Abstract
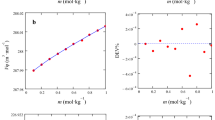
Densities, conductivities, and fluorescence spectra of two imidazolium ionic liquids ([Cnmim]Br, n = 10, 14)-glycyl dipeptide-water mixtures were measured as a function of temperature. The density data have been utilized to calculate the apparent molar volumes, standard partial molar volumes (V 02,φ ), standard partial molar volumes of transfer from water to aqueous ionic liquids solutions (Δt V°), the hydration number (N H), partial molar expansibility (E 0φ ), and Hepler’s constant of glycyl dipeptides. Through the electrical conductivity measurements, the c cmc values at different temperatures and a series of thermodynamic parameters (ΔG o m , ΔH o m , and ΔS o m ) of micellization of [Cnmim]Br (n = 10, 14) in aqueous glycyl dipeptides solutions are evaluated. The pyrene fluorescence spectra were used to study the change of micropolarity produced by the interaction of [Cnmim]Br with glycyl dipeptide, and the aggregation behavior of [Cnmim]Br (n = 10, 14). The results shown above have been explained in terms of solute–solvent interactions and structural changes in the mixed solutions. The interaction between [Cnmim]Br (n = 10, 14) and glycyl dipeptide is affected by temperature and hydrocarbon chain length of the dipeptides.







Similar content being viewed by others
References
Rogers RD, Seddon KR (2003) Science 302:792–793
Welton T (1999) Chem Rev 99:2071–2084
Wang JJ, Zhang LM, Wang HY, Wu CZ (2011) J Phys Chem B 115:4955–4962
Hemmateenejad B, Safavi A, Dorostkar S (2011) J Mol Liq 160:35–39
Fang S, Ren DH (2013) J Chem Eng Data 58:845–850
Shekaari H, Jebali F (2010) J Solution Chem 39:1409–1427
Shekaari H, Jebali F (2010) J Chem Eng Data 55:2517–2523
Tome LIN, Jorge M, Gomes JRB, Coutinho JAP (2010) J Phys Chem B 114:16450–16459
Singh V, Chhotaray PK, Gardas RL (2014) J Chem Thermodyn 71:37–49
Shekaari H, Kazempour A (2012) J Taiwan Inst Chem E 43:650–657
Shekaari H, Kazempour A, Ghasedi-Khajeh Z (2012) Fluid Phase Equilib 316:102–108
Yan ZN, Geng R, Gu BX, Pan Q, Wang JJ (2014) Fluid Phase Equilib 367:125–134
Mazur K, Heisler IA, Meech SR (2010) J Phys Chem B 114:10684–10691
Klotz I, Rosenberg RM (1972) Chemical thermodynamics, basic theory and methods, 3rd edn. Benjamin Cummings, Upper Saddle River
Wadi RK, Goyal RK (1992) J Solution Chem 21:163–170
Anil KN, Renu P, Neetu (2014) J Chem Thermodyn 68:169–182
Millero FJ, Surdo AL, Shin C (1978) J Phys Chem 82:784–792
Yan ZN, Wang XL, Bai XR, Wang SQ, Wang JJ (2012) J Chem Thermodyn 52:89–94
Yan ZN, Wang XG, Zhao Y, Wang JJ (2009) Acta Chim Sinica 67:115–121
Yan ZN, Zhao Y, Xing RH, Wang XG, Wang JJ (2010) J Chem Eng Data 55:759–764
Yan ZN, Wang XG, Xing RH, Wang JJ (2009) J Chem Eng Data 54:1787–1792
Roy MN, De P, Sikdar PS (2013) Fluid Phase Equilib 352:7–13
Roy MN, Ekka D, Dewan R (2011) Acta Chim Slov 58:792–796
Rajagopal K, Gladson SE (2011) J Chem Thermodyn 43:852–867
Anwar A, Vidiksha B, Priyanka B (2013) J Mole Liq 177:209–214
Hepler L (1969) Can J Chem 47:4613–4617
Banipal TS, Kaur D, Banipal PK, Singh G (2008) J Mol Liq 140:54–60
Gurney RW (1953) Ionic processes in solution. McGraw Hill, New York
Friedman H, Krishnan CV (1973) Plenum press, New York, pp. 118
Franks F, Quickenden MA, Reid DS, Watson B (1970) Trans Faraday Soc 66:582–589
Carpena P, Aguiar J, Bernaola-Galvan P, Ruiz CC (2002) Langmuir 18:6054–6058
Aguiar J, Carpena P, Molina-Bolivar JA, Ruiz CC (2003) J Colloid Interface Sci 258:116–122
Wang JJ, Wang HY, Zhang SL, Zhang HC, Zhao Y (2007) J Phys Chem B 111:6181–6188
Inoue T, Ebina H, Dong B (2007) J Colloid Interface Sci 314:236–241
Geng F, Liu J, Zheng LQ, Yu L, Li Z, Li GZ, Tung CH (2010) J Chem Eng Data 55:147–151
Li N, Zhang SH, Zheng LQ, Li XW, Yu L (2008) Phys Chem Chem Phys 10:4375–4377
Philips JNE (1955) Trans Faraday Soc 51:561–569
Luczak J, Jungnickel C, Joskowska M, Thoming J, Hupka J (2009) J Colloid Interface Sci 336:111–116
Turro NJ, Yekta A (1978) J Am Chem Soc 100:5951–5952
Vanyur R, Biczok L, Miskolczy Z (2007) Colloids Surfaces A: Physicochem Eng Aspects 299:256–261
Goodchild I, Collier L, Millar SL, Prokes I, Lord JCD, Butts CP, Bowers J, Webster JRP, Heenan RK (2007) J Colloid Interface Sci 307:455–468
Tanford C (1973) The hydrophobic effect: formation of micelles and biological membranes. Wiley, New York
Das D, Ismail K (2008) J Colloid Interface Sci 327:198–203
Israelachvili JN (1985) Physics of amphiphiles: micelles, vesicles and microemulsions. Degiorgio V. and Corti M. (eds) Amsterdam: North-Holland
Acknowledgment
The project is financially supported by the Natural Science Foundation of China (No. 20973158).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wen, X., Yan, Z., Kang, Y. et al. Apparent molar volume, conductivity, and fluorescence studies of ternary systems of dipeptides + ionic liquids ([Cnmim]Br, n = 10, 14) + water at different temperatures. Colloid Polym Sci 293, 2485–2495 (2015). https://doi.org/10.1007/s00396-015-3644-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3644-2