Abstract
Purpose
Abdominal recurrences of gastrointestinal malignancies are common. Evidence in clinical studies has shown that re-irradiation (Re-I) is tolerable and efficient in different tumor locations. In contrast, little clinical data are available on normal long-term Re‑I tolerance doses. A systematic review of upper abdominal Re‑I was performed with the aim of exploring the cumulative dose, toxicity, and outcomes.
Methods
A computerized search was undertaken in MEDLINE, EMBASE, OVID, and the Cochrane database. Only studies reporting toxicity and/or outcomes were taken into consideration. To improve the comparability of the different Re‑I regimens and assess the relationship between Radiotherapy (RT) dose and toxicity, the equivalent dose in 2‑Gy fractions was calculated according to the linear quadratic model.
Results
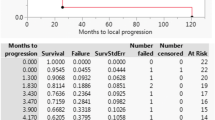
Sixteen studies met the inclusion criteria, with the total patients numbering 408. Median follow-up Re‑I ranged from 5.9 to 45 months. The median time elapsed since previous RT treatment was 15 months (2–162 months). Re‑I prescription doses were variable (22.5 Gy in 3 fractions to 126.5 Gy with 125I). Cumulative doses calculated for acute- and late-responding tissues ranged from 67.25 to 136 Gy and 30.3 to 188.38 Gy, respectively. Comprehensively, the pooled ≥G3 toxicity was 12% (95%CI: 7.6–19%). The overall 1‑year survival and local recurrence-free survival rates were 53.7% (95%CI: 45.6–63.2%) and 66.5% (95% CI: 58.7–75.4%), respectively. Pain improvement was reported in 66.9% of patients.
Conclusion
Due to limited evidence as a result of the retrospective design of the majority of the studies, our review suggests that upper abdominal Re‑I is effective in terms of local control and palliation, with a moderate rate of severe toxicities.
Zusammenfassung
Zweck
Bei gastrointestinalen Malignomen sind abdominale Rezidive häufig. Klinische Studien haben belegt, dass eine Re-I-Bestrahlung (Re-I) bei verschiedenen Tumorlokalisationen tolerierbar und wirksam ist. Dagegen liegen nur wenige klinische Daten über langfristige normale Re-I-Toleranzdosen vor. Es wurde eine systematische Überprüfung der Oberbauch-Re‑I durchgeführt, mit dem Ziel, kumulative Dosis, Toxizität und Ergebnisse zu untersuchen.
Methoden
Eine computergestützte Suche wurde anhand von MEDLINE, EMBASE, OVID und der Cochrane-Datenbank durchgeführt. Es wurden nur Studien mit Angabe der Toxizität und/oder Gefahrlosigkeit als Ergebnis berücksichtigt. Um die Vergleichbarkeit der verschiedenen Re-I-Regime zu verbessern und den Zusammenhang zwischen Strahlentherapie(RT)-Dosis und Toxizität zu beurteilen, wurde die Äquivalentdosis in 2‑Gy-Fraktionen nach dem linear-quadratischen Modell berechnet.
Ergebnisse
Sechzehn Studien erfüllten die Einschlusskriterien bei einer Gesamtzahl von 408 Patienten. Das mediane Follow-up-Re-I-Range betrug 5,9–45 Monate. Die seit der vorhergehenden RT verstrichene mediane Zeit betrug 15 Monate (Spanne 2–162 Monate). Die Re-I-Verschreibungsdosen waren variabel (22,5 Gy in 3 Fraktionen bis 126,5 Gy mit 125I). Die für akutes und spät ansprechendes Gewebe berechneten kumulativen Dosen lagen zwischen 67,25 und 136 Gy bzw. 30,3 und 188,38 Gy. Insgesamt betrug die gepoolte >G3-Toxizität 12% (95%-Konfidenzintervall [KI] 7,6–19%). Die auf 1 Jahr bezogene Gesamtüberlebensrate und die lokale rezidivfreie Überlebensrate betrugen jeweils 53,7% (95%-KI 45,6–63,2%) bzw. 66,5% (95%-KI 58,7–75,4%). Bei 66,9% der Patienten wurde eine Verbesserung der Schmerzsituation verzeichnet.
