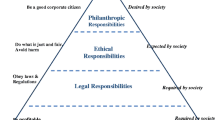
Abstract
The pharmaceutical industry has been criticised for pervasive misconduct. These concerns have generally resulted in increasing regulation. While such regulation is no doubt necessary, it tends to assume that everyone working for pharmaceutical companies is equally motivated by commerce, without much understanding of the specific views and experiences of those who work in different parts of the industry. In order to gain a more nuanced picture of the work that goes on in the “medical affairs” departments of pharmaceutical companies, we conducted 15 semi-structured interviews with professionals working in medical departments of companies in Sydney, Australia. We show that this group of pharmaceutical professionals are committed to their responsibilities both to patients, research participants, and the public and to their companies. Despite the discrepancies between these commitments, our participants did not express much cognitive dissonance, and this appeared to stem from their use of two dialectically related strategies, one of which embraces commerce and the other of which resists the commercial imperative. We interpret these findings through the lens of institutional theory and consider their implications for pharmaceutical ethics and governance.

Similar content being viewed by others
References
Angell, M. 2004. The truth about the drug companies: How they deceive us and what to do about it. New York: Random House.
Biedenbach, T. 2011. The power of combinative capabilities: Facilitating the outcome of frequent innovation in pharmaceutical R&D projects. Project Management Journal 42(2): 63–80.
Blind, K. 2012. The influence of regulations on innovation: A quantitative assessment for OECD countries. Research Policy 41(2): 391–400.
Califf, R.M., D.A. Zarin, J.M. Kramer, R.E. Sherman, L.H. Aberle, and A. Tasneem. 2012. Characteristics of clinical trials registered in ClinicalTrials.gov, 2007–2010. The Journal of the American Medical Association 307(17): 1838–1847.
Charmaz, K. 2006. Constructing grounded theory: A practical guide through qualitative analysis. London: Sage.
DeMartino, J.K. 2012. The Physician Payment Sunshine Act. Journal of the National Comprehensive Cancer Network 10(3): 423–424.
DiMaggio, P.J. 1991. Constructing an organizational field as a professional project: U.S. art museums. In The new institutionalism in organizational analysis: 1920–1940, ed. W.W. Powell and P.J. DiMaggio, 267–292. Chicago: University of Chicago Press.
Dingel, M., A. Hicks, M. Robinson, and B. Koenig. 2012. Integrating genetic studies of nicotine addiction into public health practice: Stakeholder views on challenges, barriers and opportunities. Public Health Genomics 15(1): 46–55.
Eisenstein, E., R. Collins, B. Cracknell, et al. 2008. Sensible approaches for reducing clinical trial costs. Clinical Trials 5(1): 75–84.
Elliot, C. 2010. White coat, black hat: Adventures on the dark side of medicine. Boston: Beacon Press.
Forbes, T., J. Hallier, and L. Kelly. 2004. Doctors as managers: Investors and reluctants in a dual role. Health Services Management Research 17(3): 167–176.
Gatrell, J., and T. White. 1996. Doctors and management—the development dilemma. Journal of Management in Medicine 10(2): 6–12.
Glynn, M.A., and M. Lounsbury. 2005. From the critics’ corner: Logic blending, discursive change and authenticity in a cultural production system. Journal of Management Studies 42(5): 1031–1055.
Goodrick, E., and T. Reay. 2011. Constellations of institutional logics: Changes in the professional work of pharmacists. Work and Occupations 38(3): 372–416.
Ham, C., J. Clark, P. Spurgeon, H. Dickinson, and K. Armit. 2011. Doctors who become chief executives in the NHS: From keen amateurs to skilled professionals. Journal of the Royal Society of Medicine 104(3): 113–119.
Hemminki, E., H. Toiviainen, and L. Vuorenkoski. 2010. Co-operation between patient organisations and the drug industry in Finland. Social Science and Medicine 70(8): 1171–1175.
Hoffman, A.J. 1999. Institutional evolution and change: Environmentalism and the U.S. chemical industry. Academy of Management Journal 42(4): 351–371.
Horikawa, H., M. Tsubouchi, and K. Kawakami. 2009. Industry views of biosimilar development in Japan. Health Policy 91(2): 189–194.
Iedema, R., P. Degeling, J. Braithwaite, and L. White. 2004. “It’s an interesting conversation I’m hearing”: The doctor as manager. Organization Studies 25(1): 15–33.
Kesselheim, A.S., M.M. Mello, and D.M. Studdert. 2011. Strategies and practices in off-label marketing of pharmaceuticals: A retrospective analysis of whistleblower complaints. PLoS Medicine 8(4): e1000431.
Kitchener, M. 2002. Mobilizing the logic of managerialism in professional fields: The case of academic health centre mergers. Organization Studies 23(3): 391–420.
Laeeque, H., H. Boon, N. Kachan, J. Cohen, and J. D’Cruz. 2006. The Canadian Natural Health Products (NHP) regulations: Industry perceptions and compliance factors. BMC Health Services Research 6: 63. doi:10.1186/1472-6963-6-63.
