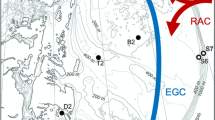
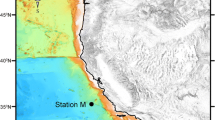
Abstract
In the Southern Ocean, pteropods play an important role in biogeochemical cycling, and sediment traps are a valuable tool for investigating this role through the collection of passively sinking matter from productive surface waters to deep sea layers. Observations of ‘swimmers’ (e.g. organisms that actively swim into traps) can also prove valuable for studying zooplankton community structure. In this study, we used two separate sediment trap studies during the 2016–2017 summer to study pteropod population structure over time scales of 24 h and 28 days. In both studies, highest densities were measured for veliger-stage Limacina helicina antarctica (0.09–0.3 mm) relative to all species and age classes. Increases in shell diameters of veligers in all traps over time enabled the calculation of an intraseasonal potential growth rate of 0.0068 mm d−1. Swimmer flux rates ranged from 121 to 2674 ind. m−2 d−1 at 53 m depth, and the 24-h vertical flux study measured 960 ind. m−2 d−1 at 57 m depth and 6692 m−2 d−1 at 90 m depth. Among a suite of environmental and biological covariates tested, fluorescence and sinking particulate organic and inorganic carbon (POC and PIC) possessed the most predictive power to explain abundances of near-surface pteropod age class and species composition. Gymnosome abundances were largely influenced by increasing adult L. helicina antarctica counts. Changes to pteropod population and community dynamics in response to climate change will have cascading effects throughout Antarctic epipelagic food webs, and these results provide a regional snapshot of patterns in structure and sedimentation from an under-surveyed region of the Southern Ocean.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
The code generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Accornero A, Manno C, Esposito F, Gambi MC (2003) The vertical flux of particulate matter in the polynya of Terra Nova Bay. Part II. Biological components. Antarct Sci 15:175–188. https://doi.org/10.1017/S0954102003001214
Akiha F, Hashida G, Makabe R, Hattori H, Sasaki H (2017) Distribution in the abundance and biomass of shelled pteropods in surface waters of the Indian sector of the Antarctic Ocean in mid-summer. Polar Sci. https://doi.org/10.1016/j.polar.2017.02.003
Almogi-Labin A, Hemleben C, Deuser WG (1988) Seasonal variation in the flux of euthecosomatous pteropods collected in a deep sediment trap in the Sargasso Sea. Deep Sea Res Part A Oceanogr Res Pap 35:441–464. https://doi.org/10.1016/0198-0149(88)90020-9
Arrigo KR, Thomas DN (2004) Large scale importance of sea ice biology in the Southern Ocean. Antarct Sci 16:471–486. https://doi.org/10.1017/S0954102004002263
Ashjian CJ, Campbell RG, Welch HE, Butler M, Van Keuren D (2003) Annual cycle in abundance, distribution, and size in relation to hydrography of important copepod species in the western Arctic Ocean. Deep Res Part I Oceanogr Res Pap 50:1235–1261. https://doi.org/10.1016/S0967-0637(03)00129-8
Augustin NH, Borchers DL, Clarke ED, Buckland ST, Walsh M (2011) Spatiotemporal modelling for the annual egg production method of stock assessment using generalized additive models. Can J Fish Aquat Sci 55:2608–2621. https://doi.org/10.1139/f98-143
