Abstract
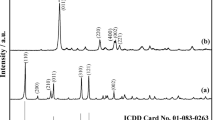
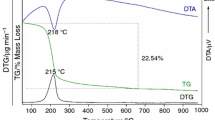
The dehydration of LiCl·H2O was studied under inert helium atmosphere by DTA/TG for different heating rates. The dehydration of LiCl·H2O proceeds through a two step reaction between 99–110 and 160–186°C, respectively. It leads to the formation of LiCl·0.5H2O as intermediate compound. The proposed mechanism is:

and

Based on the temperature peak of the DTA signals the activation energies of the two reactions were determined to be 240 kJ mol−1 (step 1) and 137 kJ mol−1 (step 2), respectively.
Similar content being viewed by others
References
P. Masset, A. Henry, J. Y. Poinso and J. C. Poignet, J. Power Sources, 160 (2006) 752.
M. Manewa and H. P. Fritz, A. Anorg. Allg. Chem., 296 (1973) 279.
G. F. Huttig and W. Steudemann, Z. Phys. Chem., 126 (1927) 279.
W. Voigt and D. Zeng, Pure Appl. Chem., 74 (2002) 1909.
M. R. Conde, Intern. J. Thermal Sci., 43 (2004) 367.
P. J. Masset, Ph.D. Thesis, National Polytechnic Institute of Grenoble (2002).
R. Parash, F. Broitman, U. Mor, D. Ozer and A. Bettelheim, J. Electrochem. Soc., 131 (1984) 2531.
H. E. Kissinger, J. Res. Nat. Bur. Stand., 57 (1956) 217.
H. E. Kissinger, Anal. Chem., 29 (1957) 1702.
A. K. Galwey, Thermochim. Acta, 355 (2000) 181.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Masset, P.J. Thermogravimetric study of the dehydration reaction of LiCl·H2O. J Therm Anal Calorim 96, 439–441 (2009). https://doi.org/10.1007/s10973-008-9399-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-008-9399-y