Abstract
Purpose
To determine the optimal levels of thyroid-stimulating hormone (TSH) levels after administration of recombinant human TSH (rhTSH) to patients with differentiated thyroid cancer (DTC), we have analyzed the clinical parameters that affected the degree of the increase in serum levels of TSH.
Methods
We retrospectively analyzed 276 patients with differentiated thyroid cancer (DTC), post-thyroidectomy and remnant ablation. Pearson’s correlation coefficient test was used to evaluate the correlation between serum levels of TSH after rhTSH stimulation and various clinical factors, including age, sex, height, weight, body mass index (BMI), body surface area (BSA), serum blood urea nitrogen, creatinine, and estimated glomerular filtration rate (GFR). Linear regression analysis was used to determine the predictors of the degree of increase in serum TSH level after rhTSH stimulation.
Results
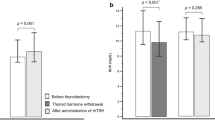
After the rhTSH injections, all subjects achieved TSH levels of >30 μU/mL, with a mean of 203.8 ± 83.4 μU/mL. On univariate analysis, age (r = 0.255) and serum creatinine (r = 0.169) level were positive predictors for higher levels of serum TSH after rhTSH stimulation, while weight (r = –0.239), BMI (r = –0.223), BSA (r = –0.217), and estimated GFR (r = –0.199) were negative predictors. Multiple linear regression analysis revealed that serum creatinine was the most powerful independent predictor for serum levels of TSH, followed by age, BSA, and BMI.
Conclusions
An increment in serum TSH after rhTSH stimulation was significantly affected by age, BSA, BMI, and creatinine, with creatinine being the most powerful predictor. By understanding the difference in the increased levels of TSH in various subjects, their dose of rhTSH can be adjusted during scheduling for radioiodine ablation, or during follow-up (recurrence surveillance) after surgery and ablation.


Similar content being viewed by others
References
Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartofsky L, et al. A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocr Metab. 2003;88:1433–41.
Schlumberger M, Berg G, Cohen O, Duntas L, Jamar F, Jarzab B, et al. Follow-up of low-risk patients with differentiated thyroid carcinoma: a European perspective. Eur J Endocrinol. 2004;150:105–12.
Dietlein M, Busemeyer S, Kobe C, Schmidt M, Theissen P, Schicha H. Recombinant human TSH versus hypothyroidism. cost-minimization-analysis in the follow-up care of differentiated thyroid carcinoma. Nuklearmedizin. 2010;49:216–24.
Heinzel A, Kley K, Mueller HW, Hautzel H. A comparison of rh-TSH and thyroid hormone withdrawal in patients with differentiated thyroid cancer: preliminary evidence for an influence of age on the subjective well-being in hypothyroidism. Horm Metab Res. 2012;44:54–9.
Lee J, Yun MJ, Nam KH, Chung WY, Soh EY, Park CS. Quality of life and effectiveness comparisons of thyroxine withdrawal, triiodothyronine withdrawal, and recombinant thyroid-stimulating hormone administration for low-dose radioiodine remnant ablation of differentiated thyroid carcinoma. Thyroid. 2010;20:173–9.
Chow SM, Au KH, Choy TS, Lee SH, Yeung NY, Leung A, et al. Health-related quality-of-life study in patients with carcinoma of the thyroid after thyroxine withdrawal for whole body scanning. Laryngoscope. 2006;116:2060–6.
Sanchez R, Espinosa-de-los-Monteros AL, Mendoza V, Brea E, Hernandez I, Sosa E, et al. Adequate thyroid-stimulating hormone levels after levothyroxine discontinuation in the follow-up of patients with well-differentiated thyroid carcinoma. Arch Med Res. 2002;33:478–81.
Serhal DI, Nasrallah MP, Arafah BM. Rapid rise in serum thyrotropin concentrations after thyroidectomy or withdrawal of suppressive thyroxine therapy in preparation for radioactive iodine administration to patients with differentiated thyroid cancer. J Clin Endocr Metab. 2004;89:3285–9.
Pacini F, Castagna MG. Diagnostic and therapeutic use of recombinant human TSH (rhTSH) in differentiated thyroid cancer. Best Pract Res Clin Endocrinol Metab. 2008;22:1009–21.
Castagna MG, Pinchera A, Marsili A, Giannetti M, Molinaro E, Fierabracci P, et al. Influence of human body composition on serum peak thyrotropin (TSH) after recombinant human TSH administration in patients with differentiated thyroid carcinoma. J Clin Endocr Metab. 2005;90:4047–50.
