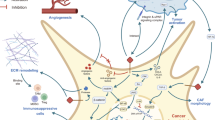
Abstract
Background
The development of tumor tissue-infiltrating regulatory T cell (Treg) is incompletely understood. This study investigates the role of retinoblastoma cell (Rbc)-derived Twist‑related protein 1 (Twist) in the Treg development.
Methods
The surgically removed Rb tissues were collected. Rbcs were cultured with CD4+ T cells to assess the role of Rbc-derived Twist in the Treg generation.
Results
We found that more than 90% Rbcs expressed Twist. Foxp3+ Tregs were detected in the Rb tissues that were positively correlated with the Twist expression in Rbcs, negatively associated with Rb patient survival and sight survival. Treating Rbcs with hypoxia promoted the Twist expression that could be detected in the cytoplasm, nuclei and on the cell surface. Twist activated CD4+ T cells by binding the TLR4/myeloid differentiation factor 2 complex and promoted the transforming growth factor-β-inducible early gene 1 product and Foxp3 expression. These Rbc-induced Foxp3+ Tregs showed immune-suppressive function on CD8+ T cell proliferation.
Conclusions
Rbcs express Twist, that induces IL-4+ Foxp3+ Tregs; the latter can inhibit CD8+ cytotoxic T cell activities. Therefore, Twist may play an important role in the pathogenesis of Rb.







Similar content being viewed by others
References
Dimaras H, Corson TW, Cobrinik D et al (2015) Retinoblastoma. Nat Rev Dis Prim 1:15021. https://doi.org/10.1038/nrdp.2015.21
Le Rhun E, Preusser M, Roth P, Reardon DA, van den Bent M, Wen P, Reifenberger G, Weller M (2019) Molecular targeted therapy of glioblastoma. Cancer Treat Rev 80:101896. https://doi.org/10.1016/j.ctrv.2019.101896
Mendoza PR, Grossniklaus HE (2015) The biology of retinoblastoma. Prog Mol Biol Transl Sci 134:503–516. https://doi.org/10.1016/bs.pmbts.2015.06.012
Xu XL, Singh HP, Wang L, Qi DL, Poulos BK, Abramson DH, Jhanwar SC, Cobrinik D (2014) Rb suppresses human cone-precursor-derived retinoblastoma tumours. Nature 514:385–388. https://doi.org/10.1038/nature13813
Andre P, Denis C, Soulas C et al (2018) Anti-NKG2A mAb is a checkpoint inhibitor that promotes anti-tumor immunity by unleashing both T and NK cells. Cell 175:1731–43.e13. https://doi.org/10.1016/j.cell.2018.10.014
Schietinger A, Greenberg PD (2014) Tolerance and exhaustion: defining mechanisms of T cell dysfunction. Trends Immunol 35:51–60. https://doi.org/10.1016/j.it.2013.10.001
Ali K, Soond DR, Pineiro R et al (2014) Inactivation of PI(3)K p110delta breaks regulatory T-cell-mediated immune tolerance to cancer. Nature 510:407–411. https://doi.org/10.1038/nature13444
Togashi Y, Shitara K, Nishikawa H (2019) Regulatory T cells in cancer immunosuppression—implications for anticancer therapy. Nat Rev Clin Oncol. 16:356–371. https://doi.org/10.1038/s41571-019-0175-7
Puisieux A, Valsesia-Wittmann S, Ansieau S (2006) A twist for survival and cancer progression. Br J Cancer 94:13–17. https://doi.org/10.1038/sj.bjc.6602876
Kaliki S, Patel A, Iram S, Palkonda VAR (2017) Clinical presentation and outcomes of stage III or stage IV retinoblastoma in 80 Asian Indian patients. J Pediatr Ophthalmol Strabismus 54:177–184. https://doi.org/10.3928/01913913-20161019-01