Schlussfolgerung
Trotz der Einschränkungen aufgrund weniger Beweise und aufgrund der vorwiegend retrospektiv gestalteten Studien spricht unsere Überprüfung dafür, dass Oberbauch-Re‑I durchführbar ist, was schwere Toxizität bei guter lokaler Kontrolle und Linderung der Symptome anbelangt.





Similar content being viewed by others
References
Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Azevedo E, Silva G, Chen WQ, Eser S, Engholm G, Stiller CA, Monnereau A, Woods RR, Visser O, Lim GH, Aitken J, Weir HK, Coleman MP, CONCORD Working Group (2018) Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 391:1023–1075. https://doi.org/10.1016/S0140-6736(17)33326-3
Hishinuma S, Ogata Y, Tomikawa M, Ozawa I, Hirabayashi K, Igarashi S (2006) Patterns of recurrence after curative resection of pancreatic cancer, based on autopsy findings. J Gastrointest Surg 10:511–518. https://doi.org/10.1016/j.gassur.2005.09.016
Baumann M, Krause M, Overgaard J, Debus J, Bentzen SM, Daartz J, Richter C, Zips D, Bortfeld T (2016) Radiation oncology in the era of precision medicine. Nat Rev Cancer 16:234–249. https://doi.org/10.1038/nrc.2016.18
Høyer M (2017) Re-irradiation with stereotactic body radiation therapy (SBRT). Chin Clin Oncol 6(Suppl 2):S15. https://doi.org/10.21037/cco.2017.07.01
Fernandes A, Berman AT, Mick R, Both S, Lelionis K, Lukens JN, Ben-Josef E, Metz JM, Plastaras JP (2016) A prospective study of proton beam reirradiation for esophageal cancer. Int J Radiat Oncol Biol Phys 95:483–487. https://doi.org/10.1016/j.ijrobp.2015.12.005
Boimel PJ, Berman AT, Li J, Apisarnthanarax S, Both S, Lelionis K, Larson GL, Teitelbaum U, Lukens JN, Ben-Josef E, Metz JM, Plastaras JP (2017) Proton beam reirradiation for locally recurrent pancreatic adenocarcinoma. J Gastrointest Oncol 8:665–674. https://doi.org/10.21037/jgo.2017.03.04
Merlotti A, Mazzola R, Alterio D, Alongi F, Bacigalupo A, Bonomo P, Maddalo M, Russi EG, Orlandi E (2017) What is the role of postoperative re-irradiation in recurrent and second primary squamous cell cancer of head and neck? A literature review according to PICO criteria. Crit Rev Oncol Hematol 111:20–30. https://doi.org/10.1016/j.critrevonc.2017.01.008
De Bari B, Filippi AR, Mazzola R, Bonomo P, Trovò M, Livi L, Alongi F (2015) Available evidence on re-irradiation with stereotactic ablative radiotherapy following high-dose previous thoracic radiotherapy for lung malignancies. Cancer Treat Rev 41:511–518. https://doi.org/10.1016/j.ctrv.2015.04.002
Abusaris H, Hoogeman M, Nuyttens JJ (2012) Re-irradiation: outcome, cumulative dose and toxicity in patients retreated with stereotactic radiotherapy in the abdominal or pelvic region. Technol Cancer Res Treat 11:591–597. https://doi.org/10.7785/tcrt.2012.500261
Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement—Journal of Clinical Epidemiology. (2018) https://www.jclinepi.com/article/S0895-4356(09)00179-6/fulltext.