Bauerfeind E, Nöthig EM, Pauls B, Kraft A, Beszczynska-Möller A (2014) Variability in pteropod sedimentation and corresponding aragonite flux at the Arctic deep-sea long-term observatory HAUSGARTEN in the eastern Fram Strait from 2000 to 2009. J Mar Syst 132:95–105. https://doi.org/10.1016/j.jmarsys.2013.12.006
Bayne BL (1986) Marine mussels: their ecology and physiology. Cambridge University Press, Cambridge
Bednaršek N, Možina J, Vogt M, O’Brien C, Tarling GA (2012a) The global distribution of pteropods and their contribution to carbonate and carbon biomass in the modern ocean. Earth Syst Sci Data 1:1–20. https://doi.org/10.5194/essd-1-1-2012
Bednaršek N, Tarling GA, Fielding S, Bakker DCE (2012b) Population dynamics and biogeochemical significance of Limacina helicina antarctica in the Scotia Sea (Southern Ocean). Deep Sea Res Part II Top Stud Oceanogr 59–60:105–116. https://doi.org/10.1016/j.dsr2.2011.08.003
Bednaršek N, Harvey CJ, Kaplan IC, Feely RA, Možina J (2016) Pteropods on the edge: Cumulative effects of ocean acidification, warming, and deoxygenation. Prog Oceanogr 145:1–24. https://doi.org/10.1016/j.pocean.2016.04.002
Bednaršek N, Klinger T, Harvey CJ, Weisberg S, McCabe RM, Feely RA, Newton J, Tolimieri N (2017) New ocean, new needs: application of pteropod shell dissolution as a biological indicator for marine resource management. Ecol Indic 76:240–244. https://doi.org/10.1016/j.ecolind.2017.01.025
Bergami C, Capotondi L, Langone L, Giglio F, Ravaioli M (2009) Distribution of living planktonic foraminifera in the Ross Sea and the Pacific sector of the Southern Ocean (Antarctica). Mar Micropaleontol 73:37–48. https://doi.org/10.1016/j.marmicro.2009.06.007
Borchers DL, Buckland ST, Priede IG, Ahmadi S (2011) Improving the precision of the daily egg production method using generalized additive models. Can J Fish Aquat Sci 54:2727–2742. https://doi.org/10.1139/f97-134
Buesseler KO, Antia AN, Chen M, Fowler SW, Gardner WD, Gustafsson O, Harada K, Michaels AF, Rutgers van der Loeff M, Sarin M, Steinberg DK, Trull T (2007) An assessment of the use of sediment traps for estimating upper ocean particle fluxes. J Mar Res 65:345–416. https://doi.org/10.1357/002224007781567621
Busch K, Bauerfeind E, Nöthig EM (2015) Pteropod sedimentation patterns in different water depths observed with moored sediment traps over a 4-year period at the LTER station HAUSGARTEN in eastern Fram Strait. Polar Biol 38:845–859. https://doi.org/10.1007/s00300-015-1644-9
Chiba S, Ishimaru T, Hosie GW, Fukuchi M (2001) Spatio-temporal variability of zooplankton community structure off east Antarctica (90 to 160°E). Mar Ecol Prog Ser 216:95–108. https://doi.org/10.3354/meps216095
Comeau S, Alliouane S, Gattuso JP (2012) Effects of ocean acidification on overwintering juvenile Arctic pteropods Limacina helicina. Mar Ecol Prog Ser 456:279–284. https://doi.org/10.3354/meps09696
Dadon JR, de Cidre LL (1992) The reproductive cycle of the Thecosomatous pteropod Limacina retroversa in the western South Atlantic. Mar Biol 114:439–442
Elliott DT, Tang KW, Shields AR (2009) Mesozooplankton beneath the summer sea ice in McMurdo Sound, Antarctica: Abundance, species composition, and DMSP content. Polar Biol 32:113–122. https://doi.org/10.1007/s00300-008-0511-3
