Summary
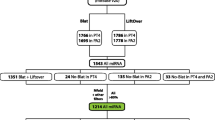
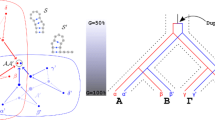
The existing classification of human Alu sequences is revised and expanded using a novel methodology and a larger set of sequence data. Our study confirms that there are two major Alu subfamilies, Alu-J and Alu-S. The Alu-S subfamily consists of at least five distinct subfamilies referred to as Alu-Sx, Alu-Sq, Alu-Sp, Alu-Sc, and Alu-Sb. The Alu-Sp and Alu-Sq subfamilies have been revealed by this study. Alu subfamilies differ from one another in a number of positions called diagnostic. In this paper the diagnostic positions are defined in quantitative terms and are used to evaluate statistical significance of the observed subfamilies. Each Alu subfamily most likely represents pseudogenes retroposed from evolving functional source Alu genes. Evidence presented in this paper indicates that Alu-Sp and Alu-Sc pseudogenes were retroposed from different source genes, during overlapping periods of time, and at different rates. Our analysis also indicates that the previously identified Alu-type transcript BC200 comes from an active Alu gene that might have existed even before the origin of dimeric Alu sequences. The source genes for Alu pseudogene families are reconstructed. It is assumed that diagnostic differences between reconstructed source genes reflect mutations that have occurred in true source Alu genes under natural selection. Some of these mutations are compensatory and are used to reconstruct a common secondary structure of Alu RNAs transcribed from the source genes. The biological function of Alu RNA is discussed in the context of its homology to the elongation-arresting domain of 7SL RNA. Practical implications of our analyses for studies of the human and of other primate genomes are outlined. A computer program that identifies diagnostic bases in individual Alu repeats is designed and made available on-line.
Similar content being viewed by others
References
Britten RJ, Baron WF, Stout D, Davidson EH (1988) Sources and evolution of human Alu repeated sequences. Proc Natl Acad Sci USA 85: 4770–4774
Britten RJ, Stout D, Davidson EH (1989) The current source of human Alu retroposons is a conserved gene shared with Old World monkey. Proc Natl Acad Sci USA 86: 3718–3722
Deininger PL, Daniels GR (1986) The recent evolution of mammalian repetitive DNA elements. Trends Genet 2:76–80
Deininger PL, Slagel VK (1988) Recently amplified Alu family members share a common parental Alu sequence. Mol Cell Biol 8: 4566–4569
Ellis NA, Goodfellow PJ, Pym B, Smith M, Palmer M, Frischauf A-M, Goodfellow PN (1989) The pseudoautosomal boundary in man is defined by an Alu repeat sequence inserted on the Y chromosome. Nature 337: 81–84
Faulkner DV, Jurka J (1988) Multiple aligned sequence editor (MASE). Trends Biochem Sci 13: 321–322
Fox GE, Woese CR (1975) 5S RNA secondary structure. Nature 256: 505–507
Gundelfinger ED, Di Carlo M, Zopf D, Melli M (1984) Structure and evolution of the 7SL RNA component of the signal recognition particle. EMBO J 3: 2325–2332
Jang KL, Latchman DS (1989) HSV infection induces increased transcription of Alu repeated sequences by RNA polymerase III. FEBS Lett 258: 255–258
Jurka J (1989) Subfamily structure and evolution of the human L1 family of repetitive sequences. J Mol Evol 29: 496–503
Jurka J (1990) Novel families of interspersed repetitive elements from the human genome. Nucleic Acids Res 18: 137–141
Jurka J, Smith T (1988) A fundamental division in the Alu family of repeated sequences. Proc Natl Acad Sci USA 85: 4775–4778
Kazazian HH Jr, Wong C, Youssoufian H, Scott AF, Philips DG, Atonarakis SE (1988) Haemophilia A resulting from de novo insertion of L1 sequences represents a novel mechanism for mutation in man. Nature 332: 164–166
Labuda D, Striker G (1989) Sequence conservation in Alu evolution. Nucleic Acids Res 17: 2477–2491
Lehrman MA, Schneider WJ, Sudhof TC, Brown MS, Goldstein JL, Russell DW (1985) Mutation in LDL receptor: Alu-Alu tecombination deletes exons encoding transmembrane and cytoplasmic domains. Science 227: 140–146
Lehrman MA, Goldstein J, Russell DW, Brown MS (1987a) Duplication of seven exons in LDL receptor genes caused by Alu-Alu recombination in a subject with familial hypercholesterolemia. Cell 48: 827–835