Institute of Health Economics (2018) About methodology development. https://www.ihe.ca/research-programs/rmd/cssqac/cssqac-about
Julious SA (2005) Two-sided confidence intervals for the single proportion: comparison of seven methods by Robert G. Newcombe. Stat Med 24:3383–3384. https://doi.org/10.1002/sim.2164
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Harms W, Krempien R, Grehn C, Berns C, Hensley FW, Debus J (2005) Daytime pulsed dose rate brachytherapy as a new treatment option for previously irradiated patients with recurrent oesophageal cancer. Br J Radiol 78:236–241. https://doi.org/10.1259/bjr/51982166
Haque W, Crane CH, Krishnan S, Delclos ME, Javle M, Garrett CR, Wolff RA, Das P (2009) Reirradiation to the abdomen for gastrointestinal malignancies. Radiat Oncol 4:55. https://doi.org/10.1186/1748-717X-4-55
Yamaguchi S, Ohguri T, Imada H, Yahara K, Moon SD, Higure A, Yamaguchi K, Yoshikawa I, Harada M, Korogi Y (2011) Multimodal approaches including three-dimensional conformal re-irradiation for recurrent or persistent esophageal cancer: preliminary results. J Radiat Res 52(22020080):812–820
Lominska CE, Unger K, Nasr NM, Haddad N, Gagnon G (2012) Stereotactic body radiation therapy for reirradiation of localized adenocarcinoma of the pancreas. Radiat Oncol 7:74. https://doi.org/10.1186/1748-717X-7-74
Wild AT, Hiniker SM, Chang DT, Tran PT, Khashab MA, Limaye MR, Laheru DA, Le DT, Kumar R, Pai JS, Hargens B, Sharabi AB, Shin EJ, Zheng L, Pawlik TM, Wolfgang CL, Koong AC, Herman JM (2013) Re-irradiation with stereotactic body radiation therapy as a novel treatment option for isolated local recurrence of pancreatic cancer after multimodality therapy: experience from two institutions. J Gastrointest Oncol 4:343–351. https://doi.org/10.3978/j.issn.2078-6891.2013.044
Yao L, Jiang Y, Jiang P, Wang H, Meng N, Qu A, Tian S, Sun H, Liu C, Wang J, Zhang K (2015) CT-guided permanent 125I seed interstitial brachytherapy for recurrent retroperitoneal lymph node metastases after external beam radiotherapy. Brachytherapy 14:662–669. https://doi.org/10.1016/j.brachy.2015.05.008
Seol SW, Yu JI, Park HC, Lim DH, Oh D, Noh JM, Cho WK, Paik SW (2015) Treatment outcome of hepatic re-irradiation in patients with hepatocellular carcinoma. Radiat Oncol J 33:276–283. https://doi.org/10.3857/roj.2015.33.4.276
Dagoglu N, Callery M, Moser J, Tseng J, Kent T, Bullock A, Miksad R, Mancias JD, Mahadevan A (2016) Stereotactic body radiotherapy (SBRT) reirradiation for recurrent pancreas cancer. J Cancer 7(13295):283–288. https://doi.org/10.7150/jca
Huang Y, Chen SW, Fan CC, Ting LL, Kuo CC, Chiou JF (2016) Clinical parameters for predicting radiation-induced liver disease after intrahepatic reirradiation for hepatocellular carcinoma. Radiat Oncol 11:89. https://doi.org/10.1186/s13014-016-0663-1
Koong AJ, Toesca DAS, von Eyben R, Pollom EL, Chang DT (2017) Reirradiation with stereotactic body radiation therapy after prior conventional fractionation radiation for locally recurrent pancreatic adenocarcinoma. Adv Radiat Oncol 2:27–36. https://doi.org/10.1016/j.adro.2017.01.003
Hunt A, Das P, Minsky BD, Koay EJ, Krishnan S, Herman JM, Taniguchi C, Koong A, Smith GL, Holliday EB (2018) Hyperfractionated abdominal reirradiation for gastrointestinal malignancies. Radiat Oncol 13:143. https://doi.org/10.1186/s13014-018-1084-0
Gkika E, Strouthos I, Kirste S, Adebahr S, Schultheiss M, Bettinger D, Fritsch R, Brass V, Maruschke L, Neeff HP, Lang SA, Nestle U, Grosu AL, Brunner TB (2019) Repeated SBRT for in- and out-of-field recurrences in the liver. Strahlenther Onkol 195:246–253. https://doi.org/10.1007/s00066-018-1385-0