Hautzel H, Pisar E, Lindner D, Schott M, Grandt R, Muller HW. Impact of renal function and demographic/anthropomorphic variables on peak thyrotropin after recombinant human thyrotropin stimulation: a stepwise forward multiple-regression analysis. Thyroid. 2013;23:662–70.
Montesano T, Durante C, Attard M, Crocetti U, Meringolo D, Bruno R, et al. Age influences TSH serum levels after withdrawal of l-thyroxine or rhTSH stimulation in patients affected by differentiated thyroid cancer. Biomed Pharmacother. 2007;61:468–71.
Over R, Nsouli-Maktabi H, Burman KD, Jonklaas J. Age modifies the response to recombinant human thyrotropin. Thyroid. 2010;20:1377–84.
Vitale G, Lupoli GA, Ciccarelli A, Lucariello A, Fittipaldi MR, Fonderico F, et al. Influence of body surface area on serum peak thyrotropin (TSH) levels after recombinant human TSH administration. J Clin Endocr Metab. 2003;88:1319–22.
Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition. 1989;5:303–11. discussion 12-3.
Levey AS, Coresh J, Greene T, Stevens LA, Zhang YP, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–54.
Bakke J, Lawrence N, Roy S. Disappearance rate of exogenous thyroid-stimulating hormone (TSH) in man. J Clin Endocr Metab. 1962;22:352–63.
Cuttelod S, Lemarchand-Beraud T, Magnenat P, Perret C, Poli S, Vannotti A. Effect of age and role of kidneys and liver on thyrotropin turnover in man. Metabolism. 1974;23:101–13.
Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, et al. Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin Chem. 2006;52:5–18.
Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–54.
Perros P. Recombinant human thyroid-stimulating hormone (rhTSH) in the radioablation of well-differentiated thyroid cancer: preliminary therapeutic experience. J Endocrinol Invest. 1999;22(11 Suppl):30–4.
Luster M, Reinhardt W, Korber C, Lassmann M, Haenscheid H, Michalowski U, et al. The use of recombinant human TSH in a patient with metastatic follicular carcinoma and insufficient endogenous TSH production. J Endocrinol Invest. 2000;23:473–5.
Jarzab B, Handkiewicz-Junak D, Roskosz J, Puch Z, Wygoda Z, Kukulska A, et al. Recombinant human TSH-aided radioiodine treatment of advanced differentiated thyroid carcinoma: a single-centre study of 54 patients. Eur J Nucl Med Mol Imaging. 2003;30:1077–86.
Adler ML, Macapinlac HA, Robbins RJ. Radioiodine treatment of thyroid cancer with the aid of recombinant human thyrotropin. Endocr Pract. 1998;4:282–6.
Lippi F, Capezzone M, Angelini F, Taddei D, Molinaro E, Pinchera A, et al. Radioiodine treatment of metastatic differentiated thyroid cancer in patients on L-thyroxine, using recombinant human TSH. Eur J Endocrinol. 2001;144:5–11.
Vargas GE, Uy H, Bazan C, Guise TA, Bruder JM. Hemiplegia after thyrotropin alfa in a hypothyroid patient with thyroid carcinoma metastatic to the brain. J Clin Endocr Metab. 1999;84:3867–71.
Braga M, Ringel MD, Cooper DS. Sudden enlargement of local recurrent thyroid tumor after recombinant human TSH administration. J Clin Endocr Metab. 2001;86:5148–51.
Valle LA, Gorodeski Baskin RL, Porter K, Sipos JA, Khawaja R, Ringel MD, et al. In thyroidectomized patients with thyroid cancer, a serum thyrotropin of 30 µU/mL after thyroxine withdrawal is not always adequate for detecting an elevated stimulated serum thyroglobulin. Thyroid. 2013;23:185–93.
Submission Statement
This manuscript has not been published before or is not under consideration for publication anywhere else and has been approved by all co-authors.
Funding
This study was funded by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST) (no. 2009-0078222, 2009-0078234); a grant of the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (grant no. A111345); and a grant of the Kyungpook National University Research Fund, 2012.
Conflict of Interest
Seung Hyun Son, Sang-Woo Lee, Ji-hoon Jung, Choon-Young Kim, Do-Hoon Kim, Shin Young Jeong, Byeong-Cheol Ahn, and Jaetae Lee declare that they have no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Son, S.H., Lee, SW., Jung, Jh. et al. Analysis of Clinical Factors for the Determination of Optimal Serum Level of Thyrotropin After Recombinant Human Thyroid-Stimulating Hormone Administration. Nucl Med Mol Imaging 49, 268–275 (2015). https://doi.org/10.1007/s13139-015-0348-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-015-0348-y