Summary
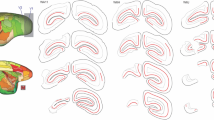
Retrograde axonal transport of horseradish peroxidase (HRP) was used to identify two populations of cells in the lateral geniculate nucleus (LGN) of the cat. HRP was injected into area 17 and 18 separately in the same animal, and the neuronal somata giving rise to thalamo-cortical axons, identified by the presence of granular HRP reaction product within them, were measured. The mean size of LGN neurones labelled by injections in area 17 (“17-relay” cells) was less than of neurones filled from area 18 (“18-relay” cells). Similar separate injections into area 17 and 18 of monocularly deprived kittens also showed that in non-deprived LGN laminae 17-relay cells were, on average, smaller than 18-relay cells. In deprived laminae, 17-relay cells were some 20% smaller than in nondeprived laminae, but deprived 18-relay cells were 50–60% smaller than normal, being on average, actually smaller than deprived 17-relay cells. We conclude that the population of large LGN neurones projecting to area 18 is more severely affected by monocular deprivation than the smaller neurones projecting to area 17, and discuss the relationship of the morphological results to physiologically defined X and Y cells in the LGN.
Similar content being viewed by others
References
Bilge, M., Bingle, A., Seneviratne, K.N., Whitteridge, D.: A map of the visual cortex in the cat. J. Physiol. (Lond.) 191, 116–118P (1967)
Bishop, P.O., Kozak, W., Levick, W.R., Vakkur, G.J.: The determination of the projection of the visual field on to the lateral geniculate nucleus in the cat. J. Physiol. (Lond.) 163, 503–539 (1962)
Blakemore, C., Van Sluyters, R.C.: Innate and environmental factors in the development of the kitten's visual cortex J. Physiol. (Lond.) 248, 663–716 (1975)
Boycott, B.B., Wässle, H.: The morphological types of ganglion cells of the domestic cat's retina. J. Physiol. (Lond.) 240, 397–419 (1974)
Bunt, A.H., Hendrickson, A.E., Lund, J.S., Lund, R.D., Fuchs, A.F.: Monkey retinal ganglion cells: morphometric analysis and tracing of axonal projections, with a consideration of the peroxidase technique. J. comp. Neurol. 164, 265–286 (1975)
Chow, K.L., Stewart, D.L.: Reversal of structural and functional effects of long-term visual deprivation in cats. Exp. Neurol. 34, 409–433 (1972)
Cleland, B.G., Dubin, M.W., Levick, W.R.: Sustained and transient neurones in the cat's retina and lateral geniculate nucleus. J. Physiol. (Lond.) 217, 473–496 (1971)
Cleland, B.G., Levick, W.R., Morstyn, R., Wagner, H.G.: Lateral geniculate relay of slowly conducting retinal afferents to cat visual cortex. J. Physiol. (Lond.) 255, 299–320 (1976)
Dürsteier, M.R., Blakemore, C, Garey, L.J.: Uptake of horseradish peroxidase: analysis by computer reconstruction. In preparation (1977)
Enroth-Cugell, C., Robson, J.G.: The contrast sensitivity of retinal ganglion cells of the cat. J. Physiol. (Lond.) 187, 517–552 (1966)
Fukada, Y.: Receptive field organization of cat optic nerve fibers with special reference to conduction velocity. Vision Res. 11, 209–226 (1971)
Fukuda, Y.: Differentiation of principal cells of the rat lateral geniculate body into two groups: fast and slow cells. Exp. Brain Res. 17, 242–260 (1973)
Fukuda, Y., Stone, J.: Retinal distribution and central projections of Y-, X- and W-cells of the cat's retina. J. Neurophysiol. 37, 749–772 (1974)
Garey, L.J., Blakemore, C.: Monocular deprivation: morphological effects on different classes of neurons in the lateral geniculate nucleus. Science 195, 414–416 (1977)
Garey, L.J., Fisken, R.A., Powell, T.P.S.: Effects of experimental deafferentation on cells in the lateral geniculate nucleus of the cat. Brain Res. 52, 363–369 (1973)
