Abstract
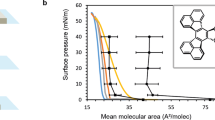
The nanoscale architectures evident in the thin films of self-assembling hybrid block copolymers—which are tailored to inherit the advantageous properties of their constituent synthetic (homo)polymer and polypeptide blocks—have continued to inspire a variety of new applications in different fields, including biomedicine. The thin films of symmetric hybrid block copolymer, α-methoxy-poly(ethylene glycol)-block-poly[ε-(benzyloxycarbonyl)-l-lysine], MPEG112-b-PLL(Z)17, were prepared by solvent casting in five different solvents and characterized using Attenuated Total Reflectance-Fourier Transform Infrared spectroscopy, Thermogravimetric analysis, Derivative Thermogravimetric analysis, Differential Scanning Calorimetry, Contact Angle goniometry, Wide-Angle X-ray Diffraction, and Scanning Electron Microscopy. Film thickness was estimated to be 51 ± 23 μm by the “step-height” method, using a thickness gauge. Although no significant change to the block copolymer’s microstructure was observed, its solvent-cast films displayed divergent physical and thermal properties. The resulting cast films proved more thermally stable than the bulk but indicated greater block miscibility. Additionally, the thin films of MPEG112-b-PLL(Z)17 preserved the microphase separation exhibited by the bulk copolymer albeit with appreciable loss of crystallinity. The surface properties of the polymer–air interface were diverse as were the effects of the casting solvents. Oriented equilibrium morphologies are also evident in some of the as-cast thin films.








Similar content being viewed by others
References
Charrier J-M (1990) Polymeric materials and processing: plastics, elastomers and composites. Oxford University Press, New York
Jacob MV, Easton CD, Woods GS, Berndt CC (2008) Fabrication of a novel organic polymer thin film. Thin Solid Films 516:3884–3887
Lopes WA, Jaeger HM (2001) Hierarchical self-assembly of metal nanostructures on diblock copolymer scaffolds. Nature 414:735–738
Wang J-Y, Park S, Russell TP (2008) Block copolymer thin films. In: Tsui OKC, Russell TP (eds) Polymer thin films. World Scientific Publishing, Singapore, pp 1–25
Bourtoom T (2008) Edible films and coatings: characteristics and properties. Int Food Res J 15:237–248
Pavlukhina S, Sukhishvili S (2011) Polymer assemblies for controlled delivery of bioactive molecules from surfaces. Adv Drug Deliv Rev 63:822–836
Vendra VK, Wu L, Krishnan S (2010) In: Kumar CSSR (ed) Nanomaterials for the life sciences vol. 5: nanostructured thin films and surfaces. Wiley-VCH, Weinheim, pp 1–54
Brannon-Peppas L, Vert M (2000) Polylactic and polyglycolic acids as drug delivery carriers. In: Wise DL (ed) Handbook of pharmaceutical controlled release technology. Marcel Dekker, New York, pp 107–130
Green PF, Limary R (2001) Block copolymer thin films: pattern formation and phase behaviour. Adv Colloid Interface Sci 94:53–81
Elabd YA, Hickner MA (2011) Block copolymers for fuel cells. Macromolecules 44:1–11
Bates FS, Fredrickson GH (1990) Block copolymer thermodynamics: theory and experiment. Annu Rev Phys Chem 41:525–557
Segalman RA (2005) Patterning with block copolymer thin films. Mater Sci Eng R 48:191–226
Higuchi T, Motoyoshi K, Sugimori H, Jinnai H, Yabu H, Shimomura M (2010) Phase transition and phase transformation in block copolymer nanoparticles. Macromol Rapid Commun 31:1773–1778
Hamley IW (2009) Ordering in thin films of block copolymers: fundamentals to potential applications. Prog Polym Sci 34:1161–1210
Ramanathan M, Darling SB (2011) Mesoscale morphologies in polymer thin films. Prog Polym Sci 36:793–812