Gannefors C, Böer M, Kattner G, Graeve M, Eiane K, Gulliksen B, Hop H, Falk-Petersen S (2005) The Arctic sea butterfly Limacina helicina: Lipids and life strategy. Mar Biol 147:169–177. https://doi.org/10.1007/s00227-004-1544-y
Gardner WD, Richardson M, Smith Jr WO (2000) Seasonal patterns of water column particulate organic carbon and fuxes in the Ross Sea, Antarctica. Deep Sea Res II 47:3423–3449. https://doi.org/10.1016/S0967-0645(00)00074-6
Gardner J, Manno C, Bakker DCE, Peck VL, Tarling GA (2018) Southern Ocean pteropods at risk from ocean warming and acidification. Mar Biol 165:8. https://doi.org/10.1007/s00227-017-3261-3
Hopcroft RR, Clarke C, Nelson RJ, Raskoff KA (2005) Zooplankton communities of the Arctic’s Canada Basin: the contribution by smaller taxa. Polar Biol 28:198–206. https://doi.org/10.1007/s00300-004-0680-7
Hopkins TL (1987) Midwater food web in McMurdo Sound, Ross Sea, Antarctica. Mar Biol 96:93–106. https://doi.org/10.1007/BF00394842
Hunt BPV, Hosie GW (2006a) The seasonal succession of zooplankton in the Southern Ocean south of Australia, part I: The seasonal ice zone. Deep Res Part I 53:1182–1202. https://doi.org/10.1016/j.dsr.2006.05.001
Hunt BPV, Hosie GW (2006b) The seasonal succession of zooplankton in the Southern Ocean south of Australia, part II: the Sub-Antarctic to Polar Frontal Zones. Deep Res Part I 53:1203–1223. https://doi.org/10.1016/j.dsr.2006.05.002
Hunt BPV, Pakhomov EA, Hosie GW, Siegel V, Ward P, Bernard K (2008) Pteropods in Southern Ocean ecosystems. Prog Oceanogr 78:193–221. https://doi.org/10.1016/j.pocean.2008.06.001
Jia Z, Swadling KM, Meiners KM, Kawaguchi S, Virtue P (2016) The zooplankton food web under East Antarctic pack ice: a stable isotope study. Deep Res Part II Top Stud Oceanogr 131:189–202. https://doi.org/10.1016/j.dsr2.2015.10.010
Kattner G, Hagen W, Graeve M, Albers C (1998) Exceptional lipids and fatty acids in the pteropod Clione limacina (Gastropoda) from both polar oceans. Mar Chem 61:219–228. https://doi.org/10.1016/S0304-4203(98)00013-9
Kleiber C, Zeileis A (2008) Applied econometrics with R. Springer Science and Business Media.
Knauer GA, Karl DM, Martin JH, Hunter CN (1984) In situ effects of selected preservatives on total carbon, nitrogen and metals collected in sediment traps. J Mar Res 42:445–462. https://doi.org/10.1357/002224084788502710
Kobayashi HA (1974) Growth cycle and related vertical distribution of the thecosomatous pteropod Spiratella (“Limacina”) helicina in the central Arctic Ocean. Mar Biol 26:295–301. https://doi.org/10.1007/BF00391513
Kraft A, Bauerfeind E, Nöthig EM, Bathmann UV (2012) Size structure and life cycle patterns of dominant pelagic amphipods collected as swimmers in sediment traps in the eastern Fram Strait. J Mar Syst 95:1–15. https://doi.org/10.1016/j.jmarsys.2011.12.006
Lalli CM, Gilmer RW (1989) Pelagic snails: the biology of holoplanktonic gastropod mollusks. Stanford University Press.
Lalli M, Wells FE (1978) Reproduction in the genus Limacina (Opisthobranchia: Thecosomata). J Zool Lond 186:95–108
Maas AE, Elder LE, Dierssen HM, Seibel BA (2011) Metabolic response of Antarctic pteropods (Mollusca: Gastropoda) to food depriviation and regional productivity. Mar Ecol Prog Ser 441:129–139. https://doi.org/10.3354/meps09358
Macdonald P, Du J (2015) mixdist: finite mixture distribution models. R package, version 0.5–4. (http://CRAN.R-project.org/package=mixdist).