McDuff SGR, Remillard KA, Zheng H, Raldow AC, Wo JY, Eyler CE, Drapek LC, Goyal L, Blaszkowsky LS, Clark JW, Allen JN, Parikh AR, Ryan DP, Ferrone CR, Tanabe KK, Wolfgang JA, Zhu AX, Hong TS (2018) Liver reirradiation for patients with hepatocellular carcinoma and liver metastasis. Pract Radiat Oncol 8:414–421. https://doi.org/10.1016/j.prro.2018.04.012
Stewart FA, Oussoren Y, van Tinteren H, Bentzen SM (1994) Loss of reirradiation tolerance in the kidney with increasing time after single or fractionated partial tolerance doses. Int J Radiat Biol 66:169–179
Yu SK, Bhangu A, Tait DM, Tekkis P, Wotherspoon A, Brown G (2014) Chemoradiotherapy response in recurrent rectal cancer. Cancer Med 3:111–117. https://doi.org/10.1002/cam4.169
Ben-Josef E, Schipper M, Francis IR, Hadley S, Ten-Haken R, Lawrence T, Normolle D, Simeone DM, Sonnenday C, Abrams R, Leslie W, Khan G, Zalupski MM (2012) A phase I/II trial of intensity modulated radiation (IMRT) dose escalation with concurrent fixed-dose rate gemcitabine (FDR-G) in patients with unresectable pancreatic cancer. Int J Radiat Oncol Biol Phys 84:1166–1171. https://doi.org/10.1016/j.ijrobp.2012.02.051
Massaccesi M, Longo S, Caravatta L, Maddalo M, Pontoriero A, Dionisi F, Bonome P, Indrieri P, Laudati A, Micheletti S, Scorsetti M, Campoccia S, Alterio D, D’Angelo E, Buffoli A, Arcangeli S, Marzano S, Munoz F, Zinicola T, Nardangeli A, Chiesa S, Donato V, Valentini V (2018) Ontology of head and neck cancer reirradiation: fundamentals for a shared multidimensional network database within the RE.VOL.V.E.R. (RE-treatment VOLumes Value for prediction of Effects of Re-Irradiation) project framework. AIRO Abstract book.; CO059, pp 79–80
Acknowledgements
The authors wish to thank the Scientific Commission of the Italian Association of Radiotherapy and Clinical Oncology (AIRO) for the revision of the manuscript.
Author information
Authors and Affiliations
Consortia
Contributions
L. Caravatta and M. Massaccesi designed and coordinated the study and the analysis. L. Boldrini, A. Nardangeli, and A.R. Alitto performed the literature search and selected included studies. F. Dionisi revised the literature search and performed data extraction. C. Rosa, F. Munoz, and L. Bianco checked data extraction. F. Fiorica and L. Caravatta finally reviewed and approved the literature selection and data extraction. F. Fiorica performed statistical data analysis. F. Fiorica and L. Caravatta performed the main data analysis and provided graphics. F. Fiorica, L. Caravatta, A.R. Alitto, and C. Rosa drafted the manuscript. M. Massaccesi, M. Lupattelli, D. Genovesi, and G. Mantello critically revised the study and the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
L. Caravatta, F. Fiorica, C. Rosa, L. Boldrini, A.R. Alitto, A. Nardangeli, F. Dionisi, L. Bianco, F. Munoz, M. Lupattelli, G. Mantello, D. Genovesi, and M. Massaccesi declare that there is no conflict of interest regarding the publication of this article.
Rights and permissions
About this article
Cite this article
Caravatta, L., Fiorica, F., Rosa, C. et al. Role of upper abdominal reirradiation for gastrointestinal malignancies: a systematic review of cumulative dose, toxicity, and outcomes on behalf of the Re-Irradiation Working Group of the Italian Association of Radiotherapy and Clinical Oncology (AIRO). Strahlenther Onkol 196, 1–14 (2020). https://doi.org/10.1007/s00066-019-01519-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-019-01519-5