Billmeyer FW Jr (1984) Textbook of polymer science, 3rd edn. Wiley, New York
Kryszewski M (1972) Formation of supermolecular structures in thin polymer layers. Pure Appl Chem 31:21–47
Siemann U (2005) Solvent cast technology—a versatile tool for thin film production. Prog Colloid Polym Sci 130:1–14
Law PW, Longdon A, Willins GG (2004) Solvent cast cellulose diacetate film. Macromol Symp 208:293–322
Kim G, Libera M (1998) Morphological development in solvent-cast polystyrene–polybutadiene–polystyrene (SBS) triblock copolymer thin films. Macromolecules 31:2569–2577
Strawhecker KE, Kumar SK, Douglas JF, Karim A (2001) The critical role of solvent evaporation on the roughness of spin-cast polymer films. Macromolecules 34:4669–4672
Roberts MJ, Bentley MD, Harris JM (2002) Chemistry for peptide and protein PEGylation. Adv Drug Deliv Rev 54:459–476
Fruijtier-Pölloth C (2005) Safety assessment on polyethylene glycols (PEGs) and their derivatives as used in cosmetic products. Toxicology 214:1–38
Molineux G (2002) Pegylation: engineering improved pharmaceuticals for enhanced therapy. Cancer Treat Rev 28:13–16
Davis FF, Abuchowski A, van Es T, Palczuk NC, Savoca K, Chen RH-L, Pyatak P (1980) Soluble, non-antigenic polyethylene glycol-bound enzymes. In: Goldberg EP, Nakajima A (eds) Biomedical polymers: polymeric materials and pharmaceuticals for biomedical use. Academic Press, New York, pp 441–452
Nakajima A, Kugo K, Hayashi T, Sato H (1980) Formation, structure and properties of tri-block copolymer membranes: biomaterials containing poly-α-amino acids as one component. In: Goldberg EP, Nakajima A (eds) Biomedical polymers: polymeric materials and pharmaceuticals for biomedical use. Academic Press, New York, pp 243–269
Shanmugam G, Polavarapu PL (2008) Concentration- and dehydration-dependent structural transitions in poly-l-lysine. J Mol Struct 890:144–149
Katchalski E, Grossfeld I, Frankel M (1948) Poly-condensation of α-amino derivatives III: poly-lysine. J Am Chem Soc 70:2094–2101
Ottenbrite RM (1989) Controlled release technology. In: Encyclopedia of polymer science and engineering, 2nd edn, suppl vol. Wiley, New York, pp 164–187
Mazia D, Schatten G, Sale W (1975) Adhesion of cells to surfaces coated with polylysine: applications to electron microscopy. J Cell Biol 66:198–200
Nathan A, Kohn J (1994) Amino acid derived polymers. In: Shalaby SW (ed) Biomedical polymers: designed-to-degrade systems. Hanser/Gardner Publications, Cincinnati, pp 117–151
Koiso K, Komai T, Niijima T (1983) Experimental urinary bladder reconstruction using a synthetic poly(α-amino acids) membrane. Artif Organs 7:232–237
Dorn K, Hoerpel G, Ringsdorf H (1985) Polymeric antitumor agents on molecular and cellular levels. In: Gebelein CG, Carraher CE Jr (eds) Bioactive polymer systems: an overview. Plenum Press, New York, pp 531–585
Katayose S, Kataoka K (1997) Water-soluble polyion complex associates of DNA and poly(ethylene glycol)-poly(l-lysine) block copolymer. Bioconjugate Chem 8:702–707
Lussi JW, Michel R, Reviakine I, Falconnet D, Goessl A, Csucs G, Hubbell JA, Textor M (2004) A novel generic platform for chemical patterning of surfaces. Prog Surf Sci 76:55–69
Shiraishi K, Kawano K, Minowa T, Maitani Y, Yokoyama M (2009) Preparation and in vivo imaging of PEG-poly(l-lysine)-based polymeric micelle MRI contrast agents. J Control Release 136:14–20
Jang W-D, Nakagishi Y, Nishiyama N, Kawauchi S, Morimoto Y, Kikuchi M, Kataoka K (2006) Polyion complex micelles for photodynamic therapy: incorporation of dendritic photosensitizer excitable at long wavelength relevant to improved tissue-penetrating property. J Control Release 113:73–79