Lehrman MA, Russell DW, Goldstein JL, Brown MS (1987b) Alu-Alu recombination deletes splice acceptor sites and produces secreted low density lipoprotein receptor in a subject with familial hypercholesterolemia. J Biol Chem 262: 3354–3361
Levitt M (1969) Detailed molecular model for transfer ribonucleic acid. Nature 224: 759–763
Markert ML, Hutton JJ, Wiginton DA, States JC, Kaufman RE (1988) Adenosine deaminase (ADA) deficiency due to deletion of the ADA gene promoter and first exon by homologous recombination between two Alu elements. J Clin Invest 81: 1323–1327
Mayr E (1961) Cause and effect in biology. Science 134: 1501–1506
Milosavljevic A (1990) Categorization of macromolecular sequences by minimal length encoding. PhD thesis, Available as: Technical Report UCSC-CRL-90-41 from the Computer and Information Sciences Dept., University of California at Santa Cruz
Milosavljevic A, Haussler D, Jurka J (1989) Informed parsimonious inference of prototypical genetic sequences. In: Rivest R, Haussler D, Warmuth MK (eds) Proceedings of the Second Annual Workshop on Computational Learning Theory. Morgan Kaufman, San Mateo, pp 102–117
Pleij CWA (1990) Pseudoknots: a new motif in the RNA game. Trends Biochem Sci 15: 143–147
Quentin Y (1988) The Alu family developed through successive waves of fixation closely connected with primate lineage history. J Mol Evol 27: 194–202
Rouyer F, Simmler MC, Page DC, Weissenbach J (1987) A sex chromosome rearrangement in a human XX male caused by Alu-Alu recombination. Cell 51: 417–425
Ryan SC, Dugaiczyk A (1989) Newly arisen DNA repeats in primate phylogeny. Proc Natl Acad Sci USA 86: 9360–9364
Schoeniger LO, Jelinek WR (1986) 4.5 S RNA is encoded by hundreds of tandemly linked genes, has a short half-life, and is hydrogen bonded in vivo to poly(A)-terminated RNAs in the cytoplasm of cultured mouse cells. Mol Cell Biol 6: 1508–1519
Selker EU (1990) DNA methylation and chromatin structure: a view from below. Trends Biochem Sci 15: 103–107
Siegel V, Walter P (1986) Removal of the Alu structural domain from signal recognition particle leaves its protein translocation activity intact. Nature 320: 81–84
Skowronski J, Fanning TG, Singer MF (1988) Unit-length line-1 transcripts in human teratocarcinoma cells. Mol Cell Biol 8: 1385–1397
Slagel V, Flemington E, Traina-Dorge V, Bradshaw H, Deininger P (1987) Clustering and subfamily relationships of the Alu family in the human genome. Mol Biol Evol 4: 19–29
Smith TF, Waterman MS (1981) Identification of common molecular subsequences. J Mol Biol 145: 195–197
Stoppa-Lyonnet D, Carter PE, Meo T, Tosi M (1990) Clusters of intragenic Alu repeats predispose the human C1 inhibitor locus to deleterious rearrangements. Proc Natl Acad Sci USA 87: 1551–1555
Tomatsu S, Kobayashi Y, Fukumaki Y, Yubisui T, Orii T, Sakaki Y (1989) The organization and complete nucleotide sequence of the human NADH-cytochrome B5 reductase gene. Gene 80: 353–361.
Ullu E, Tschudi C (1984) Alu sequences are processed 7SL RNA genes. Nature 312: 171–172
Wagner M (1986) A consideration of the origin of processed pseudogenes. Trends Genet 2: 134–137
Walter P, Gilmore R, Bobel G (1984) Protein translocation across the endoplasmic reticulum. Cell 38: 5–8
Watson JB, Sutcliffe JG (1987) Primate brain-specific cytoplasmic transcript of the Alu repeat family. Mol Cell Biol 7: 3324–3327
Wilbur WJ, Lipman DJ (1983) Rapid similarity searches of nucleic acid and protein data bases. Proc Natl Acad Sci USA 80: 726–730
Willard C, Nguyen HT, Schmid CW (1987) Existence of at least three distinct Alu subfamilies. J Mol Evol 26: 180–186
Zuckerkandl E, Latter G, Jurka J (1989) Maintenance of function without selection: Alu sequences as “cheap genes”. J Mol Evol 29: 504–512
Zwieb C (1985) The secondary structure of the 7SL RNA in the signal recognition particle: functional implications. Nucleic Acids Res 13: 6105–6124
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jurka, J., Milosavljevic, A. Reconstruction and analysis of human alu genes. J Mol Evol 32, 105–121 (1991). https://doi.org/10.1007/BF02515383
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02515383