Abstract
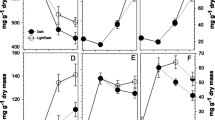
Labelling experiments in which high-specific-activity [U-14C]sucrose or [U-14C]hexoses were injected into potato (Solanum tuberosum L. cv. Desiree) tubers showed that within 1 d of detaching growing tubers from their mother plant, there is an inhibition of starch synthesis, a stimulation of the synthesis of other major cell components, and rapid resynthesis of sucrose. This is accompanied by a general increase in phosphorylated intermediates, an increase in UDP-glucose, and a dramatic decrease of ADP-glucose. No significant decline in the extracted activity of enzymes for sucrose degradation or synthesis, or starch synthesis is seen within 1 d, nor is there a significant decrease in sucrose, amino acids, or fresh weight. Over the next 7 d, soluble carbohydrates decline. This is accompanied by a decline in sucrose-synthase activity, hexose-phosphate levels, and the synthesis of structural cell components. It is argued that a previously unknown mechanism acting at ADP-glucose pyrophosphorylase allows sucrose-starch interconversions to be regulated independently of the use of sucrose for cell growth.
Similar content being viewed by others
Abbreviations
- AGPase:
-
ADP-glucose pyrophosphorylase; Glc=glucose
- Glc6P (Glc1P):
-
glucose-6-phosphate(−1-phosphate)
- Fru6P:
-
fructose-6-phosphate
- Fru 2,6bisP:
-
fructose-2,6-bisphosphate
- PEP:
-
phosphoenolpyruvate
- 3PGA:
-
3-phosphoglycerate
- SPS:
-
sucrose-phosphate synthase
- SuSy:
-
sucrose synthase
- UG-Pase:
-
UDP-glucose pyrophosphorylase
- UDPGLe:
-
urindine-5′-diphosphoglucose
References
Borchert, S., Grosse, H., Heldt, H.W. (1989) Specific transport of inorganic phosphate, glucose-6-phosphate, dihydroxyacetone phosphate and 3-phosphoglycerate into amyloplasts from pea roots. FEBS Let.253, 183–186
Burton, W.G. (1989) The potato. Longman Scientific and Technical, Harlow, UK
Edwards, J.M., green, J.H., ap Rees, T. (1988) Activity of branching enzyme as a cardinal feature of the r-locus inPisum sativum, Phytochemistry25, 2033–2039
Geigenberger, P, Stitt, M. (1993) Sucrose synthase catalyses a readily reversible reaction in vivo in developing potato tubers and other plant tissues. Planta189, 329–339
Hajirezaei, M.R., Stitt, M. (1991) Contrasting roles for pyrophosphate: fructose-6-phosphate phosphotransferase during aging of tissues from potato tubers and carrot storage tissues. Plant Sci77, 177–183
Hatzfeld, W.D., Stitt, M. (1990) A study of the rate of recycling of triose phosphates in heterotrophicChenopodium rubrum cells, potato tubers and maize endosperm. Planta180, 198–204
Heldt, H.W., Chon, C.J., Maronde, D., Herold, A., Stankovic, Z.S., Walker, D.A., Kraminer, A., Kirk, M.R., Heber, U (1977) Role of orthophosphate and other factors in the regulation of starch formation in leaves and isolated chloroplasts. Plant Physiol.59, 1146–1155
Herold, A. (1980) Regulation of photosynthesis by sink activity: the missing link. New Phytol.86, 131–144
Hill, L.M., Smith, A.M. (1992) Evidence that glucose-6-phosphate is imported as substrate for starch synthesis by plastids of developing pea embryos. Planta185, 91–96
Hnilo, J., Okita, T. (1989) Mannose feeding and its effect on starch synthsis in developing potato tuber discs. Plant Cell Physiol.30, 1007–1010
Huber, S.C., Huber, J.L.A. (1992) Role of sucrose-phosphate synthase in sucrose metabolism in leaves. Plant Physiol.99, 1275–1278
Jellito, T., Sonnewald, U., Willmitzer, L., Hajirezaei, M.R., Stitt, M. (1992) Inorganic pyrophosphate content and metabolites in leaves and tubers of potato and tobacco plants expressingE. coli pyrophosphatase in their cytosol. Planta188, 238–244
Keeling, P.I., Bacon, P.J., Holt, D.C. (1993) Elevated temperature reduces starch deposition in wheat endosperm by reducing the activity of soluble starch synthase. Planta191, 342–348
Kleczkowski, L.A., Villand, P., Lüthi, E., Olsen, O.D., Preiss, J. (1993) Insensitivity of barley endosperm ADP-glucose pyrophosphorylase to 3-phosphoglycerate and orthophosphate regulation. Plant Physiol.101, 179–186
