Abstract
Background
To evaluate the efficacy of intravitreal bevacizumab injection in the treatment of central serous chorioretinopathy (CSC).
Methods
In a prospective interventional case series, 40 eyes of 40 patients with idiopathic CSC at least 3 months in duration were included. Patients were treated with once or twice intravitreal bevacizumab injections initially and completed at least 12 months follow-up. Main outcomes measures were the resolution of neurosensory detachment, best-corrected visual acuity, and findings on fluorescein and indocyanine green angiography.
Results
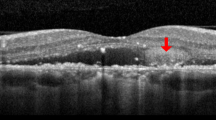
Thirty-three of 40 eyes (82.5%) showed complete absence of subretinal fluid at the macula within 3 months following initial intravitreal bevacizumab injection. Eyes exhibiting resolution of subretinal fluid revealed improvement in fluorescein and indocyanine green angiographic findings. The rate of intense hyperfluorescence on indocyanine green angiography was higher in eyes exhibiting resolution of subretinal fluid than eyes exhibiting incomplete absorption of subretinal fluid after intravitreal bevacizumab (72.7 and 28.5%, respectively, p = 0.039).
Conclusions
Intravitreal bevacizumab injections generally resulted in anatomic improvement for CSC and may constitute a therapeutic option in CSC. The effect after intravitreal bevacizumab injection for CSC might be related to the hyperfluorescence on indocyanine green angiography.



Similar content being viewed by others
References
Levine R, Brucker AJ, Robinson F (1989) Long-term follow-up of idiopathic central serous chorioretinopathy by fluorescein angiography. Ophthalmology 96:854–859
Hussain D, Gass JD (1998) Idiopathic central serous chorioretinopathy. Indian J Ophthalmol 46:131–137
Loo RH, Scott IU, Flynn HW Jr, Gass JD, Murray TG, Lewis ML, Rosenfeld PJ, Smiddy WE (2002) Factors associated with reduced visual acuity during long-term follow-up patients with idiopathic central serous chorioretinopathy. Retina 22:19–24
Robertson DM, Ilstrup D (1983) Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am J Ophthalmol 95:457–466
Taban M, Boyer DS, Thomas EL, Taban M (2004) Chronic central serous chorioretinopathy: photodynamic therapy. Am J Ophthalmol 137:1073–1080
Chan WM, Lai TY, Lai RY, Liu DT, Lam DS (2008) Half-dose verteporfin photodynamic therapy for acute central serous chorioretinopathy: one-year results of a randomized controlled trial. Ophthalmology 115:1756–1765
Battaglia Parodi M, Da Pozzo S, Ravalico G (2003) Photodynamic therapy in chronic central serous chorioretinopathy. Retina 23:235–237
Torres-Soriano ME, Garcia-Aguirre G, Kon-Jara V, Ustariz-Gonzáles O, Abraham-Marín M, Ober MD, Quiroz-Mercado H (2008) A pilot study of intravitreal bevacizumab for the treatment of central serous chorioretinopathy (case reports). Graefes Arch Clin Exp Ophthalmol 246:1235–1239
Schaal KB, Hoeh AE, Scheuerle A, Schuett F, Dithmar S (2009) Intravitreal bevacizumab for treatment of chronic central serous chorioretinopathy. Eur J Ophthalmol 19:613–617
Seong HK, Bae JH, Kim ES, Han JR, Nam WH, Kim HK (2009) Intravitreal bevacizumab to treat acute central serous chorioretinopathy: short-term effect. Ophthalmologica 223:343–347
Lim SJ, Roh MI, Kwon OW (2010) Intravitreal bevacizumab injection for central serous chorioretinopathy. Retina 30:100–106
Artunay O, Yuzbasioglu E, Rasier R, Sengul A, Bahcecioglu H (2010) Intravitreal bevacizumab in treatment of idiopathic persistent central serous chorioretinopathy: a prospective, controlled clinical study. Curr Eye Res 35:91–98
Guyer DR, Yannuzzi LA, Slakter JS, Sorenson JA, Ho A, Orlock D (1994) Digital indocyanine green videoangiography of central serous chorioretinopathy. Arch Ophthalmol 112:1057–1062
Spaide RF, Hall L, Haas A, Campeas L, Yannuzzi LA, Fisher YL, Guyer DR, Slakter JS, Sorenson JA, Orlock DA (1996) Indocyanine green videoangiography of older patients with central serous chorioretinopathy. Retina 16:203–213
Prünte C, Flammer J (1996) Choroidal capillary and venous congestion in central serous chorioretinopathy. Am J Ophthalmol 9:37–41
Piccolino FC, Borgia L, Zinicola E, Zingirian M (1995) Indocyanine green angiographic findings in central serous chorioretinopathy. Eye 9:324–332
Iida T, Kishi S, Hagimura N, Shimizu K (1999) Persistent and bilateral choroidal vascular abnormalities in central serous chorioretinopathy. Retina 19:508–551
Peters S, Heiduschka P, Julien S, Ziemssen F, Fietz H, Bartz-Schmidt KU (2007) Tübingen bevacizumab study group, Schraermeyer U: Ultrastructural findings in primate eye after intravitreal injection of bevacimuzab. Am J Ophthalmol 143:995–1002
Peters S, Julien S, Heiduschka P, Grisanti S, Ziemssen F, Adler M, Schraermeyer U, Bartz-Schmidt KU (2007) Antipermeability and antiproliferative effects of standard and frozen bevacizumab on choroidal endothelial cells. Br J Ophthalmol 91:827–831
Lim JW, Kim MU, Shin MC (2010) Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina 30:1465–1471
Inoue R, Sawa M, Tsujikawa M, Gomi F (2010) Association between the efficacy of photodynamic therapy and indocyanine green angiography findings for central serous chorioretinopathy. Am J Ophthalmol 149:441–446
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Clinical trial registration ID
NCT00864773
Rights and permissions
About this article
Cite this article
Lim, J.W., Kim, M.U. The efficacy of intravitreal bevacizumab for idiopathic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 249, 969–974 (2011). https://doi.org/10.1007/s00417-010-1581-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-010-1581-9