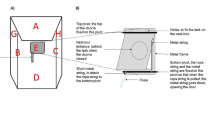
Abstract
Testing the cognitive abilities of cartilaginous fishes is important in understanding the evolutionary origins of cognitive functions in higher vertebrates. We used five South American fresh water stingrays (Potamotrygon castexi) in a learning and problem-solving task. A tube test apparatus was developed to provide a simple but sophisticated procedure for testing cognitive abilities of aquatic animals. All five subjects quickly learned to use water as a tool to extract food from the testing apparatus. The experimental protocol, which gave the animals the opportunity of correcting a wrong visual cue decision, resulted in four out of five subjects correcting an error rather than making an initial right choice. One of five subjects reached 100% correct trials in the visual discrimination task. The ability to use water as an agent to extract food from the testing apparatus is a first indication of tool use in batoid fishes. Performance in the instrumental task of retrieving food from a novel testing apparatus and the rapid learning in the subsequent discrimination/error correction task shows that cartilaginous fish can be used to study the origins of cognitive functions in the vertebrate lineage.




Similar content being viewed by others
References
Agrillo C, Dadda M, Bisazza A (2007) Quantity discrimination in female mosquito fish. Anim Cogn 10:63–70
Agrillo C, Dadda M, Serena G, Bisazza A (2008) Do fish count? Spontaneous discrimination of quantity in female mosquito fish. Anim Cogn 11:495–503
Andrew RJ (2002) The earliest origins and subsequent evolution of lateralization. In: Rogers LJ, Andrews R (eds) Comparative vertebrate lateralization. Cambridge University Press, Cambridge, pp 70–93
Aronson LR, Aronson FR, Clark E (1967) Instrumental conditioning and light- dark discrimination in young nurse sharks. Bull Mar Sci 17:249–256
Beck BB (1980) Animal tool use. Garland STPM Press, New York
Bergen Y, Laland KN, Hoppit W (2004) Social learing, innovation, and intelligence in fish. In: Rogers LJ, Kaplan G (eds) Comparative vertebrate cognition: are primates superior to non-primates? Kluwer Academic, Plenum, pp 141–168
Bisazza A, Rogers LJ, Vallortigara G (1998) The origins of cerebral asymmetry: a review of evidence of behavioural and brain lateralization in fishes, reptiles and amphibians. Neuro Behav Rev 22:411–426
Bitterman ME (1965) Phyletic differences in learning. Am Psychol 20:396–410
Bitterman ME, Mackintosh NJ (1969) Reversal and probability learning: comments. In: Gilbert RM, Sutherland NS (eds) Animal discrimination learning. Academic Press, London, pp 163–185
Brown C, Laland KN (2003) Social learning in fishes: a review. Fish Fish 4:280–288
Brown C, Laland K, Krause J (2006) Fish cognition and behavior. Blackwell Publishing, Oxford
Bshary R, Wickler W, Fricke H (2002) Fish cognition: a primate’s eye view. Anim Cogn 5:1–13
Bshary R, Grutter AS, Willener AST, Leimar O (2008) Pairs of cooperating cleaner fish provide better service quality than singletons. Nature 455:965–966
Burghardt GM (1977) Learning processes in reptiles. In: Gans C, Tinkle D (eds) The biology of the reptilia, vol 7. Academic Press, New York, pp 555–681
Burghardt GM (2005) The genesis of animal play: testing the limits. MIT Press, Cambridge
Butler AB, Hodos W (2005) Comparative vertebrate neuroanatomy: evolution and adaptation. Wiley-Liss, Hoboken
Carrier JC, Musick JA, Heithaus MR (2004) Biology of sharks and their relatives. CRC Press, Boca Raton
Clark E (1959) Instrumental conditioning in lemon sharks. Science 130:217–218
Collin SP, Whitehead D (2004) The functional roles of passive electroreception in non-electric fishes. Anim Biol 54:1–25
Fellows BJ (1967) Chance stimulus sequences for discrimination tasks. Psychol Bull 67:87–92
Gleitman H, Rozin P (1971) Learning and memory. In: Hoar WS, Randall DJ (eds) Fish physiology, vol 6. Academic Press, New York, pp 191–278
Graeber RC (1978) Behavioral studies correlated with central nervous system integration of vision in sharks. In: Hodgson ES, Mathewson RF (eds) Sensory biology of sharks, skates and rays. Office of Naval Research, Arlington, pp 195–225
Grogan ED, Lund R (2004) The origin and relationships of early chondrichthyes. In: Carrier JC, Musick JA, Heithaus MR (eds) Biology of sharks and their relatives. CRC Press, Boca Raton
Hamlett WC (2005) Reproductive biology and phylogeny of Chondrichthyes: sharks, batoids, and chimaeras. Science Publishers Inc, Enfield
Hodgson ES, Mathewson RF (1978) Sensory biology of sharks, skates and rays. Office of Naval Research, Arlington
