Summary
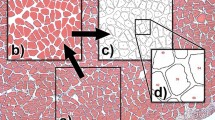
Subpopulations of fast and slow fibres within the trunk musculature of elvers were examined using morphometric analysis of electron micrographs. Fibre regions were characterised by their histochemical staining characteristics, and individual fibres located using a coordinate mapping system utilising morphological features as reference points. Percentages of fibre volume occupied by mitochondria, myofibrils, sarcoplasmic reticulum (S.R.), and T-system were determined in each of the fibre groups, along a transect from the skin to the vertebral column (fibres 1–14, respectively).
The fine structure of slow (“red”) fibres (1–2 fibres deep) is relatively homogeneous throughout its range, giving mean values for mitochondria, 21.4%; myofibrils, 61.0%; S.R., 2.10%; T-system, 0.31%. The fibres are relatively small (204 μm2) and the mitochondrial cristae poorly developed.
In contrast, there is a marked heterogeneity in the ultrastructure of fast (“white”) fibres, dependent on both position and size. The moderately small (333 μm2) superficial fast fibres (3–4 fibres deep) have a significantly higher mitochondrial content (7.6%) than the larger deep fibres (1.2%) (6–12 fibres deep, 775 μm2). The mean fractional volumes occupied by myofibrils, S.R., and T-system in the deep fibres are: 80.4%, 5.95%, and 0.38%, respectively. Fibres < 100 μm2 constitute up to 5% of the fast muscle and have a significantly higher mitochondrial volume (4.3%), more glycogen granules, and a slightly lower volume of S.R. (5.57%) than larger fibres.
It is suggested that metabolic subpopulations of fast fibres correspond to different stages of fibre growth. The relatively poorly developed S.R. of eel fast muscle is thought to be correlated with the low frequency, high amplitude nature of the propagated waveform found in anguilliform locomotion.
Similar content being viewed by others
References
Alexander RMcN (1969) The orientation of muscle fibres in the myomeres of fishes. J Mar Biol Assoc. UK 49:263–290
Altringham JD, Johnston IA (1981) Quantitative histochemical studies of the peripheral innervation of cod (Gadus morhua) fast myotomal muscle fibres. J Comp Physiol 143:123–127
Bertin L (1956) Eels. A biological study. Clever-Hulme Press, London
Bone Q (1964) Patterns of muscular innervation in the lower chordates. Int Rev Neurobiol 6:99–147
Bone Q (1978) Locomotor muscle. In: Hoar WS, Randall DJ (ds) Fish physiology 7. Academic Press, New York, pp 361–424
Bone Q, Chubb AD (1978) The histochemical demonstration of myofibrillar ATPase in elasmobranch muscle. Histochem Jnl 10:489–494
Bone Q, Kiceniuk J, Jones DR (1978) On the role of the different fibre types in fish myotomes at intermediate swimming speeds. Fishery Bulletin 76(2):691–699
Egginton S, Johnston IA (1982) Muscle fibre differentiation and vascularisation in the juvenile European eel (Anguilla anguilla, L.). Cell Tissue Res 222:563–577
Eisenberg BR, Kuda AM (1975) Stereological analysis of mammalian skeletal muscle. Ultrastruct Res 51:176–187
Flitney FW, Johnston IA (1979) Mechanical properties of isolated fish red and white muscle fibres. J Physiol 295:49–50P
Freadman MA (1979) Role partitioning of swimming musculature of striped Bass, Morone saxatilis, Walbaum and Bluefish, Pomatomus saltatrix, L. J Fish Biol 15(4):417–424
Greene CW (1913) An undescribed longitudinal differentiation of the great lateral muscle of the King salmon. Anat Rec 7:99–101
Grillner S, Kashin S (1976) On the generation and performance of swimming in fish. Adv Behavioral Biol 18:181–201
Hudson RCL (1969) Polyneuronal innervation of the fast muscles of the marine teleost Cottus scorpius, L. J Exp Biol 50:47–67
Jankowsky H-D (1966) The effect of adaptation temperature on the metabolic level of the eel Anguilla vulgaris, L. Helgoländer wiss Meeresunters 13:402–407
Johnston IA (1981) Structure and function of fish muscles. In: Day MH (ed) Vertebrate locomotion. Symp Zool Soc Lond 48:71░13
