Abstract
Purpose
To investigate PRL-3 protein expression in normal colorectal epithelia and colorectal cancers with monoclonal antibody (MAb) against PRL-3.
Methods
MAb against PRL-3 was prepared with the hybridoma technique, and its specificity was confirmed with ELISA and Western blotting assays. The expression of PRL-3 protein in normal colorectal epithelia and colorectal cancers was examined by immunohistochemistry assay. Logistic regression and survival analysis were performed to determine the clinical significance of PRL-3 expression.
Results
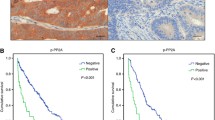
MAb 3B6 against PRL-3 was obtained and showed high specificity. PRL-3 protein was expressed in two of 28 (7.1%) normal colorectal epithelia, 21 of 88 (23.9%) primary colorectal cancers, 22 of 41 (53.7%) metastatic lymph nodes and eight of 12 (66.7%) liver metastases, respectively. The PRL-3 expression rates of metastases were significantly higher than those of primary colorectal cancers and normal colorectal epithelia (P <0.05). PRL-3 expression was significantly associated with the liver metastasis of colorectal cancer (P = 0.004) and tended to shorten survival time (P = 0.0145).
Conclusions
This is the first study demonstrating that PRL-3 is a potential marker for liver metastasis of colorectal cancer and negatively influences the prognosis of colorectal cancer patients.




Similar content being viewed by others
References
Brown-Shimer S, Johnson KA, Hill DE, Bruskin AM (1992) Effect of protein tyrosine phosphatase 1B expression on transformation by the human neu oncogene. Cancer Res 52:478–482
Cangi MG, Cukor B, Soung P, Signoretti S, Moreira G Jr, Ranashinge M, Cady B, Pagano M, Loda M (2000) Role of Cdc25A phosphatase in human breast cancer. J Clin Invest 106:753–761
Di Cristofano A, Pandolfi PP (2000) The multiple roles of PTEN in tumor suppression. Cell 100:387–390
de St. Groth SF, Scheidegger D (1980) Production of monoclonal antibodies strategy and tactics. J Immunol Methods 35:1–21
Fuller SA, Takahaski M, Hurrell JG (2002) Immunology. In: Ausubel FM, Brent R, Kingston RE, Moore D, Seidman JG, Smith J, Struhl K (eds) Current protocols in molecular biology, vol. 2. Wiley, New York, pp 11.1.1–11.11.4
Galaktionov K, Lee AK, Eckstein J, Draetta G, Meckler J, Loda M, Beach D (1995) Cdc25 phosphatases as potential human oncogenes. Science 269:1575–1577
Hunter T (2000) Signaling-2000 and beyond. Cell 100:113–127
Lindblom A, Linder S (1996) The metastatic phenotype—prognostic implications. Crit Rev Oncol Hematol 24:71–96
Ma D (2001) Tumor metastsis. In: Zeng YX (eds) Oncology. People Health, Beijing, pp 183–185
Matter WF, Estridge T, Zhang C, Belagaje R, Stancato L, Dixon J, Johnson B, Bloem L, Pickard T, Donaghue M, Acton S, Jeyaseelan R, Kadambi V, Vlahos CJ (2001) Role of PRL-3, a human muscle-specific tyrosine phosphatase, in angiotensin-II signaling. Biochem Biophys Res Commun 283:1061–1068
Neel BG, Tonks NK (1997) Protein tyrosine phosphatase in signal transduction. Curr Opin Cell Biol 9:193–204
Ponniah S, Wang DZ, Lim KL, Pallen CJ (1999) Targeted disruption of the tyrosine phosphatase PTPalpha leads to constitutive downregulation of the kinases Src and Fyn. Curr Biol 9:535–538
Saha S, Bardelli A, Buckhaults P, Velculescu VE, Rago C, St Croix B, Romans KE, Choti MA, Lengauer C, Kinzler KW, Vogelstein B (2001) A phosphatase associated with metastasis of colorectal cancer. Science 294:1343–1346
Sobin LH, Witterind C (1997) International Union Against Cancer. TNM classification of maglignant tumors (5th edn). Wiley, New York, p 53
Su J, Muranjian M, Sap J (1999) Receptor protein tyrosine phosphatase alpha activiates Src-family kinases and controls integrin-mediated responses in fibroblasts. Curr Biol 9:505–511
Susan A, Schumacher BU (2001) Metastasis research protocols, vol. 1. Humana, New Jersey, p 10
Tan YH (1993) Yin and yang of phosphorylation in cytokine signaling. Science 262:376–377
Zeng Q, Hong W, Tan YH (1998) Mouse PRL-2 and PRL-3, two potentially prenylated protein tyrosine phosphatases homologous to PRL-1. Biochem Biophys Res Commun 244:421–427
Zeng Q, Dong JM, Guo K, Li J, Tan HX, Koh V, Pallen CJ, Manser E, Hong W (2003) PRL-3 and PRL-1 Promote cell migration, invasion, and metastasis. Cancer Res 63:2716–2722
Zheng XM, Wang Y, Pallen CJ (1992) Cell transformation and activation of pp60-src by overexpression of a protein tyrosine phosphatase. Nature 359:336–339
Acknowledgements
The authors thank Dr. Jiyou Li, Dr. Jing Gu, Dr. Jiafu Ji, and Dr. Jingsheng He of Beijing Cancer Hospital for providing the tissue specimens
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the State Key Basic Research Program of China (G19980512). Key Project of Beijing Scientific Technology Committee (H020220020310), and Peking University Key Program of Oncology.
Rights and permissions
About this article
Cite this article
Peng, L., Ning, J., Meng, L. et al. The association of the expression level of protein tyrosine phosphatase PRL-3 protein with liver metastasis and prognosis of patients with colorectal cancer. J Cancer Res Clin Oncol 130, 521–526 (2004). https://doi.org/10.1007/s00432-004-0563-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-004-0563-x