Mackey AP, Atkinson A, Hill SL, Ward P, Cunningham NJ, Johnston NM, Murphy EJ (2012) Antarctic macrozooplankton of the southwest Atlantic sector and Bellingshausen Sea: baseline historical distributions (Discovery Investigations, 1928–1935) related to temperature and food, with projections for subsequent ocean warming. Deep Res Part II Top Stud Oceanogr 59–60:130–146. https://doi.org/10.1016/j.dsr2.2011.08.011
Makabe R, Hattori H, Sampei M, Darnis G, Fortier L, Sasaki H (2016) Can sediment trap-collected zooplankton be used for ecological studies? Polar Biol 39:2335–2346. https://doi.org/10.1007/s00300-016-1900-7
Manno C, Sandrini S, Tositti L, Accornero A (2007) First stages of degradation of Limacina helicina shells observed above the aragonite chemical lysocline in Terra Nova Bay (Antarctica). J Mar Syst 68:91–102. https://doi.org/10.1016/j.jmarsys.2006.11.002
Manno C, Tirelli V, Accornero A, Fonda Umani S (2010) Importance of the contribution of Limacina helicina faecal pellets to the carbon pump in Terra Nova Bay (Antarctica). J Plankton Res 32:145–152. https://doi.org/10.1093/plankt/fbp108
Manno C, Peck VL, Tarling GA (2016) Pteropod eggs released at high pCO2 lack resilience to ocean acidification. Nat Publ Gr. https://doi.org/10.1038/srep25752
Manno C, Bednaršek N, Tarling GA, Peck VL, Comeau S, Adhikari D, Bakker DCE, Bauerfeind E, Bergan AJ, Berning MI, Buitenhuis E, Burridge AK, Chierici M, Flöter S, Fransson A, Gardner J, Howes EL, Keul N, Kimoto K, Kohnert P, Lawson GL, Lischka S, Maas A, Mekkes L, Oakes RL, Pebody C, Peijnenburg KTCA, Seifert M, Skinner J, Thibodeau PS, Wall-Palmer D, Ziveri P (2017) Shelled pteropods in peril: assessing vulnerability in a high CO2 ocean. Earth-Science Rev 169:132–145. https://doi.org/10.1016/j.earscirev.2017.04.005
Manno C, Giglio F, Stowasser G, Fielding S, Enderlein P, Tarling GA (2018) Threatened species drive the strength of the carbonate pump in the northern Scotia Sea. Nat Commun 9:4592. https://doi.org/10.1038/s41467-018-07088-y
Matsuno K, Yamaguchi A, Fujiwara A, Onodera J, Watanabe E, Imai I, Chiba S, Harada N, Kikuchi T (2014) Seasonal changes in mesozooplankton swimmers collected by sediment trap moored at a single station on the Northwind Abyssal Plain in the western Arctic Ocean. J Plankton Res 36:490–502. https://doi.org/10.1093/plankt/fbt092
Michaels AF, Silver MW, Gowing MM, Knauer GA (1990) Cryptic zooplankton “swimmers” in upper ocean sediment traps. Deep Sea Res Part A, Oceanogr Res Pap 37:1285–1296. https://doi.org/10.1016/0198-0149(90)90043-U
Moteki M, Odate T, Hosie GW, Takahashi KT, Swadling KM, Tanimura A (2017) Ecosystem studies in the Indian Ocean sector of the Southern Ocean undertaken by the training vessel Umitaka-maru. Polar Sci 12:1–4. https://doi.org/10.1016/j.polar.2017.04.002
Motoda S (1985) Devices of Simple Plankton Apparatus–VII. Bull Mar Sci 37:776–777
Možina J (2013) Pteropods in the world atlas of marine plankton functional types (Doctoral dissertation, Univerza v Novi Gorici, Fakulteta za znanosti o okolju)
Nishizawa Y, Sasaki H, Takahashi KT (2016) Interannual variability in euthecosomatous pteropods (Limacina spp.) in the Indian sector of the Southern Ocean during austral summer. Antarct Rec 60:35–48
Noji TT, Bathmann UV, Von Bodungen B, Voss M, Antia A, Krumbholz M, Klein B, Peeken I, Noji CIM, Rey F (1997) Clearance of picoplankton-sized particles and formation of rapidly sinking aggregates by the pteropod, Limacina retroversa. J Plankton Res 19:863–875. https://doi.org/10.1093/plankt/19.7.863
Orsi AH, Whitworth T, Nowlin WD (1995) On the meridional extent and fronts of the Antarctic Circumpolar Current. Deep Res Part I 42:641–673. https://doi.org/10.1016/0967-0637(95)00021-W
Paranjape MA (1968) The egg mass and veligers of Limacina helicina Phipps, in: Veliger. pp. 322–326.