Xu LZ, Xie RD, Xie H et al (2020) Chimeric specific antigen epitope-carrying dendritic cells induce interleukin-17(+) regulatory T cells to suppress food allergy. Clin Exp Allergy 50:231–243. https://doi.org/10.1111/cea.13528
Krishnakumar S, Mohan A, Mallikarjuna K, Venkatesan N, Biswas J, Shanmugam MP, Ren-Heidenreich L (2004) EpCAM expression in retinoblastoma: a novel molecular target for therapy. Invest Ophthalmol Vis Sci 45:4247–4250. https://doi.org/10.1167/iovs.04-0591
Takeuchi Y, Nishikawa H (2016) Roles of regulatory T cells in cancer immunity. Int Immunol 28:401–409. https://doi.org/10.1093/intimm/dxw025
Pezzuto A, Carico E (2018) Role of HIF-1 in cancer progression: novel insights. A review. Curr Mol Med. 18:343–351. https://doi.org/10.2174/1566524018666181109121849
Mami-Chouaib F, Tartour E (2019) Editorial: tissue resident memory T cells. 10. https://doi.org/10.3389/fimmu.2019.01018
Park BS, Song DH, Kim HM, Choi BS, Lee H, Lee JO (2009) The structural basis of lipopolysaccharide recognition by the TLR4-MD-2 complex. Nature 458:1191–1195. https://doi.org/10.1038/nature07830
Peng DJ, Zeng M, Muromoto R, Matsuda T, Shimoda K, Subramaniam M, Spelsberg TC, Wei WZ, Venuprasad K (2011) Noncanonical K27-linked polyubiquitination of TIEG1 regulates Foxp3 expression and tumor growth. J Immunol. 186:5638–5647. https://doi.org/10.4049/jimmunol.1003801
Khanbabaei H, Teimoori A, Mohammadi M (2016) The interplay between microRNAs and Twist1 transcription factor: a systematic review. Tumour Biol 37:7007–7019. https://doi.org/10.1007/s13277-016-4960-y
Lin MC, Lin JJ, Hsu CL, Juan HF, Lou PJ, Huang MC (2017) GATA3 interacts with and stabilizes HIF-1alpha to enhance cancer cell invasiveness. Oncogene 36:4243–4252. https://doi.org/10.1038/onc.2017.8
Chang YC, Chan YC, Chang WM, Lin YF, Yang CJ, Su CY, Huang MS, Wu ATH, Hsiao M (2017) Feedback regulation of ALDOA activates the HIF-1alpha/MMP9 axis to promote lung cancer progression. Cancer Lett 403:28–36. https://doi.org/10.1016/j.canlet.2017.06.001
Vichalkovski A, Gresko E, Hess D, Restuccia DF, Hemmings BA (2010) PKB/AKT phosphorylation of the transcription factor Twist-1 at Ser42 inhibits p53 activity in response to DNA damage. Oncogene 29:3554–3565. https://doi.org/10.1038/onc.2010.115
Kishton RJ, Sukumar M, Restifo NP (2017) Metabolic regulation of T Cell longevity and function in tumor immunotherapy. Cell Metab 26:94–109. https://doi.org/10.1016/j.cmet.2017.06.016
Mohanty R, Chowdhury CR, Arega S, Sen P, Ganguly P, Ganguly N (2019) CAR T cell therapy: a new era for cancer treatment (Review). Oncol Rep 42:2183–2195. https://doi.org/10.3892/or.2019.7335
Romano M, Fanelli G, Albany CJ, Giganti G, Lombardi G (2019) Past, present, and future of regulatory T cell therapy in transplantation and autoimmunity. Front Immunol. 10:43. https://doi.org/10.3389/fimmu.2019.00043
Joseph JP, Harishankar MK, Pillai AA, Devi A (2018) Hypoxia induced EMT: a review on the mechanism of tumor progression and metastasis in OSCC. Oral Oncol 80:23–32. https://doi.org/10.1016/j.oraloncology.2018.03.004
Hajizadeh F, Okoye I, Esmaily M, Ghasemi Chaleshtari M, Masjedi A, Azizi G, Irandoust M, Ghalamfarsa G, Jadidi-Niaragh F (2019) Hypoxia inducible factors in the tumor microenvironment as therapeutic targets of cancer stem cells. Life Sci 237:116952. https://doi.org/10.1016/j.lfs.2019.116952
