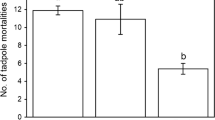
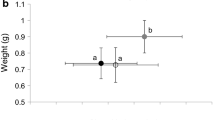
Abstract
When confronted by signals of predators presence, many aquatic organisms modify their phenotype (e.g., behaviour or morphology) to reduce their risk of predation. A principal means by which organisms assess predation risk is through chemical cues produced by the predators and/or prey during predation events. Such responses to predation risk can directly affect prey fitness and indirectly affect the fitness of species with which the prey interacts. Accurate assessment of the cue will affect the adaptive nature, and hence evolution, of the phenotypic response. It is therefore, important to understand factors affecting the assessment of chemical cues. Here I examined the effect of the age of chemical cues arising from an invertebrate predator, a larval dragonfly (Anax junius), which was fed bullfrog tadpoles, on the behavioural response (activity level and position) of bullfrog tadpoles. The bullfrog response to chemical cues declined as a function of chemical cue age, indicating the degradation of the chemical cue was on the order of 2–4 days. Further, the decay occurred more rapidly when the chemical cue was placed in pond water rather than well water. These results indicate a limitation of the tadpoles to interpret factors that affect the magnitude of the chemical cue and hence accurately assess predation risk. These findings also have implications for experimental design and the adaptation of phenotypic responses to chemical cues of predation risk.
Similar content being viewed by others
References
Agrawal A. A. (2001). Phenotypic plasticity in the interactions and evolution of species. Science 294: 321–326
Chivers D. P. and Smith R. J. F. (1998). Chemical alarm signalling in aquatic predator–prey systems: a review and prospect us. Écoscience 5: 338–352
Kats L. B. and Dill L. M. (1998). The scent of death: chemosensory assessment of predation risk by prey animals. Écoscience 5: 361–394
Lass S. and Spaak P. (2003). Chemically induced anti-predator defences in plankton: a review. Hydrobiologia 491: 221–239
Lima S. L. (1998). Stress and decision making under the risk of predation: recent developments from behavioural, reproductive and ecological perspectives. Stress and Behaviour 27: 215–290
Lima S. L. and Bednekoff P. A. (1999). Temporal variation in danger drives antipredator behaviour: the predation risk allocation hypothesis. American Naturalist 153: 649–659
Loose C. J. and Dawidowicz P. (1994). Trade-offs in diel vertical migration by zooplankton – the costs of predator avoidance. Ecology 75: 2255–2263
Loose C. J., Vonelert E. and Dawidowicz P. (1993). Chemically-induced vertical migration in daphnia – a bioassay for kairomones exuded by fish. Archiv für Hydrobiologie 126: 329–337
Peacor S. D. (2002). Positive effects of predators on prey growth rate through induced modifications of prey behavior. Ecology Letters 5: 77–85
Peacor S. D. and Werner E. E. (2001). The contribution of trait-mediated indirect effects to the net effects of a predator. Proceedings of the National Academy of Sciences of the United States of America 98: 3904–3908
Persons M. H., Walker S. E., Rypstra A. L. and Marshall S. D. (2001). Wolf spider predator avoidance tactics and survival in the presence of diet-associated predator cues (Araneae: Lycosidae). Animal Behaviour 61: 43–51
Relyea R. A. (2003). How prey respond to combined predators: a review and an empirical test. Ecology 84: 1827–1839
Schmitz O. J., Krivan V. and Ovadia O. (2004). Trophic cascades: the primacy of trait-mediated indirect interactions. Ecology Letters 7: 153–163
Schoeppner N. M. and Relyea R. A. (2005). Damage, digestion and defense: the roles of alarm cues and kairomones for inducing prey defenses. Ecology Letters 8: 505–512
SPSS 12.0 for Windows, Rel. 12.0.0., 2003 Chicago: SPSS Inc
Stearns S. C. (1989). The evolutionary significance of phenotypic plasticity. BioScience 39: 436–445
Tollrian, R. and & C. D. Harvell (eds), (1999). The Ecology and Evolution of Inducible Defenses. Princeton University Press, New Jersey
Turner A. M. and Montgomery S. L. (2003). Spatial and temporal scales of predator avoidance: experiments with fish and snails. Ecology 84: 616–622
Van Buskirk J. and Arioli M. (2002). Dosage response of an induced defense: how sensitive are tadpoles to predation risk?. Ecology 83: 1580–1585
Wade J. W. (1992). Sewall Wright: gene interaction and the Shifting Balance Theory. In: Antonovics, J. and Futuyma, D. (eds) Oxford Surveys of Evolutionary Biology VI, pp 35–62. Oxford University Press, Oxford
Weber A. (2003). More than one ‘fish kairomone’? Perch and stickleback kairomones affect Daphnia life history traits differently. Hydrobiologia 498: 143–150
Werner E. E. and Anholt B. R. (1996). Predator-induced behavioural indirect effects: consequences to competitive interactions in anuran larvae. Ecology 77: 157–169
Werner E. E. and Peacor S. D. (2003). A review of trait-mediated indirect interactions. Ecology 84: 1083–1100
West-Eberhard M. J. (1989). Phenotypic plasticity and the origins of diversity. Annual Review of Ecology and Systematics 20: 249–278
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peacor, S.D. Behavioural response of bullfrog tadpoles to chemical cues of predation risk are affected by cue age and water source. Hydrobiologia 573, 39–44 (2006). https://doi.org/10.1007/s10750-006-0256-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-006-0256-3