Raby D, Lagadeuc Y, Dodson JJ, Mingelbier M (1994) Relationship between feeding and vertical distribution of bivalve larvae in stratified and mixed waters. Mar Ecol Prog Ser 103:275–284. https://doi.org/10.3354/meps103275
Rines JEB, McFarland MN, Donaghay PL, Sullivan JM (2010) Thin layers and species-specific characterization of the phytoplankton community in Monterey Bay, California, USA. Cont Shelf Res 30:66–80. https://doi.org/10.1016/j.csr.2009.11.001
Roberts D, Howard WR, Moy AD, Hopcroft RR (2011) Interannual pteropod variability in sediment traps deployed above and below the aragonite saturation horizon in the Sub-Antarctic Southern Ocean. Polar Biol 3:1739–1750. https://doi.org/10.1007/s00300-011-1024-z
Roberts D, Howard WR, Roberts JL, Bray SG, Moy AD, Trull TW, Hopcroft RR (2014) Diverse trends in shell weight of three Southern Ocean pteropod taxa collected with Polar Frontal Zone sediment traps from 1997 to 2007. Polar Biol 37:1445–1458. https://doi.org/10.1007/s00300-014-1534-6
Ross RM, Quetin LB, Lascara C (1996) Distribution of Antarctic krill and dominant zooplankton west of the Antarctic Peninsula. Found Ecol Res west Antarct Penins 70:199–217. https://doi.org/10.1029/AR070p0199
Ryan KG, Tay ML, Martin A, McMinn A, Davy SK (2011) Chlorophyll fluorescence imaging analysis of the responses of Antarctic bottom-ice algae to light and salinity during melting. J Exp Mar Bio Ecol 399:156–161. https://doi.org/10.1016/j.jembe.2011.01.006
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez J-Y, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Seibel BA, Dierssen HM (2003) Cascading trophic impacts of reduced biomass in the Ross Sea, Antarctica: just the tip of the iceberg? Biol Bull 205:93–97. https://doi.org/10.2307/1543229
Seibel BA, Maas AE, Dierssen HM (2012) Energetic plasticity underlies a variable response to ocean acidification in the pteropod Limacina helicina antarctica. PLoS ONE. https://doi.org/10.1371/journal.pone.0030464
Sokolov S, Rintoul SR (2009) Circumpolar structure and distribution of the antarctic circumpolar current fronts: 2. Variability and relationship to sea surface height. J Geophys Res Ocean 114:1–15. https://doi.org/10.1029/2008JC005248
Teniswood CMH, Roberts D, Howard WR, Bray SG, Bradby JE (2016) Microstructural shell strength of the Subantarctic pteropod Limacina helicina antarctica. Polar Biol 39:1643–1652. https://doi.org/10.1007/s00300-016-1888-z
Thabet AA, Maas AE, Lawson GL, Tarrant AM (2015) Life cycle and early development of the thecosomatous pteropod Limacina retroversa in the Gulf of Maine, including the effect of elevated CO2 levels. Mar Biol 162:2235–2249. https://doi.org/10.1007/s00227-015-2754-1
Thibodeau PS, Steinberg DK, Stammerjohn SE, Hauri C (2019) Environmental controls on pteropod biogeography along the Western Antarctic Peninsula. Limnol Oceanogr 64:S240–S256. https://doi.org/10.1002/lno.11041
Tremblay M, Sinclair M (2007) Sea scallop larvae Placopecten magellanicus on Georges Bank: vertical distribution in relation to water column stratification and food. Mar Ecol Prog Ser 61:1–15. https://doi.org/10.3354/meps061001
van der Spoel S, Dadon JR, Boltovskoy D (1999) South Atlantic Zooplankton, Vol. 1. In: Boltovskoy D (ed) South Atlantic Zooplankton, vol 1. Backhuys Publishers, Leiden, pp 649–706
Venables WN, Ripley BD (2002) Random and Mixed Effects, in: Modern Applied Statistics with S. Springer: New York, pp. 271–300. https://doi.org/10.1007/978-0-387-21706-2_10
Wakabayashi K (2017) Embryonic development of the sea butterfly Desmopterus papilio (Gastropoda: Thecosomata). Invertebr Reprod Dev 61:142–146. https://doi.org/10.1080/07924259.2017.1311952