Garey, L.J., Powell, T.P.S.: The projection of the lateral geniculate nucleus upon the cortex in the cat. Proc. roy. Soc. B 169, 107–126 (1967)
Garey, L.J., Powell, T.P.S.: The projection of the retina in the cat. J. Anat. (Lond.) 102, 189–222 (1968)
Garey, L.J., Powell, T.P.S.: An experimental study of the termination of the lateral geniculocortical pathway in the cat and monkey. Proc. roy. Soc. B 179, 41–63 (1971)
Gilbert, C.D., Kelly, J.P.: The projections of cells in different layers of the cat's visual cortex. J. comp. Neurol. 163, 81–106 (1975)
Glickstein, M., King, R.A., Miller, J., Berkley, M.: Cortical projections from the dorsal lateral geniculate nucleus of cats. J. comp. Neurol. 130, 55–76 (1967)
Graybiel, A.: Some extrageniculate visual pathways in the cat. Invest. Ophthal. 11, 322–332 (1972)
Guillery, R.W.: A study of Golgi preparations from the dorsal lateral geniculate nucleus of the adult cat. J. comp. Neurol. 128, 21–50 (1966)
Guillery, R.W.: Survival of large cells in the dorsal lateral geniculate laminae after interruption of retinogeniculate afferents. Brain Res. 28, 541–544 (1971)
Guillery, R.W., Stelzner, D.J.: The differential effects of unilateral lid closure upon the monocular and binocular segments of the dorsal lateral geniculate nucleus in the cat. J. comp. Neurol. 139, 413–422 (1970)
Hayhow, W.R.: The cytoarchitecture of the lateral geniculate body in the cat in relation to the distribution of crossed and uncrossed optic fibers. J. comp. Neurol. 110, 1–64 (1958)
Hoffmann, K.P.: Conduction velocity in pathways from retina to superior colliculus in the cat; a correlation with receptive-field properties. J. Neurophysiol. 36, 409–424 (1973)
Hoffmann, K.P., Stone, J.: Conduction velocity of afferents to cat visual cortex: a correlation with cortical receptive field properties. Brain Res. 32, 460–466 (1971)
Hoffmann, K.P., Stone, J., Sherman, S.M.: Relay of receptive-field properties in dorsal lateral geniculate nucleus of the cat. J. Neurophysiol. 35, 518–531 (1972)
Hubel, D.H., Wiesel, T.N.: Receptive fields and functional architecture in two non-striate visual areas (18 and 19) of the cat. J. Neurophysiol. 28, 229–289 (1965)
Hubel, D.H., Wiesel, T.N.: The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J. Physiol. (Lond.) 206, 419–436 (1970)
Ikeda, H., Wright, M.J.: Receptive field organization of “sustained” and “transient” retinal ganglion cells which subserve different functional roles. J. Physiol. (Lond.) 227, 769–800 (1972)
Kupfer, C., Palmer, P.: Lateral geniculate nucleus: histological and cytochemical changes following afferent denervation and visual deprivation. Exp. Neurol. 9, 400–409 (1964)
Laemle, L.K.: Cell populations of the lateral geniculate nucleus of the cat as determined with horseradish peroxidase. Brain Res. 100, 650–656 (1975)
LaVail, J.H., LaVail, M.M.: Retrograde axonal transport in the central nervous system. Science 176, 1416–1417 (1972)
Maciewicz, R.J.: Thalamic afferents to areas 17, 18 and 19 of cat cortex traced with horseradish peroxidase. Brain Res. 84, 308–312 (1975)
Marrocco, R.T.: Sustained and transient cells in monkey lateral geniculate nucleus: conduction velocities and response properties. J. Neurophysiol. 39, 340–353 (1976)
Movshon, J. A., Dürsteier, M. R.: The effects of brief periods of unilateral eye closure on the kitten's visual system. J. Neurophysiol. (1977) In Press
Nauta, H.J.W., Fritz, M.B., Lasek, R.J.: Afferents to the rat caudoputamen studied with horseradish peroxidase. An evaluation of a retrograde neuroanatomical research method. Brain Res. 67, 219–238 (1974)
Otsuka, R., Hassler, R.: Über Aufbau und Gliederung der corticalen Sehsphäre bei der Katze. Arch. Psychiat. Neurol. 203, 212–234 (1962)