Lee, M., and J. Kohler. 2010. Benchmarking and transparency: Incentives for the pharmaceutical industry’s corporate social responsibility. Journal of Business Ethics 95(4): 641–658.
Lexchin, J., L. Bero, B. Djulbegovic, and O. Clark. 2003. Pharmaceutical industry sponsorship and research outcome and quality: Systematic review. British Medical Journal 326(7400): 1167B–1170B.
Mackenzie, F.J., C.F.C. Jordens, R.A. Ankeny, J. McPhee, and I.H. Kerridge. 2007. Direct-to-consumer advertising under the radar: The need for realistic drugs policy in Australia. Internal Medicine Journal 37(4): 224–228.
Martin, E. 2006. Pharmaceutical virtue. Culture, Medicine and Psychiatry 30(2): 157–174.
Meyer, R.E., and G. Hammerschmid. 2006. Changing institutional logics and executive identities: A managerial challenge to public administration in Austria. The American Behavioral Scientist 49(7): 1000–1014.
Montgomery, K. 2001. Physician executives: The evolution and impact of a hybrid profession. Advances in Health Care Management 2: 215–241.
Montgomery, K., and A.L. Oliver. 2009. Shifts in guidelines for ethical scientific conduct: How public and private organizations create and change norms of research integrity. Social Studies of Science 39(1): 137–155.
Morse, J.M. 1994. “Emerging from the data”: The cognitive processes of analysis in qualitative inquiry. In Critical issues in qualitative research methods, ed. J.M. Morse, 23–42. Thousand Oaks, CA: Sage.
Moynihan, R., I. Heath, and D. Henry. 2002. Selling sickness: The pharmaceutical industry and disease mongering. British Medical Journal 324(7342): 886–890.
Nelson, A.J. 2005. Cacophany or harmony? Multivocal logics and technology licensing by the Stanford University Department of Music. Industrial and Corporate Change 14(1): 93–118.
Pache, A.-C., and F. Santos. 2011. Inside the hybrid organization: An organizational level view of responses to conflicting institutional demands. Cergy-Pontoise Cedex: ESSEC Business School. http://www.essec.edu/faculty/showDeclFileRes.do?declId=9761&key=__workpaper__.
Reay, T., and C.B. Hinings. 2009. Managing the rivalry of competing institutional logics. Organization Studies 30(6): 629–652.
Scott, W.R. 2008. Approaching adulthood: The maturing of institutional theory. Theory and Society 37(5): 427–442.
Scott, W.R., M. Reuf, P. Mendel, and C.A. Caronna. 2000. Institutional change and health care organization: From professional dominance to managed care. Chicago: University of Chicago Press.
Shapin, S. 2008. The scientific life: A moral history of a late modern vocation. Chicago: University of Chicago Press.
Suddaby, R., and R. Greenwood. 2005. Rhetorical strategies of legitimacy. Administrative Science Quarterly 50(1): 35–67.
Tabarrok, A. 2009. From off-label prescribing towards a new FDA. Medical Hypotheses 72(1): 11–13.
Thornton, P.H. 2004. Markets from culture. Stanford: Stanford University Press.
Trouiller, P., P. Olliaro, E. Torreele, J. Orbinski, R. Laing, and N. Ford. 2002. Drug development for neglected diseases: A deficient market and a public-health policy failure. Lancet 359(9324): 2188–2194.
Wager, E., and A. Herxheimer. 2003. Peer review and the pharmaceutical industry. In Peer review in health sciences, ed. F. Godlee and T. Jefferson, 130–139. London: BMJ Books.
Wang, N., W.L. Lipworth, J.E. Ritchie, K.M. Williams, and R.O. Day. 2010. Eliciting views of Australian pharmaceutical industry employees on collaboration and the concept of Quality Use of Medicines. Internal Medicine Journal 41(4): 314–320.
Wazana, A. 2000. Physicians and the pharmaceutical industry: Is a gift ever just a gift? The Journal of the American Medical Association 283(3): 373–380.
Wertheimer, A., and J. Norris. 2009. Safeguarding against substandard/counterfeit drugs: Mitigating a macroeconomic pandemic. Research in Social and Administrative Pharmacy 5(1): 4–16.
Zaharia, R., and M. Ghenghea. 2011. Corporate social responsibility in the pharmaceutical industry: The Romania’s case. Transformations in Business and Economics 10(2B [Special Issue: S1]): 730–740.
Funding Acknowledgement
This research was funded by a National Health and Medical Research Council Postdoctoral Fellowship [630726] and the University of California Academic Senate.
Conflict of Interest
The authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lipworth, W., Montgomery, K. & Little, M. How Pharmaceutical Industry Employees Manage Competing Commitments in the Face of Public Criticism. Bioethical Inquiry 10, 355–367 (2013). https://doi.org/10.1007/s11673-013-9449-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11673-013-9449-4