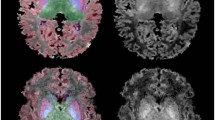
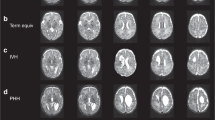
Abstract
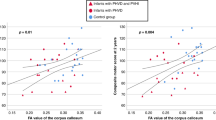
Preterm birth is associated with alteration in corticothalamic development, which underlies poor neurodevelopmental outcomes. Our hypothesis was that preterm neonates with CHD would demonstrate abnormal thalamic microstructure when compared to critically ill neonates without CHD. A secondary aim was to identify any association between thalamic microstructural abnormalities and perioperative clinical variables. We compared thalamic DTI measurements in 21 preterm neonates with CHD to two cohorts of neonates without CHD: 28 term and 27 preterm neonates, identified from the same neonatal intensive care unit. Comparison was made with three other selected white matter regions using ROI manual-based measurements. Correlation was made with post-conceptional age and perioperative clinical variables. In preterm neonates with CHD, there were age-related differences in thalamic diffusivity (axial and radial) compared to the preterm and term non-CHD group, in contrast to no differences in anisotropy. Contrary to our hypothesis, abnormal thalamic and optic radiation microstructure was most strongly associated with an elevated first arterial blood gas pO2 and elevated preoperative arterial blood gas pH (p < 0.05). Age-related thalamic microstructural abnormalities were observed in preterm neonates with CHD. Perinatal hyperoxemia and increased perioperative serum pH were associated with abnormal thalamic microstructure in preterm neonates with CHD. This study emphasizes the vulnerability of thalamocortical development in the preterm neonate with CHD.


Similar content being viewed by others
Abbreviations
- CHD:
-
Congenital heart disease
- NICU:
-
Neonatal intensive care unit
- ASD:
-
Atrial septal defect
- VSD:
-
Ventricular septal defect
- HLHS:
-
Hypoplastic left heart syndrome
- TGA:
-
Transposition of the great arteries
- DORV:
-
Double-outlet right ventricle
- FA:
-
Fractional anisotropy
- RD:
-
Radial diffusivity
- AD:
-
Axial diffusivity
References
Andropoulos DB, Hunter JV, Nelson DP, Stayer SA, Stark AR, McKenzie ED, Heinle JS, Graves DE, Fraser CD Jr (2010) Brain immaturity is associated with brain injury before and after neonatal cardiac surgery with high-flow bypass and cerebral oxygenation monitoring. J Thorac Cardiovasc Surg 139(3):543–556
Ball G, Counsell SJ, Anjari M, Merchant N, Arichi T, Doria V, Rutherford MA, Edwards AD, Rueckert D, Boardman JP (2010) An optimised tract-based spatial statistics protocol for neonates: applications to prematurity and chronic lung disease. Neuroimage 53:94–102
Ball G, Boardman JP, Rueckert D, Aljabar P, Arichi T, Merchant N, Gousias IS, Edwards AD, Counsell SJ (2012) The effect of preterm birth on thalamic and cortical development. Cereb Cortex 22:1016–1024
Ballweg JA, Wernovsky G, Gaynor JW (2007) Neurodevelopmental outcomes following congenital heart surgery. Pediatr Cardiol 28:126–133
Bartha AI, Yap KR, Miller SP, Jeremy RJ, Nishimoto M, Vigneron DB, Barkovich AJ, Ferriero DM (2007) The normal neonatal brain: MR imaging, diffusion tensor imaging, and 3D MR spectroscopy in healthy term neonates. AJNR 28:1015–1021
Bellinger DC, Bernstein JH, Kirkwood MW (2003) Visual-spatial skills in children after open-heart surgery. J Dev Behav Pediatr 24:169–179
Bellinger DC, Wypij D, Rivkin MJ et al (2011) Adolescents with d-transposition of the great arteries corrected with the arterial switch procedure: neuropsychological assessment and structural brain imaging. Circulation 124:1361–1369
Chau V, Synnes A, Grunau RE, Poskitt KJ, Brant R, Miller SP (2013) Abnormal brain maturation in preterm neonates associated with adverse developmental outcomes. Neurology 81:2082–2089
Cheng HH, Almodovar MC, Laussen PC et al (2011) Outcomes and risk factors for mortality in premature neonates with critical congenital heart disease. Pediatr Cardiol 32:1139–1146
Clouchoux C, du Plessis AJ, Bouyssi-Kobar M et al (2012) Delayed cortical development in fetuses with complex congenital heart disease. Cereb Cortex. doi:10.1093/cerecor/bhs281
Costello JM, Polito A, Brown DW et al (2010) Birth before 39 weeks’ gestation is associated with worse outcomes in neonates with heart disease. Pediatrics 126:277–284
Counsell SJ, Dyet LE, Larkman DJ (2007) Thalamo-cortical connectivity in children born preterm mapped using probabilistic magnetic resonance tractography. Neuroimage 34:896–904
Dees E, Lin H, Cotton RB, Graham TP, Dodd DA (2000) Outcome of preterm infants with congenital heart disease. J Pediatr 137:653–659
