Abstract
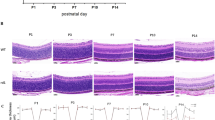
Recent gene expression studies on mouse models for retinal degeneration identified deregulation of Pituitary tumor transforming gene 1 (Pttg1) as a potential susceptibility factor involved in photoreceptor cell death. Pttg1 is a transcription regulatory protein involved in sister chromatid segregation, and Pttg1 −/− mice exhibit testicular and splenic hypoplasia, thymic hyperplasia, aberrant cell cycle progression, chromosome instability, and impaired glucose homeostasis leading to diabetes, particularly in older males. Due to Pttg1 deregulation in dystrophic retinas, we characterized Pttg1 −/− retinas using Hematoxylin and Eosin (H&E) staining, immunohistochemistry (IHC), and electroretinography (ERG). Seven month old Pttg1 −/− mice were also examined for a diabetic retinopathy phenotype using Fluorescein Angiography (FA) to test for neovascularization. Our data reveal that up to 9 months of age, Pttg1 −/− retinas have a healthy morphology and normal photoreceptor function. This study lays the groundwork for further investigation into the relevance of Pttg1 in retinal dystrophy.






Similar content being viewed by others
References
Wang Z, Yu R, Melmed S (2001) Mice lacking pituitary tumor transforming gene show testicular and splenic hypoplasia, thymic hyperplasia, thrombocytopenia, aberrant cell cycle progression, and premature centromere division. Mol Endocrinol 15:1870–1879
Caporali S, Levati L, Starace G et al (2008) AKT is activated in an ataxia-telangiectasia and Rad3-related-dependent manner in response to temozolomide and confers protection against drug-induced cell growth inhibition. Mol Pharmacol 74:173–183
Wang Z, Moro E, Kovacs K et al (2003) Pituitary tumor transforming gene-null male mice exhibit impaired pancreatic beta cell proliferation and diabetes. Proc Natl Acad Sci USA 100:3428–3432
Yetemian RM, Brown BM, Craft CM (2010) Loss of G-protein coupled receptor kinase 1 in Nrl −/− mice leads to neovascularization, enhanced inflammatory response, and age-related cone dystrophy. Invest Ophthalmol Vis Sci 51:6196–6206
van de Pavert SA, Sanz AS, Aartsen WM et al (2007) Crb1 is a determinant of retinal apical Muller glia cell features. Glia 55:1486–1497
Fernandez-Medarde A, Barhoum R, Riquelme R et al (2009) RasGRF1 disruption causes retinal photoreception defects and associated transcriptomic alterations. J Neurochem 110:641–652
Kim SR, Fishkin N, Kong J et al (2004) Rpe65 Leu450Met variant is associated with reduced levels of the retinal pigment epithelium lipofuscin fluorophores A2E and iso-A2E. Proc Natl Acad Sci USA 101:11668–11672
Pittler SJ, Baehr W (1991) Identification of a nonsence mutation in the rod photoreceptor cGMP phosphodies-terase β-subunit gene of the rd mouse. Proc Natl Acad Science USA 88:8322–8326
Brown BM, Ramirez T, Rife L, Craft CM (2010) Visual arrestin 1 contributes to cone photoreceptor survival and light-adaptation. Invest Ophthalmol Vis Sci 51:2372–2380
Zhu X, Brown B, Rife L, Craft CM (2006) Slowed photoresponse recovery and age-related degeneration in cones lacking G protein-coupled receptor kinase 1. In: Hollyfield JG, Anderson RE, LaVail MM (eds) Advances in experimental medicine and biology, vol 572. Springer, pp 133–139
Zhu X, Li A, Brown B, Weiss ER et al (2002) Mouse cone arrestin expression pattern: light induced translocation in cone photoreceptors. Mol Vis 8:462–471
Zhu X, Wu K, Rife L et al (2005) Carboxypeptidase E is required for normal synaptic transmission from photoreceptors to the inner retina. J Neurochem 95:1351–1362
Zhu X, Craft CM (2000) Modulation of CRX transactivation activity by phosducin isoforms. Mol Cell Biol 20:5216–5226
Daniele LL, Lillo C, Lyubarsky AL et al (2005) Cone-like morphological, molecular, and electrophysiological features of the photoreceptors of the Nrl knockout mouse. Invest Ophthalmol Vis Sci 46:2156–2167
Oh EC, Cheng H, Hao H et al (2008) Rod differentiation factor NRL activates the expression of nuclear receptor NR2E3 to suppress the development of cone photoreceptors. Brain Res 1236:16–29
Caporali A, Pani E, Horrevoets AJ et al (2008) Neurotrophin p75 receptor (p75NTR) promotes endothelial cell apoptosis and inhibits angiogenesis: implications for diabetes-induced impaired neovascularization in ischemic limb muscles. Circ Res 103:e15–e26
Acknowledgments
Cheryl M. Craft, Ph.D. is the Mary D. Allen Chair in Vision Research, Doheny Eye Institute and a Research to Prevent Blindness Senior Scientific Investigator. Supported by NIH EY015851 (CMC), National Eye Institute Core Grant EY03040 (Doheny Eye Institute), Research to Prevent Blindness (DEI), the Foundation Fighting Blindness (DEI), Mary D. Allen Foundation (Richard Newton Lolley Memorial Scholarship (RMY), William Hansen Sandberg Memorial Foundation (RMY), Tony Gray Foundation, Dorie Miller, and Dr. Paul R. Burton. We sincerely appreciate the generous continued support for our vision research program by Mrs. Mary D. Allen. We also thank Dr. Shlomo Melmed for the Pttg1 −/− mice and Dr. Larry A. Donoso for the Arr1 D9F2 antibody. The authors acknowledge the expert technical support of Bruce Brown, Alexander Wang for his help with the experiments, Lawrence Rife for ERG analysis, Fernando Gallardo for FA, and Ernesto Barron for figures. Also, we thank Dr. Shun-Ping Huang, Dr. Freddi Zuniga and Leng-Ying Chen for their scientific contributions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: In Honor of Dr. Dianna Johnson.
Rights and permissions
About this article
Cite this article
Yetemian, R.M., Craft, C.M. Characterization of the Pituitary Tumor Transforming Gene 1 Knockout Mouse Retina. Neurochem Res 36, 636–644 (2011). https://doi.org/10.1007/s11064-010-0334-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-010-0334-9