Johnston IA (1982) Quantitative analyses of ultrastructure and vascularisation of the slow muscle fibres of the European Anchovy (Engraulis encrasicolus). Tiss Cell (in press)
Johnston IA, Ward PS, Goldspink G (1975) Studies on the swimming musculature of the rainbow trout. 1. Fibre types. J Fish Biol 7:451–458
Johnston IA, Davison W, Goldspink G (1977) Energy metabolism of carp swimming muscles. J Comp Physiol 114:203–216
Johnston IA, Lucking M (1978) Temperature induced variation in the distribution of different types of muscle fibre in the goldfish (Carassius auratus). J Comp Physiol 124:111–116
Johnston IA, Moon TW (1980) Endurance exercise training in the fast and slow muscles of a teleostfish (Pollachius virens). J Comp Physiol 135B:147–156
Johnston IA, Moon TW (1981) Fine structure and metabolism of multiply innervated fast muscle fibres in teleost fish. Cell Tissue Res 219:93–109
Kilarski W, Kozlowska W (1979) Myofibrillogenesis of the lateral musculature in the trout (Salmo trutta L.). Dev Growth and Differ 21(4):349–360
Kryvi H (1977) Ultrastructure of the different fibre types in axial muscles of the sharks Etmopterus spinax and Galeus melastomus. Cell Tissue Res 184:287–300
Kryvi H, Totland GK (1977) Histochemical studies with microphotometric determinations of the lateral muscles in the sharks Etomopterus spinax and Galeus melastomus. J Mar Biol Assoc, UK 57:261–271
Kryvi H, Flood P, Guljaeu D (1980) The ultrastructure of the fibre types in the axial muscle of the sturgeon Acipenser stellatus. Cell Tissue Res (in press)
Lindsey CC (1978) Form, function, and locomotory habits in fish. In: Hoar WS, Randall DJ (eds) Fish physiology 7. Academic Press, New York, pp 1–100
McArdle HJ, Johnston IA (1981) Ca2+-uptake by tissue sections and biochemical characteristics of sarcoplasmic reticulum isolated from fish fast and slow muscles. Eur J Cell Biol 25:103–107
Nag AC (1972) Ultrastructure and adenosine triphosphatase activity of red and white muscle fibres of the caudal region of a fish, Salmo gairdneri. J Cell Biol 55:42–57
Nag AC, Nursall JR (1972) Histogenesis of white and red muscle fibres of trunk muscles of a fish Salmo gairdneri. Cytobios 6:227–246
Patterson S, Goldspink G (1976) Mechanism of myofibril growth and proliferation in fish muscle. J Cell Sci 22:607–616
Pette D, Wimmer M, Nemeth P (1980) Do enzyme activities vary along muscle fibres? Histochemistry 67:225–231
Raamsdonk W, van Pool CW, Cronnie G (1978) Differentiation of muscle fibres types in the teleost Brachydanio rerio. Anat Embryol 153:137–155
Rasmussen SW, Bulow A, Bachholm P (1975) An improved method of making formvar film for electron microscopy. Science Tools 22(3):40
Robinson G (1977) In: Bancroft JD, Stevens A (eds) Theory and practice of histological techniques. Churchill, Edinburgh, pp 350–352
Wardle CS (1975) Limit of fish swimming speed. Nature (Lond) 255:725–727
Waterman RE (1969) Development of the lateral musculature in the teleost Brachdanio rerio: a fine structural study. Am J Anat 25:457–494
Weatherley AH, Gill HS, Rogers SC (1979) Growth dynamics of muscle fibres: dry weight and condition in relation to somatic growth rate in yearling trout (Salmo gairdneri). Can J Zool 57:2385–2392
Webb PW (1978) Hydrodynamics: nonscombroid fish. In: Hoar WS, Randall DJ (eds) Fish physiology 7. Academic Press, New York, pp 189–237
Weibel ER (1973) Stereological techniques for electron microscopic morphometry. Chapt 6. In: Hayat MA (ed) Principles and techniques of electron microscopy. Vol 3. Van Nostrand-Reinhold, Amsterdam, pp 237–296
Willemse JJ, van den Berg PG (1978) Growth of striated muscle fibres in the M. lateralis of the European eel Anguilla anguilla (L.) (Pisces, Teleostei). J Anat 125(3):447–460
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Egginton, S., Johnston, I.A. A morphometric analysis of regional differences in myotomal muscle ultrastructure in the juvenile eel (Anguilla anguilla L.). Cell Tissue Res. 222, 579–596 (1982). https://doi.org/10.1007/BF00213856
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00213856