Abstract
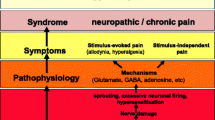
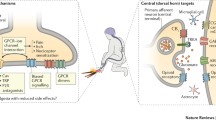
Chronic pain of neuropathic origin is an unmet therapeutic challenge that may require the development of novel drug entities. The resources involved in developing novel drugs and testing them in clinical trials are so large that few candidates can be fully tested in practice. A poor choice can have major repercussions. At the same time, the research community has identified large numbers of potential pharmacological targets for analgesia in recent years, with individual investigators promoting favorites with increasing zeal. Because of the uncertain predictive value of animal models of neuropathic pain, it is essential to consider rational strategic approaches to selecting the most likely candidate targets.
Similar content being viewed by others
References and Recommended Reading
Ruda MA, Ling QD, Hohmann AG, et al.: Altered nociceptive neuronal circuits after neonatal peripheral inflammation. Science 2000, 289:628–631.
Devor M: Pain and “state” induced analgesia: an introduction. In Mechanisms of Stress-Induced Analgesia. Edited by Curzon G, Trickebank MD. Sussex: Wiley; 1984:1–18.
Jordt SE, Julius D: Molecular basis for species-specific sensitivity to “hot” chili peppers. Cell 2002, 108:421–430.
Froy O, Gurevitz M: Membrane potential modulators: a thread of scarlet from plants to humans. FASEB J 1998, 12:1793–1796.
Devor M: Neuropathic pain: What do we do with all these theories ? Acta Anesthesiologica Scandinavica 2001, 45:1121–1127.
Hokfelt T, Zhang X, Xu ZQ, et al.: Phenotype regulation in dorsal root ganglion neurons after nerve injury: focus on peptides and their receptors. In Molecular Neurobiolgy of Pain. Edited by Borsook D. Seattle: IASP Press; 1997:115–143.
Costigan M, Befort K, Karchewski L, et al.: Replicate highdensity rat genome oligonucleotide microarrays reveal hundreds of regulated genes in the dorsal root ganglion after peripheral nerve injury. BMC Neurosci 2002, 3:16–28.
Xiao HS, Huang QH, Zhang FX, et al.: Identification of gene expression profile of dorsal root ganglion in the rat peripheral axotomy model of neuropathic pain. Proc Natl Acad Sci U S A 2002, 99:8360–8365.
Tachibana T, Noguchi K, Ruda MA: Analysis of gene expression following spinal cord injury in rat using complementary DNA microarray. Neurosci Lett 2002, 327:133–137.
Ji RR, Kohno T, Moore KA, Woolf CJ: Central sensitization and LTP: Do pain and memory share similar mechanisms? Trends Neurosci 2003, 26:696–705.
Devor M, Seltzer Z: Pathophysiology of damaged nerves in relation to chronic pain. In Textbook of Pain, edn 4. Edited by Wall PD, Melzack R. London: Churchill Livingstone; 1999:129–164.
Amir R, Michaelis M, Devor M: Membrane potential oscillations in dorsal root ganglion neurons: role in normal electrogenesis and in neuropathic pain. J Neurosci 1999, 19:8589–8596.
Amir R, Liu CN, Kocsis JD: Oscillatory mechanism in primary sensory neurons. Brain 2002, 125:421–435. This paper may be a bit heavy for some readers of this journal, but it is the latest on a phenomenon, subthreshold oscillations, that lies at the very heart of ectopic hyperexcitability and neuropathic pain.
Chung JM, Dib-Hajj SD, Lawson SN: 2003 Sodium channel subtypes and neuropathic pain. In Proceedings of the 10th World Congress on Pain: Progress in Pain Research and Management, vol 24. Edited by Dostrovsky JO, Carr DB, Koltzenburg M. Seattle: IASP Press; 2003:99–114. A detailed discussion of the role played by Na+-channels in nerve excitability and chronic pain. This is currently a “hot topic” and this is an accessible review of the current status of the field.
Waxman SG: 2002, Sodium Channels and Neuronal Hyperexcitability, vol 241. London: Novartis Foundation; 2002.
Liu CN, Wall PD, Ben-Dor E, et al.: Tactile allodynia in the absence of C-fiber activation: altered firing properties of DRG neurons following spinal nerve injury. Pain 2000, 85:503–521.
Lyu YS, Park SK, Chung K, Chung JM: Low dose of tetrodotoxin reduces neuropathic pain behaviors in an animal model. Brain Res 2000, 871:98–103.
Kerr BJ, Souslova V, McMahon SB, Wood JN: A role for the TTX-resistant sodium channel Nav1.8 in NGF-induced hyperalgesia, but not neuropathic pain. Neuroreport 2001, 12:3077–3080.
Okuse K, Chaplin R, McMahon SB, et al.: Regulation of expression of the sensory neuron-specific sodium channel SNS in inflammatory and neuropathic pain. Mol Cell Neurosci 1997, 10:196–207.
Roza C, Laird J, Souslova V, et al.: The tetrodotoxin-resistant Na + channel Nav1.8 is essential for the expression of spontaneous activity in damaged sensory axons of mice. J Physiol 2003, 550:921–926.
Devor M, Raber P: Heritability of symptoms in an animal model of neuropathic pain. Pain 1990, 42:51–67.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Devor, M. Strategies for finding new pharmacological targets for neuropathic pain. Current Science Inc 8, 187–191 (2004). https://doi.org/10.1007/s11916-004-0050-8
Issue Date:
DOI: https://doi.org/10.1007/s11916-004-0050-8