Sakurai Y, Akaike T, Kataoka K, Okano T (1980) Interfacial phenomena in biomaterials chemistry. In: Goldberg EP, Nakajima A (eds) Biomedical polymers: polymeric materials and pharmaceuticals for biomedical use. Academic Press, New York, pp 335–379
Saravia V, Küpcü S, Nolte M, Huber C, Pum D, Fery A, Sleytr UB, Toca-Herrera JL (2007) Bacterial protein patterning by micro-contact printing of PLL-g-PEG. J Biotechnol 130:247–252
Männistö M, Vanderkerken S, Toncheva V, Elomaa M, Ruponen M, Schacht E, Urtti A (2002) Structure–activity relationships of poly(l-lysines): effects of pegylation and molecular shape on physicochemical and biological properties in gene delivery. J Control Release 83:169–182
Tian H, Tang Z, Zhuang X, Chen X, Jing X (2012) Biodegradable synthetic polymers: preparation, functionalization and biomedical application. Prog Polym Sci 37:237–280
Mui SC, Trapa PE, Huang B, Soo PP, Lozow MI, Wang TC, Cohen RE, Mansour AN, Mukerjee S, Mayes AM, Sadoway DR (2002) Block copolymer-templated nanocomposite electrodes for rechargeable lithium batteries. J Electrochem Soc 149:A1610–A1615
Kugo K, Murashima M, Hayashi T, Nakajima A (1983) Structure and properties of membrane surfaces of A-B-A triblock copolymers consisting of poly(γ-methyl d, l-glutamate) as the A component and polybutadiene as the B component. Polym J 15:267–277
Müller-Buschbaum P, Gutmann JS, Lorenz-Haas C, Mahltig B, Stamm M, Petry W (2001) Early stages of film creation in thin diblock copolymer films. Macromolecules 34:7463–7470
Izunobi JU, Higginbotham CL (2010) Microstructure characterization and thermal analysis of hybrid block copolymer α-methoxy-poly(ethylene glycol)-block-poly[ε-(benzyloxycarbonyl)-l-lysine] for biomedical applications. J Mol Struct 977:153–164
Izunobi JU, Higginbotham CL (2011) Polymer molecular weight analysis by 1H NMR spectroscopy. J Chem Educ 88:1098–1104
Woodward RP (1999) Contact angle measurements using the drop shape method. First Ten Angstroms Inc, Portsmouth
Bellamy LJ (1975) The infrared spectra of complex molecules, vol 1, 3rd edn. Chapman and Hall, London
Izunobi JU, Higginbotham CL (2013) Conformational and thermal analyses of α-methoxy-poly(ethylene glycol)-block-poly[ε-(benzyloxycarbonyl)-l-lysine] hybrid block copolymers. Polym Int 62:1169–1178
Lever T (2007) Optimizing DSC experiments. In: Craig DQM, Reading M (eds) Thermal analysis of pharmaceuticals. CRC Press, Boca Raton, pp 23–51
Dawkins JV (1973) In: Allport DC, Janes WH (eds) Block copolymers. Applied Science, London, pp 363–408
Tang ZG, Black RA, Curran JM, Hunt JA, Rhodes NP, Williams DF (2004) Surface properties and biocompatibility of solvent-cast poly[ε-caprolactone] films. Biomaterials 25:4741–4748
Chan C-M (1994) Polymer surface modification and characterization. Hanser, Munich
Inoue H, Matsumoto A, Matsukawa K, Ueda A, Nagai S (1990) Surface characteristics of polydimethyl siloxane–poly(methyl methacrylate) block copolymers and their PMMA blends. J Appl Polym Sci 41:1815–1829
Loudon GM (1988) Organic Chemistry, 2nd edn. Benjamin/Cummings, Menlo Park, CA, pp 289–291
Acknowledgments
We thank Dr. James Kennedy for SEM, and Dr. Sean Lyons and Dr. Declan Devine for useful discussion. This work was funded, in part, under the Irish National Development Plan (NDP) Technological Sector Research program (Strand III).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Izunobi, J.U., Geever, L.M. & Higginbotham, C.L. Preparation and characterization of poly(ethylene glycol)-block-poly[ε-(benzyloxycarbonyl)-l-lysine] thin films for biomedical applications. Polym. Bull. 71, 1691–1709 (2014). https://doi.org/10.1007/s00289-014-1149-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-014-1149-7