Abstract
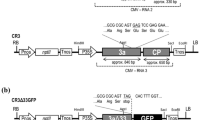
We have developed Cucumber mosaic virus (CMV) as a plant virus vector especially for production of pharmaceutical proteins. The CMV vector is a vector modifiable for different host plants and does not require further engineering steps. CMV contains three genomic RNA molecules (RNAs 1–3) necessary for infectivity. With this system, instead of creating different vector constructs for each plant we use, we take advantage of the formation of pseudrecombinants between two CMV isolates by simply reassembling a vector construct (RNA 2 base) and an RNA molecule containing the host determinant (mostly RNA 3). In this study, the gene for acidic fibroblast growth factor (aFGF), one of the human cytokines, was cloned under the control of the subgenomic promoter for RNA 4A of the CMV-based vector, C2-H1. Infected Nicotiana benthamiana plants produced aFGF at levels up to 5–8% of the total soluble protein. The tobacco-produced aFGF was purified, and its biological activity was confirmed. Using this system, which provides a versatile and viable strategy for the production of therapeutic proteins in plants, we also demonstrated a high level of aFGF in Glycine max (soybean) and Arabidopsis thaliana.







Similar content being viewed by others
Abbreviations
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- aFGF:
-
Acidic fibroblast growth factor
- CMV:
-
Cucumber mosaic virus
- TMV:
-
Tobacco mosaic virus
References
Arazi T, Slutsky SG, Shiboleth YM, Wang Y, Rubinstein M, Barak S, Yang J, Gal-On A (2001) Engineering zucchini yellow mosaic potyvirus as a non-pathogenic vector for expression of heterologous proteins in cucurbits. J Biotechnol 87:67–82
Brigneti G, Voinnet O, Li WX, Ji LH, Ding SW, Baulcombe DC (1998) Viral pathogenicity determinants are suppressors of transgene silencing in Nicotiana benthamiana. EMBO J 17:6739–6746
Burgess WH (1991) Structure–function studies of acidic fibroblast growth factor. Ann N Y Acad Sci 638:89–97
Burgess WH, Maciag T (1989) The heparin-binding (fibroblast) growth factor family of proteins. Ann Rev Biochem 58:575–606
Carrère L, Tepfer M, Jacquemond M (1999) Recombinants of cucumber mosaic virus (CMV): determinants of host range and symptomatology. Arch Virol 144:365–379
Charmandari E, Pincus SM, Matthews DR, Johnston A, Brook CG, Hindmarsh PC (2002) Oral hydrocortisone administration in children with classic 21-hydroxylase deficiency leads to more synchronous joint GH and cortisol secretion. J Clin Endocrinol Metab 87:2238–2244
Cummins MJ, Pruitt B (1999) Low-dose oral use of human interferon-alpha in cancer patients. J Interferon Cytokine Res 19:937–941
De Zoeten GA, Penswick JR, Horisberger MA, Ahl P, Schultze M, Hohn T (1989) The expression, localization, and effect of a human interferon in plants. Virology 172:213–222
Ding B, Li Q, Nguyen L, Palukaitis P, Lucas WJ (1995) Cucumber mosaic virus 3a protein potentiates cell-to-cell trafficking of CMV RNA in tobacco plants. Virology 207:345–353
Dirnberger D, Steinkellner H, Abdennebi L, Remy JJ, van deWiel D (2001) Secretion of biologically active glycoforms of bovine follicle stimulating hormone in plants. Eur J Biochem 268:4570–4579
Edwardson JR, Christie RG (1991) Cucumoviruses. In: CRC Handbook of viruses infecting legumes, CRC Press, Boca Raton, pp 293–319
Gils M, Kandzia R, Marillonnet S, Klimyuk V, Bleba Y (2005) High-yield production of authentic human growth hormone using a plant virus-based expression system. Plant Biotech J 3:613–620
Hong JS, Masuta C, Nakano M, Abe J, Uyeda I (2003) Adaptation of Cucumber mosaic virus soybean strains (SSVs) to cultivated and wild soybeans. Theor Appl Genet 107:49–53
James EA, Wang C, Wang Z, Reeves R, Shin JH, Magnuson NS, Lee JM (2000) Production and characterization of biologically active human GM-CSF secreted by genetically modified plant cells. Protein Expr Purif 19:131–138
Kwon TH, Seo JE, Kim J, Lee JH, Jang YS, Yang MS (2003) Expression and secretion of the heterodimeric protein interleukin-12 in plant cell suspension culture. Biotechnol Bioeng 81:870–875
Landgren E, Klint P, Yokote K, Claesson-Welsh L (1998) Fibroblast growth factor receptor-1 mediates chemotaxis independently of direct SH2-domain protein binding. Oncogene 17:283–291
Lee HJ (2002) Protein drug oral delivery: the recent progress. Arch Pharm Res 25:572–584
Lee JH, Kim NS, Kwon TH, Jang YS, Yang MS (2002) Increased production of human granulocyte-macrophage colony stimulating factor (hGM-CSF) by the addition of stabilizing polymer in plant suspension cultures. J Biotechnol 96:205–211
Ma S, Huang Y, Davis A, Yin Z, Mi Q, Menassa R, Brandle JE, Jevnikar AM (2005) Production of biologically active human interleukin-4 in transgenic tobacco and potato. Plant Biotechnol J 3:309–318
Magnuson NS, Linzmaier PM, Reeves R, An G, HayGlass K, Lee JM (1998) Secretion of biologically active human interleukin-2 and interleukin-4 from genetically modified tobacco cells in suspension culture. Protein Expr Purif 13:45–52
