Abstract
Background
Although a high prevalence of macrovascular disease (MVD) has been reported in patients with stage 3 chronic kidney disease (CKD), few studies have reported its risk with respect to the underlying cause of kidney disease. This study investigated the prevalence of MVD in type 2 diabetic patients with CKD stratified by CKD stage, as defined by estimated glomerular filtration rate (eGFR), as well as the risk factors for MVD.
Methods
1493 patients with diabetic CKD (1273 males, 220 females) were stratified by CKD stage (stage 1: 39, stage 2: 272, stage 3: 1052, stage 4: 101, stage 5: 29) based on eGFR calculated by the Japanese formula and averaged over 8 months. MVD was defined as one of the following: coronary heart disease (CHD), stroke or arteriosclerosis obliterans (ASO).
Results
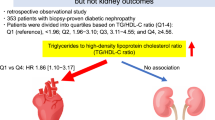
The prevalence of MVD was 18.6%. A significant increasing trend in MVD prevalence was observed from stage 3 (17.78%) to 4 (52.48%). According to a receiver operating characteristic curve analysis on MVD prevalence in stage 3 patients, an eGFR of 46.4 ml/min/1.73 m2 was determined to be a critical cut-off level. Proteinuria, eGFR <60 ml/min/1.73 m2 and hyperuricemia were independent risk factors for MVD.
Conclusions
In patients with diabetic CKD, a significant increase in MVD prevalence was observed from stage 3 to 4. An eGFR of 46.4 ml/min/1.73 m2 is a critical level that affects MVD prevalence. From the perspective of cardiorenal association, CKD stage 3 should be divided into two substages. As hyperuricemia is related to an increased risk of MVD, uric acid control may be important in reducing MVD risk in diabetic CKD.


Similar content being viewed by others
References
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266.
Mann JFE, Gerstein HC, Pogue J, Lonn E, Dagenais GR, McQueen M, for the HOPE Investigators, et al. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE Randomized Trial. Ann Intern Med. 2001;134:629–36.
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296–305.
Anavekar NS, McMurray JJV, Velazquez EJ, Solomon SD, Kober L, Rouleau JL, et al. Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N Engl J Med. 2004;351:1285–95.
Keith DS, Nichols GA, Gullion CM, Brown JB, Smith DH. Longitudinal follow-up and outcomes among a population with chronic kidney disease in a large managed care organization. Arch Intern Med. 2004;164:659–63.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Irie F, Iso H, Sairenchi T, Fukasawa N, Yamagishi K, Ikehara S, et al. The relationships of proteinuria, serum creatinine, glomerular filtration rate with cardiovascular disease mortality in Japanese general population. Kidney Int. 2006;69:1264–71.
Ninomiya T, Kiyohara Y, Kubo M, Tanizaki Y, Doi Y, Okubo K, et al. Chronic kidney disease and cardiovascular disease in a general Japanese population: the Hisayama Study. Kidney Int. 2005;68:228–36.
Yamamoto R, Kanazawa A, Shimizu T, Hirose T, Tanaka Y, Kawamori R, et al. Association between atherosclerosis and newly classified chronic kidney disease stage for Japanese patients with type 2 diabetes. Diabetes Res Clin Pract. 2009;84:39–45.
Hsieh MC, Tien KJ, Perng DS, Hsiao JY, Chang SJ, Liang HT, et al. Diabetic nephropathy and risk factors for peripheral artery disease in Chinese with type 2 diabetes mellitus. Metabolism. 2009;58:504–9.
Kannel WB, McGee DL. Diabetes and cardiovascular risk factors: the Framingham study. Circulation. 1979;59:8–13.
Booth GL, Kapral MK, Fung K, Tu JV. Relation between age and cardiovascular disease in men and women with diabetes compared with nondiabetic people: a population-based retrospective cohort study. Lancet. 2006;368:29–36.
Adler AI, Stevens RJ, Manley SE, Bilous RW, Cull CA, Holman RR, UKPDS GROUP. Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int. 2003;63:225–32.
Stephenson JM, Kenny S, Stevens LK, Fuller JH, Lee E. Proteinuria and mortality in diabetes: the WHO Multinational Study of Vascular Disease in Diabetes. Diabet Med. 1995;12:149–55.
Pugh JA, Medina R, Ramirez M. Comparison of the course to end-stage renal disease of type 1 (insulin-dependent) and type 2 (non-insulin-dependent) diabetic nephropathy. Diabetologia. 1993;36:1094–8.
Nakagawa N, Takahashi F, Chinda J, Kobayashi M, Hayashi Y, Abe M, et al. A newly estimated glomerular filtration rate is independently associated with arterial stiffness in Japanese patients. Hypertens Res. 2008;31:193–201.
Zoppini G, Targher G, Negri C, Stoico V, Perrone F, Muggeo M, et al. Elevated serum uric acid concentrations independently predict cardiovascular mortality in type 2 diabetic patients. Diabetes Care. 2009;32:1716–20.
Kodama S, Saito K, Yachi Y, Asumi M, Sugawara A, Totsuka K, et al. Association between serum uric acid and development of type 2 diabetes. Diabetes Care. 2009;32:1737–42.
Kosugi T, Nakayama T, Heinig M, Zhang L, Yuzawa Y, Sanchez-Lozada LG, et al. The effect of lowering uric acid on renal disease in the type 2 diabetic db/db mice. Am J Physiol Renal Physiol. 2009;297:F481–8.
Bo S, Cavallo-Perin P, Gentile L, Repetti E, Pagano G. Hypouricemia and hyperuricemia in type 2 diabetes: two different phenotypes. Eur J Clin Invest. 2001;31:318–21.
Madero M, Sarnak MJ, Wang X, Greene T, Beck GJ, Kusek JW, et al. Uric acid and long-term outcomes in CKD. Am J Kidney Dis. 2009;53:796–803.
Verdecchia P, Schillaci G, Brunetti P, Santeusanio F, Porcellati C, Brunetti P. Relation between serum uric acid and risk of cardiovascular disease in essential hypertension. The PIUMA Study. Hypertension. 2003;36:1072–8.
Kang DH, Nakagawa T, Feng L, Watanabe S, Han L, Mazzali M, et al. A role for uric acid in the progression of renal disease. J Am Soc Nephrol. 2002;13:2888–97.
Mazzali M, Kanellis J, Han L, Feng L, Xia YY, Chen Q, et al. Hyperuricemia induces a primary renal arteriolopathy in rats by a blood pressure-independent mechanism. Am J Physiol. 2002;282:F991–7.
Siu YP, Leung KT, Tong MK, Kwan TH. Use of allopurinol in slowing the progression of renal disease through its ability to lower serum uric acid level. Am J Kidney Dis. 2006;47:51–9.
Conflict of interest
The authors have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tanaka, K., Hara, S., Kushiyama, A. et al. Risk of macrovascular disease stratified by stage of chronic kidney disease in type 2 diabetic patients: critical level of the estimated glomerular filtration rate and the significance of hyperuricemia. Clin Exp Nephrol 15, 391–397 (2011). https://doi.org/10.1007/s10157-011-0420-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-011-0420-6