Wang K, Hunt BPV, Liang C, Pauly D, Pakhomov EA (2017) Reassessment of the life cycle of the pteropod Limacina helicina from a high resolution interannual time series in the temperate North Pacific. ICES J Mar Sci 74:1906–1920. https://doi.org/10.1093/icesjms/fsx014
Willis K, Cottier F, Kwasniewski S, Wold A, Falk-Petersen S (2006) The influence of advection on zooplankton community composition in an Arctic fjord (Kongsfjorden, Svalbard). J Mar Syst 61:39–54. https://doi.org/10.1016/j.jmarsys.2005.11.013
Wormuth JH (1981) Vertical distributions and diel migrations of Euthecosomata in the northwest Sargasso Sea. Deep Sea Res Part A Oceanogr Res Pap 28:1493–1515. https://doi.org/10.1016/0198-0149(81)90094-7
Acknowledgements
We are grateful to all of the reviewers, including Drs. Guglielmo, Maas, and Vecchione, who have offered valuable editorial suggestions that have vastly improved the quality of this work. We would like to acknowledge the master and crew, technical and scientific teams aboard the TRV Umitaka-maru (Tokyo University of Marine Science and Technology (TUMSAT)) and the icebreaker Shirase for assistance with both deployment and retrieval of the sediment traps used for this research. Cruises were funded for “Physical and chemical observations” by the Japanese Antarctic Research Expedition (JARE) under the Ministry of Education, Culture, Sports, Science and Technology (MEXT) through a Grant-in-Aid for Scientific Research (Grant No. 24255001), as well as Research Project Funds of National Institute of Polar Research (Grant No. KP-308). This work was supported by Australian Antarctic Science Grant No. 4331 and the Holsworth Wildlife Research Endowment (Grant No. 109804) through the Ecological Society of Australia and the Equity Trustees Charitable Foundation. CW was supported, in part, by an internship from the National Institute of Polar Research. RT was supported by the RJL Hawke Postdoctoral Fellowship. We dedicate this manuscript to Dr. Tsuneo Odate who championed strong Japan-Australia research collaborations. He will be greatly missed.
Funding
Research cruises were funded for “Physical and chemical observations” by the Japanese Antarctic Research Expedition (JARE) under the Ministry of Education, Culture, Sports, Science and Technology (MEXT) through a Grant-in-Aid for Scientific Research (Grant No. 24255001), as well as Research Project Funds of National Institute of Polar Research (Grant No. KP-308). This work was supported by Australian Antarctic Science Grant No. 4331 and the Holsworth Wildlife Research Endowment (Grant No. 109804) through the Ecological Society of Australia and the Equity Trustees Charitable Foundation. CW was supported, in part, by an internship from the National Institute of Polar Research. RT was supported by the RJL Hawke Postdoctoral Fellowship.
Author information
Authors and Affiliations
Contributions
Conceptualization: CKW; methodology: CKW; formal analysis and investigation: CKW; writing—original draft preparation: CKW; writing—review and editing: CKW, RT, KMS, RM; funding acquisition: CKW, MM, TO, KMS; resources: RM, KM, ST; supervision: RM, MM, TO, RT, KMS.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Weldrick, C.K., Makabe, R., Mizobata, K. et al. The use of swimmers from sediment traps to measure summer community structure of Southern Ocean pteropods. Polar Biol 44, 457–472 (2021). https://doi.org/10.1007/s00300-021-02809-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02809-4