Abstract
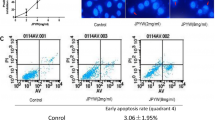
We studied the inhibitory effects of lentinan alone or lentinan combined with docetaxel and cisplatin on growth of gastric cancer cell line BGC823. The cells were divided into lentinan group, docetaxel combined with cisplatin group, and lentinan combined with docetaxel and cisplatin group. Gastric cancer cell line BGC823 was treated with different concentrations of drugs in each group. Tetrazolium-based colorimetric assay (MTT), Annexin V/propidium iodide method and flow cytometry were used to determine the proliferation and apoptosis of the cells in each group. The inhibition ratio was positively related with the concentrations of drugs when BGC823 cells were treated with docetaxel combined with cisplatin from low to high dose. The inhibition ratio of each group further increased after lentinan was added into the medium. The apoptosis rate of 6.25 μg/ml lentinan on BGC823 cells was 19.84 %. The apoptosis rate of BGC823 cells was significantly increased from 50.22 % to 72.06 % after treatment with 6.25 μg/ml lentinan combined with 2.5 μg/ml docetaxel and 50 μg/ml cisplatin. Lentinan has an inhibitory effect on the proliferation of gastric cancer cell line BGC823. Lentinan combined with docetaxel and cisplatin increases the inhibitory effect on the proliferation of BGC823 cells mediated by docetaxel combined with cisplatin. Low concentration of lentinan combined with docetaxel and cisplatin has better therapeutic effects on the proliferation of BGC823 cells compared with high concentrations of docetaxel combined with cisplatin. Lentinan has the ability of inducing BGC823 cell apoptosis and this effect is enhanced when combined with docetaxel and cisplatin.



Similar content being viewed by others
References
Compare D, Rocco A, Nardone G. Risk factors in gastric cancer. Eur Rev Med Pharmacol Sci. 2010;14(4):302–8.
Wagner AD, Grothe W, Haerting J, Kleber G, Grothey A, Fleig WE. Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol. 2006;24(18):2903–9.
Bang YJ, Kang WK, Kang YK, Kim HC, Jacques C, Zuber E, et al. Docetaxel 75 mg/m(2) is active and well tolerated in patients with metastatic or recurrent gastric cancer: a phase II trial. Jpn J Clin Oncol. 2002;32(7):248–54.
Chihara G, Maeda Y, Hamuro J, Sasaki T, Fukuoka F. Inhibition of mouse sarcoma 180 by polysaccharides from Lentinus edodes (Berk.) sing. Nature. 1969;222(5194):687–8.
Suga T, Shiio T, Maeda YY, Chihara G. Antitumor activity of lentinan in murine syngeneic and autochthonous hosts and its suppressive effect on 3-methylcholanthrene-induced carcinogenesis. Cancer Res. 1984;44(11):5132–7.
Fujimoto K, Tomonaga M, Goto S. A case of recurrent ovarian cancer successfully treated with adoptive immunotherapy and lentinan. Anticancer Res. 2006;26(6A):4015–8.
Hamuro J. Anticancer immunotherapy with perorally effective lentinan. Gan To Kagaku Ryoho. 2005;32(8):1209–15.
Zhang LJQ, Ni ZH, Sun J. Prohibitin induces apoptosis in BGC823 gastric cancer cells through the mitochondrial pathway. Asian Pac J Cancer Prev. 2012;13:3803–7.
Ina K, Furuta R, Kataoka T, Kayukawa S, Yoshida T, Miwa T, et al. Lentinan prolonged survival in patients with gastric cancer receiving S-1-based chemotherapy. World J Clin Oncol. 2011;2(10):339–43. doi:10.5306/wjco.v2.i10.339.
Wang JL, Bi Z, Zou JW, Gu XM. Combination therapy with lentinan improves outcomes in patients with esophageal carcinoma. Mol Med Rep. 2012;5(3):745–8. doi:10.3892/mmr.2011.718.
Kamano T, Mikami Y, Shirasaka T. Continuous infusion of 5-fluorouracil plus low-dose cisplatin in tumor-bearing mice. Anticancer Drugs. 1997;8(6):632–6.
Trudeau ME. Docetaxel: a review of its pharmacology and clinical activity. Can J Oncol. 1996;6(1):443–57.
Kupfahl C, Geginat G, Hof H. Lentinan has a stimulatory effect on innate and adaptive immunity against murine Listeria monocytogenes infection. Int Immunopharmacol. 2006;6(4):686–96. doi:10.1016/j.intimp.2005.10.008.
Ina K, Ando T. The use of lentinan for treating gastric cancer. Anticancer Agents Med Chem. 2012. doi:CMCACA-EPUB-20121018-2 [pii].
Oba K, Kobayashi M, Matsui T, Kodera Y, Sakamoto J. Individual patient based meta-analysis of lentinan for unresectable/recurrent gastric cancer. Anticancer Res. 2009;29(7):2739–45.
Kataoka H, Shimura T, Mizoshita T, Kubota E, Mori Y, Mizushima T, et al. Lentinan with S-1 and paclitaxel for gastric cancer chemotherapy improve patient quality of life. Hepatogastroenterology. 2009;56(90):547–50.
Hori T, Ikehara T, Takatsuka S, Fukuoka T, Tendo M, Tezuka K, et al. Combination chemotherapy of S-1/low-dose CDDP/lentinan for advanced gastric cancer. Gan To Kagaku Ryoho. 2011;38(2):293–5.
Murata T, Hatayama I, Kakizaki I, Satoh K, Sato K, Tsuchida S. Lentinan enhances sensitivity of mouse colon 26 tumor to cis-diamminedichloroplatinum (II) and decreases glutathione transferase expression. Jpn J Cancer Res. 1996;87(11):1171–8.
Zhang L, Li X, Xu X, Zeng F. Correlation between antitumor activity, molecular weight, and conformation of lentinan. Carbohydr Res. 2005;340(8):1515–21. doi:10.1016/j.carres.2005.02.032.
Ng ML, Yap AT. Inhibition of human colon carcinoma development by lentinan from shiitake mushrooms (Lentinus edodes). J Altern Complement Med. 2002;8(5):581–9. doi:10.1089/107555302320825093.
Harada K, Itashiki Y, Takenawa T, Ueyama Y. Effects of lentinan alone and in combination with fluoropyrimidine anticancer agent on growth of human oral squamous cell carcinoma in vitro and in vivo. Int J Oncol. 2010;37(3):623–31.
Chen HL, Li DF, Chang BY, Gong LM, Piao XS, Yi GF, et al. Effects of lentinan on broiler splenocyte proliferation, interleukin-2 production, and signal transduction. Poult Sci. 2003;82(5):760–6.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, L., Xiao, Y. & Xiao, N. Effect of lentinan combined with docetaxel and cisplatin on the proliferation and apoptosis of BGC823 cells. Tumor Biol. 34, 1531–1536 (2013). https://doi.org/10.1007/s13277-013-0680-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-0680-8