Merlo, L., Geigenberger, P., Hajirezaei, M.R., Stitt, M. (1993) Changes of carbohydrates, metabolites and enzyme activities in potato tubers during development, and within a single tuber along a stolon-apex gradient. J. Plant Physiol.142, 392–402
Morrell, S., ap Rees, T. (1986) Sucrose metabolism in developing tubers ofSolanum tuberosum. Phytochemistry25, 1579–18585
Müller-Röber, B.T., Sonnewald, U., Willmitzer, L. (1992) Inhibition of ADP-glucose pyrophosphorylase leads to sugar storing tubers and influences tuber formation and expression of tuber storage protein genes. EMBO J.2, 1229–1238
Neuhaus, H.E., Stitt, M. (1990) Control analysis of photosynthate partitioning. Impact of reduced activity of ADP-glucose pyrophosphorylase or plastid phosphoglucomutase on the fluxes to starch and sucrose inArabidopsis thaliana L. Heynh., Planta182, 445–454
Neuhaus, H.E., Kruckeberg, A.L., Feil, R., Stitt, M. (1989) Decreased-activity mutants of phosphoglucose isomerase in the cytosol and chloroplast ofClarkia xanthiana. Planta178, 110–122
Oparka, K.J., Wright, K.M. (1988) Influence of cell turgor on sucrose partitioning in potato tuber storage tissues. Planta175, 520–526
Oparka, K.J., Davies, H.V., Wright, K.M., Viola, R., Prior, D.A.M. (1990) Effect of sink isolation on sugar uptake and starch synthesis by potato-tuber storage parenchyma. Planta182, 113–117
Plaxton, W.C., Preiss, J. (1987) Purification and properties of nonproteolytically degraded ADP-glucose pyrophosphorylase from maize endosperm. Plant Physiol.85, 105–112
Preiss, J (1988) Biosynthesis of starch and its regulation. In: The biochemistry of plants, carbohydrates, vol. 14 pp. 181–254, Preiss, J., ed. Academic Press, San Diego
Preiss, J. (1991) Biology and molecular biology of starch synthesis and its regulation. Oxford Surv. Plant Mol. Biol.7, 59–114
Preiss, J. (1993) Biosynthesis of Starch: ADPglucose pyrophosphorylase, the regulatory enzyme of starch synthesis: structure-function relationships. Denpun Kayaku40, 117–131
Pressey, R. (1969) Potato sucrose synthetase: purification, properties and changes in activity associated with maturation. Plant Physiol.44, 759–764
Reimholz, R., Geigenberger, P., Stitt, M. (1994) Sucrose-phosphate synthase is regulated via metabolites and protein phosphorylation in potato tubers, in a manner analogous to the enzyme in leaves. Planta192, 480–488
Renz, A., Merlo, L., Stitt, M. (1993) Partial purification of three fructokinases and three hexokinases from potato tubers which show differing organ and developmental specifity. Planta190, 156–165
Ross, H.A., Davies, H.V. (1992) Sucrose metabolism in tubers of potato (Solanum tuberosum L.). Plant Physiol.98, 287–293
Smith, A.M., Denyer, K. (1992) Starch synthesis in developing pea embryos. New Phytol.122, 21–33
Smith, A.M., Martin, C. (1994) Starch biosynthesis and the potential for its manipulation. In: Plant biotechnology, vol. 3, Biosynthesis and manipulation of plant products, Grierson, D., ed. Blackie, Glasgow, in press
Smith-White, B.J., Preiss, J. (1992) Comparison of proteins of ADP-glucose pyrophosphorylase from diverse sources. J. Mol. Evol34, 449–464
Sonnewald, U. (1992) Expression ofE. coli inorganic pyrophosphatase in transgenic plants alters photoassimilate partitioning in leaves. Plant J.2, 571–581
Sowokinos, J.R., Preiss, J. (1982) Phosphorylases inSolanum tuberosum. III Purification, physical and catalytical properties of ADP-glucose pyrophosphorylase in potatoes. Plant Physiol.69, 1459–1466
Stitt, M. (1990) Fructose-2,6-bisphosphate as a regulatory molecule in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol.41, 153–158
Stitt, M. (1991) Rising CO2 levels and their potential significance for carbon flow in photosynthetic cells. Plant Cell Environ.14, 741–762
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Geigenberger, P., Merlo, L., Reimholz, R. et al. When growing potato tubers are detached from their mother plant there is a rapid inhibition of starch synthesis, involving inhibition of ADP-glucose pyrophosphorylase. Planta 193, 486–493 (1994). https://doi.org/10.1007/BF02411552
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02411552