Marcus PI, van der Heide L, Sekellick MJ (1999) Interferon action on avian viruses. I. Oral administration of chicken interferon-alpha ameliorates Newcastle disease. J Interferon Cytokine Res 19:881–885
Marillonnet S, Giritch A, Gils M, Kandzia R, Klimyuk V, Gleba Y (2004) In planta engineering of viral RNA replicons: efficient assembly by recombination of DNA modules delivered by Agrobacterium. Proc Natl Acad Sci USA 101:6852–6857
Matsumoto S, Ikura K, Ueda M, Sasaki R (1995) Characterization of a human glycoprotein (erythropoietin) produced in cultured tobacco cells. Plant Mol Biol 27:1163–1172
Matsumura T, Ohya K, Itchouda N, Sugimoto C (2000) Expression of human interferon alpha and tumor necrosis factor alpha in plants. In: Toutant JP, Balázs E (eds) Molecular farming. INRA Editions, Versailles Cedex, pp 177–185
Matuo Y, Nishi N, Matsumoto K, Miyazaki K, Matsumoto K, Suzuki F, Nishikawa K (1991) 3-[(3-cholamidopropyl)dimethylammonio]-1-propane sulfonate as noncytotoxic stabilizing agent for growth factors. Methods Enzymol 198:511–518
Menassa R, Jevnikar A, Brandle J (2000) A contained system for the field production of plant recombinant proteins. In: Toutant JP, Balázs E (eds) Molecular farming. INRA Editions, Versailles Cedex, pp 197–205
Mori M, Fujihara N, Mise K, Furusawa I (2001) Inducible high-level mRNA amplification system by viral replicase in transgenic plants. Plant J 27:79–86
Nitta N, Takanami Y, Kuwata S, Kubo S (1988) Inoculation with RNAs 1 and 2 of cucumber mosaic virus induces viral RNA replicase activity in tobacco mesophyll protoplasts. J Gen Virol 69:2695–2700
Ohya K, Itchoda N, Ohashi K, Onuma M, Sugimoto C, Matsumura T (2002) Expression of biologically active human tumor necrosis factor-alpha in transgenic potato plant. J Interferon Cytokine Res 22:371–378
Palukaitis P, García-Arenal F (2003) Cucumoviruses. Adv Virus Res 62:241–323
Park Y, Cheong H (2002) Expression and production of recombinant human interleukin-2 in potato plants. Protein Expr Purif 25:160–165
Pérez Filgueira DM, Zamorano PI, Domínguez MG, Taboga O, Del Médico Zajac MP, Puntel M, Romera SA, Morris TJ, Borca MV, Sadir AM (2003) Bovine herpes virus gD protein produced in plants using a recombinant tobacco mosaic virus (TMV) vector possesses authentic antigenicity. Vaccine 21:4201–4209
Sardana RK, Alli Z, Dudani A, Tackaberry E, Panahi M, Narayanan M, Ganz P, Altosaar I (2002) Biological activity of human granulocyte-macrophage colony stimulating factor is maintained in a fusion with seed glutelin peptide. Transgenic Res 11:521–531
Sawahel WA (2002) The production of transgenic potato plants expressing human alpha-interferon using lipofectin-mediated transformation. Cell Mol Biol Lett 7:19–29
Schwinghamer MW, Symons RH (1975) Fractionation of cucumber mosaic virus RNA and its translation in a wheat embryo cell-free system. Virology 63:252–262
Senda M, Masuta C, Ohnishi S, Goto K, Kasai A, Sano T, Hong JS, MacFarlane S (2004) Patterning of virus-infected Glycine max seed coat is associated with suppression of endogenous silencing of chalcone synthase genes. Plant Cell 16:807–818
Shin YJ, Hong SY, Kwon TH, Jang YS, Yang MS (2003) High level of expression of recombinant human granulocyte-macrophage colony stimulating factor in transgenic rice cell suspension culture. Biotechnol Bioeng 82:778–783
Suzuki M, Kuwata S, Kataoka J, Masuta C, Nitta N, Takanami Y (1991) Functional analysis of deletion mutants of cucumber mosaic virus RNA3 using an in vitro transcription system. Virology 183:106–113
Wagner B, Hufnagl K, Radauer C, Wagner S, Baier K, Scheiner O, Wiedermann U, Breiteneder H (2004) Expression of the B subunit of the heat-labile enterotoxin of Escherichia coli in tobacco mosaic virus-infected Nicotiana benthamiana plants and its characterization as mucosal immunogen and adjuvant. J Immunol Methods 287:203–215
Wang ZD, Ueda S, Uyeda I, Yagihashi H, Sekiguchi H, Takahashi Y, Sato M, Ohya K, Sugimoto C, Matsumura T (2003) Positional effect of gene insertion on genetic stability of a clover yellow vein virus-based expression vector. J Gen Plant Pathol 69:327–334
Yamaguchi N, Seshimo Y, Yoshimoto E, Ahn HI, Ryu KH, Choi JK, Masuta C (2005) Genetic mapping of the compatibility between a lily isolate of Cucumber mosaic virus and a satellite RNA. J Gen Virol 86:2359–2369
Zhao Y, Hammond J, Tousignant ME, Hammond RW (2000) Development and evaluation of a complementation-dependent gene delivery system based on cucumber mosaic virus. Arch Virol 145:2285–2295
Acknowledgments
We would like to thank Dr. I. Uyeda (Hokkaido University, Sapporo, Japan) for reviewing the manuscript. This work was supported in part by Grants-in-Aid for Scientific Research (B) (no. 17380197) from the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsuo, K., Hong, J.S., Tabayashi, N. et al. Development of Cucumber mosaic virus as a vector modifiable for different host species to produce therapeutic proteins. Planta 225, 277–286 (2007). https://doi.org/10.1007/s00425-006-0346-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0346-5