-y
Tiriac A, Uitermarkt BD, Fanning AS, Sokoloff G, Blumberg MS (2012) Rapid whisker movements in sleep in new born rats. Curr Biol 22:2075–2080
Tiriac A, Del Rio-Bermudez C, Blumberg MS (2014) Self-generated movements with “unexpected” sensory consequences. Curr Biol 24:2136–2141
Valencia Garcia S, Libourel PA, Lazarus M, Grassi D, Luppi PH, Fort P (2017) Genetic inactivation of glutamate neurons in the rat sublaterodorsal tegmental nucleus recapitulates REM sleep behaviour disorder. Brain 140:414–428
Valencia Garcia S, Brischoux F, Clément O, Libourel PA, Arthaud S, Lazarus M, Luppi PH, Fort P (2018) Ventromedial medulla inhibitory neuron inactivation induces REM sleep without atonia and REM sleep behavior disorder. Nat Commun 9:504. https://doi.org/10.1038/s41467-017-02761-0
Verhave PS, Jongsma MJ, Van den Berg RM, Vis JC, Vanwersch RA, Smit AB, Van Someren EJ, Philippens IH (2011) REM sleep behaviour disorder in the marmoset MPTP model of early Parkinson disease. Sleep 34:119–1125
Vetrivelan R, Fuller PM, Tong Q, Lu J (2009) Medullary circuitry regulating rapid eye movement sleep and motor atonia. J Neurosci 29:9361–9369
Vilas D, Iranzo A, Tolosa E, Aldecoa I, Berenguer J, Vilaseca I, Martí C, Serradell M, Lomeña F, Alós L, Gaig C, Santamaria J, Gelpi E (2016) Assessment of α-synuclein in submandibular glands of patients with idiopathic rapid-eye-movement sleep behaviour disorder: a case-control study. Lancet Neurol 15:708–718
Weber F, Chung S, Beier KT, Xu M, Luo L, Dan Y (2015) Control of REM sleep by ventral medulla GABAergic neurons. Nature 526:435–438
Wu FS, Yang YC, Tsai JJ (1999) Melatonin potentiates the GABA(A) receptor-mediated current in cultured chick spinal cord neurons. Neurosci Lett 260:177–180
Zagrodzka J, Hedberg CE, Mann GL, Morrison AR (1998) Contrasting expressions of aggressive behavior related by lesions of the central nucleus of the amygdala during wakefulness and rapid eye movement sleep without atonia in cats. Behav Neurosci 112:589–602
Acknowledgments
I thank Dr. Ellen Gelpi for providing Fig. 10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iranzo, A. The REM sleep circuit and how its impairment leads to REM sleep behavior disorder. Cell Tissue Res 373, 245–266 (2018). https://doi.org/10.1007/s00441-018-2852-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-018-2852-8