Felderhoff-Mueser U, Bittigau P, Sifringer M, Jarosz B, Korobowicz E, Mahler L, Piening T, Moysich A, Grune T, Thor F, Heumann R, Buhrer C, Ikonomidou C (2004) Oxygen causes cell death in the developing brain. Neurobiol Dis 13:273–282
Gelehrter S, Fifer CG, Armstrong A, Hirsch J, Gajarski R (2011) Outcomes of hypoplastic left heart syndrome in low-birth-weight patients. Pediatr Cardiol 32:1175–1181
Goff DA, Luan X, Gerdes M et al (2012) Younger gestational age is associated with worse neurodevelopmental outcomes after cardiac surgery. J Thorac Cardiovasc Sur 143:535–542
Haynes RL, Folkerth RD, Keefe RJ, Sung I, Swzeda LI, Rosenberg PA, Volpe JJ, Kinney HC (2013) Nitrosative and oxidative injury to premyelinating oligodendrocytes in periventricular leukomalacia. J Neuropathol Exp Neurol 62:441–450
Kaindl AM, Favrais G, Gressens P (2009) Molecular mechanism involved in injury to the preterm brain. J Child Neurol 24(9):1112–1118
Kanold PO, Luhmann HJ (2010) The subplate and early cortical circuits. Annu Rev Neurosci 33:23–48
Karsdorp PA, Everaerd W, Kindt M, Mulder BJM (2007) Physiological and cognitive functioning in children and adolescents with congenial heart disease. Pediatr Psychol 32:527–541
Kostovic I, Judas M (2007) Patterns of cortical lamination during prenatal life: Do they have implications for treatment? Neurosci Biobehav Rev 31:1157–1168
Kostovic I, Judas M (2010) The development of the subplate and thalamocortical connections in the human foetal brain. Acta Paediatr 99:1119–1127
Licht DJ, Shera DM, Clancy RR (2009) Brain maturation is delayed in infants with complex congenital heart defects. J Thorac Cardiovasc Surg 137:529–536
Mahle WT, Clancy RR, Moss EM, Gerdes M, Jobes DR, Wernovsky G (2000) Neurodevelopmental outcome and lifestyle assessment in school-aged and adolescent children with hypoplastic left heart syndrome. Pediatrics 105:1082–1089
Markus T, Hansson S, Amer-Wahlin I, Hellstrom-Westas L, Saugstad OD, Ley D (2007) Cerebral inflammatory response after fetal asphyxia and hyperoxic resuscitation in newborn sheep. Pediatr Res 62(1):71–77
McQuillen PS, Ferriero DM (2005) Perinatal subplate neuron injury: implications for cortical development and plasticity. Brain Pathol 15:250–260
Miller SP, Ferriero DM, Leonard C et al (2005) Early brain Injury in premature newborns detected with magnetic resonance imaging is associated with adverse neurodevelopmental outcomes. J Pediatr 147:609–616
Miller SP, McQuillen PS, Hamrick S et al (2007) Abnormal brain development in newborns with congenital heart disease. New Engl J Med 357:1928–1938
Natarajan G, Anne SR, Aggarwal S (2011) Outcomes of congenital heart disease in late preterm infants: double jeopardy? Acta Paediatr 100:1104–1107
Newburger JW, Sleeper LA, Bellinger DC et al (2012) Early developmental outcome in children with hypoplastic left heart syndrome and related anomalies: the single ventricle reconstruction trial. Circulation 125:2081–2091
Paquette LB, Wisnowski JL, Pruetz JD et al (2013) Abnormal cerebral microstructure in premature neonates with congenital heart disease. AJNR 34:2026–2033
Shillingford AJ, Glanzman MM, Ittenbach RF, Clancy RR, Gaynor JW, Wernovsky G (2008) Inattention, hyperactivity, and school performance in a population of school-age children with complex congenital heart disease. Pediatrics 121:e759–e767
Tanner K, Sabrine N, Wren C (2005) Cardiovascular malformations among preterm infants. Pediatrics 116:e833–e838
Watanabe K, Matsui M, Matsuzawa J et al (2009) Impaired neuroanatomic development in infants with congenital heart disease. J Thorac Cardiov Sur 137:146–153
Acknowledgments
The authors thank Julie Castro, Lisa Padilla, and Hesham Mahmoud for their assistance with patient recruitment and data management. We thank Melanie Gieraltowski for manuscript preparation. Support for this study is provided by the National Institutes of Health: K23NS063371 to AP, P50NA019632 to JLW, 1UL1RR031986 to AP, JLW, LP; and Rudi Schulte Foundation to SB, CHLA Clinical Investigation Center, The Children’s Heart Foundation, supported in part by the NIH NCRR GCRC GRANT 5 MO1 RR00043-44 AT Childrens Hospital Los Angeles.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paquette, L.B., Votava-Smith, J.K., Ceschin, R. et al. Abnormal Development of Thalamic Microstructure in Premature Neonates with Congenital Heart Disease. Pediatr Cardiol 36, 960–969 (2015). https://doi.org/10.1007/s00246-015-1106-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-015-1106-8