Palmer, L.A., Rosenquist, A.C.: Visual receptive fields of single striate cortical units projecting to the superior colliculus in the cat. Brain Res. 67, 27–42 (1974)
Powell, T.P.S., Mountcastle, V.B.: Some aspects of the functional organization of the cortex of the postcentral gyrus of the monkey: a correlation of the findings obtained in a single unit analysis with cytoarchitecture. Johns Hopk. Hosp. Bull. 105, 133–162 (1959)
Rose, J.E., Woolsey, C.N.: The relations of thalamic connections, cellular structure and evocable electrical activity in the auditory region of the cat. J. comp. Neurol. 91, 441–466 (1949)
Rosenquist, A.C., Edwards, S.B., Palmer, L.A.: An autoradiographic study of the projections of the dorsal lateral geniculate nucleus and the posterior nucleus in the cat. Brain Res. 80, 71–93 (1974)
Sanides, F., Hoffmann, J.: Cyto- and myeloarchitecture of the visual cortex of the cat and of surrounding integration cortices. J. Hirnforsch. 11, 79–104 (1969)
Sherman, S.M., Guillery, R.W., Kaas, J.H., Sanderson, K.J.: Behavioral, electrophysiological and morphological studies of binocular competition in the development of the geniculo-cortical pathways of cats. J. comp. Neurol. 158, 1–18 (1974)
Sherman, S.M., Hoffmann, K.P., Stone, J.: Loss of a specific cell type from dorsal lateral geniculate nucleus in visually deprived cats. J. Neurophysiol. 35, 532–541 (1972)
Sherman, S.M., Norton, T.T., Casagrande, V.A.: X- and Y-cells in the dorsal lateral geniculate nucleus of the tree shrew (Tupaia glis). Brain Res. 93, 152–157 (1975)
Sherman, S.M., Wilson, J.R., Kaas, J.H., Webbe, S.V.: X- and Y-cells in the dorsal lateral geniculate nucleus of the owl monkey (Aotus trivirgatus). Science 192, 475–477 (1976)
Singer, W., Tretter F., Cynader, M.: Organization of cat striate cortex: a correlation of receptive-field properties with afferent and efferent connections. J. Neurophysiol. 38, 1080–1098 (1975)
Sprague, J.M., Meikle, T.H.: The role of the superior colliculus in visually guided behavior. Exp. Neurol. 11, 115–146 (1965)
Stone, J., Dreher, B.: Projection of X- and Y-cells of the cat's lateral geniculate nucleus to areas 17 and 18 of visual cortex. J. Neurophysiol. 36, 551–567 (1973)
Stone, J., Fukuda, Y.: Properties of cat retinal ganglion cells: a comparison of W-cells with X- and Y-cells. J. Neurophysiol. 37, 722–748 (1974)
Stone, J., Hoffmann, K.P.: Very slow-conducting ganglion cells in the cat's retina: a major, new functional type? Brain Res. 43, 610–616 (1972)
Thorpe, P.A., Blakemore, C.: Evidence for a loss of afferent axons in the visual cortex of monocularly deprived cats. Neurosci. Letters 1, 271–276 (1975)
Tretter, F., Cynader, M., Singer, W.: Cat parastriate cortex: a primary or secondary visual area? J. Neurophysiol. 38, 1099–1113 (1975)
Wiesel, T.N., Hubel, D.H.: Effects of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J. Neurophysiol. 26, 978–993 (1963a)
Wiesel, T.N., Hubel, D.H.: Single-cell responses in striate cortex of kittens deprived of vision in one eye. J. Neurophysiol. 26, 1003–1017 (1963b)
Wilson, M.E., Cragg, B.G.: Projections from the lateral geniculate nucleus in the cat and monkey. J. Anat. (Lond.) 101, 677–692 (1967)
Wilson, P.D., Stone, J.: Evidence of W-cell input to the cat's visual cortex via the C laminae of the lateral geniculate nucleus. Brain Res. 92, 472–478 (1975)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garey, L.J., Blakemore, C. The effects of monocular deprivation on different neuronal classes in the lateral geniculate nucleus of the cat. Exp Brain Res 28, 259–278 (1977). https://doi.org/10.1007/BF00235708
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00235708