Ciesielska A, Hromada-Judycka A, Ziemlinska E, Kwiatkowska K (2019) Lysophosphatidic acid up-regulates IL-10 production to inhibit TNF-alpha synthesis in Mvarphis stimulated with LPS. J Leukoc Biol 106:1285–1301. https://doi.org/10.1002/jlb.2a0918-368rr
Nagai Y, Yanagibashi T, Watanabe Y et al (2012) The RP105/MD-1 complex is indispensable for TLR4/MD-2-dependent proliferation and IgM-secreting plasma cell differentiation of marginal zone B cells. Int Immunol 24:389–400. https://doi.org/10.1093/intimm/dxs040
Liu H, Zhang G, Huang J, Ma S, Mi K, Cheng J, Zhu Y, Zha X, Huang W (2016) Atractylenolide I modulates ovarian cancer cell-mediated immunosuppression by blocking MD-2/TLR4 complex-mediated MyD88/NF-kappaB signaling in vitro. J Transl Med. 14:104. https://doi.org/10.1186/s12967-016-0845-5
Shang P, Tang Q, Hu Z, Huang S, Hu Y, Zhu J, Liu H (2020) Procyanidin B3 alleviates intervertebral disc degeneration via interaction with the TLR4/MD-2 complex. J Cell Mol Med 24:3701–3711. https://doi.org/10.1111/jcmm.15074
Horvatinovich JM, Grogan EW, Norris M, Steinkasserer A, Lemos H, Mellor AL, Tcherepanova IY, Nicolette CA, DeBenedette MA (2017) Soluble CD83 inhibits T cell activation by binding to the TLR4/MD-2 complex on CD14(+) monocytes. J Immunol. 198:2286–2301. https://doi.org/10.4049/jimmunol.1600802
Kammoun M, Piquereau J, Nadal-Desbarats L et al (2020) Novel role of Tieg1 in muscle metabolism and mitochondrial oxidative capacities. Acta Physiol (Oxf) 228:e13394. https://doi.org/10.1111/apha.13394
Zhang X, Yao Y, Wei WZ, Yang ZQ, Gu J, Zhou L (2017) Impaired epidermal Langerhans cell maturation in TGFbeta-inducible early gene 1 (TIEG1) knockout mice. Oncotarget. 8:112875–112882. https://doi.org/10.18632/oncotarget.22843
Subramaniam M, Pitel KS, Withers SG, Drissi H, Hawse JR (2016) TIEG1 enhances Osterix expression and mediates its induction by TGFbeta and BMP2 in osteoblasts. Biochem Biophys Res Commun 470:528–533. https://doi.org/10.1016/j.bbrc.2016.01.112
Waddell KM, Kagame K, Ndamira A, Twinamasiko A, Picton SV, Simmons IG, Revill P, Johnston WT, Newton R (2015) Improving survival of retinoblastoma in Uganda. Br J Ophthalmol 99:937–942. https://doi.org/10.1136/bjophthalmol-2014-306206
Acknowledgements
This study was supported by grants of the National Nature and Science Foundation of China (81870706, 31570932, U1801286, 31000713, 31700805), Guangdong Provincial Key Laboratory of Regional Immunity and Diseases (2019B030301009), Cross Innovation Research Initiative Funds of Shenzhen University and Shenzhen science, technology and innovation committee (201002052, KQTD20170331145453160 and KQJSCX20180328095619081).
Author information
Authors and Affiliations
Contributions
RZ, YNS, XD, ZG, YS, ZZ and YL performed experiments, analyzed data and reviewed the manuscript. GN, PCY and BPM organized the study and supervised experiments. PCY designed the project and prepared manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The author declares that there is no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, R., Song, YN., Duo, X. et al. Retinoblastoma cell-derived Twist protein promotes regulatory T cell development. Cancer Immunol Immunother 70, 1037–1048 (2021). https://doi.org/10.1007/s00262-020-02744-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02744-z