Horner V, Whiten A (2007) Learning from others’ mistakes? Limits on understanding a trap- tube task by young chimpanzees (Pan troglodytes) and children (Homo sapiens). J Comp Psychol 121:12–21
Hueter RE, Mann DA, Maruska KP, Sisneros JA, Demski LS (2004) Sensory biology of elasmobranchs. In: Carrier JC, Musick JA, Heithaus MR (eds) Biology of sharks and their relatives. CRC Press, Boca Raton
Jerison HJ (1970) Gross brain indices and the analysis of fossil endocasts. In: Noback CR, Montagna W (eds) Advances in primatology, vol 1. Appleton-Century Crofts, New York
Jerison HJ (1973) Evolution of the brain and intelligence. Academic Press, New York
Keifer JD, Colgan PW (1992) The role of learning in fish behavior. Rev Fish Biol Fish 2:125–143
Klimley PA (2003) The secret life of sharks: a leading marine biologist reveals the mysteries of shark behavior. Simon and Schuster, New York
Laland KN, Brown C, Krause J (2003) Learning in fishes: from three-seconds memory to culture. Fish Fish 4:192–202
Mackintosh NJ (1969) Comparative studies of reversal and probability learning: rats, birds and fish. In: Gilbert RM, Sutherland NS (eds) Animal discrimination learning. Academic Press, London, pp 137–162
Malashichev YB (2006) One-sided limb preference is linked to alternating-limb locomotion in anuran amphibians. J Comp Psychol 120:401–410
New JG (2001) Comparative neurobiology of the elasmobranch cerebellum: theme and variation on a sensorimotor interface. Environ Biol Fish 60:93–108
Northcutt RG (1977) Elasmobranch central nervous system organization and its possible evolutionary significance. Am J Zool 17:411–429
Northcutt R (1978) Brain organization in the cartilaginous fishes. In: Hodgson ES, Mathewson RF (eds) Sensory biology of sharks, skates and rays. Office of Naval Research, Arlington, pp 117–193
Northcutt RG (1989) Brain variation and phylogenetic trends in elasmobranch fishes. J Exp Zool 2:83–100
Reaux R, Povinelli DJ (2000) The trap-tube problem. In: Povinelli DJ (ed) Folk physics for apes: a chimpanzee’s theory of how the world works. Oxford University Press, Oxford, pp 108–131
Rodrriguez F (2006) Neural mechanisms of learning in teleost fish. In: Brown C, Laland K, Krause J (eds) Fish cognition and behavior. Blackwell Publishing, Oxford, pp 243–277
Rogers LJ (2002) Lateralised brain function in anurans: comparison to lateralisation in other vertebrates. Laterality 7(3):219–293
Rogers LJ, Andrew R (2002) Comparative vertebrate lateralization. Cambridge University Press, Cambridge
Schluessel V, Bleckmann H (2005) Spatial memory and orientation strategies in the elasmobranch Potamotrygon motoro. J Comp Physiol A 191:695–706
Schuster S, Rossel S, Schmidtmann A, Jäger I, Poralla J (2004) Archer fish learn to compensate for complex optical distortions to determine the absolute size of their aerial prey. Curr Biol 14:1565–1568
Thorpe WH (1963) Learning and instinct in animals. Harvard University Press, Cambridge
Visalberghi E, Limongelli L (1994) Lack of Comprehension of cause-effect relationships in capuchin monkeys (Cebus apella). J Comp Psychol 108:15–22
Visalberghi E, Fragaszy DM, Savage-Rumbaugh S (1995) Comprehension of causal relations in a tool-using task by chimpanzees (Pan troglodytes), bonobos (Pan paniscus), orang utans (Pongo pygmaeus), and capuchins (Cebus apella). J Comp Psychol 109:52–60
Wodinsky J, Bitterman ME (1957) Discrimination-reversal in the fish. Am J Psychol 70:569–576
Yarom Y, Cohen D (2002) The olivocerebellar system as a generator of temporal patterns. Ann N Y Acad Sci 978:122–134
Yerkes RM (1943) Chimpanzees: a laboratory colony. Yale University Press, New Haven
Acknowledgments
The authors of this study want to thank the zoo of Vienna for providing the animals. We would especially like to thank E Wolf, A Weissenbacher and R Halbbauer at the zoo of Vienna for their help and cooperation during this study. We want to express our special gratitude to E Sonntag of the Detroit zoo who used food containing plastic pipes as a behavioral enrichment for the stingrays housed at the Detroit zoo. This example gave rise to our idea to create a pipe-like testing apparatus.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
This file is unfortunately not in the Publisher's archive anymore: Supplementary material 1 (DIVX 2136 kb). Animal 2 choosing the correct side of the tasting apparatus. The correct side to extract the food is marked by white tape
Rights and permissions
About this article
Cite this article
Kuba, M.J., Byrne, R.A. & Burghardt, G.M. A new method for studying problem solving and tool use in stingrays (Potamotrygon castexi). Anim Cogn 13, 507–513 (2010). https://doi.org/10.1007/s10071-009-0301-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-009-0301-5