Abstract
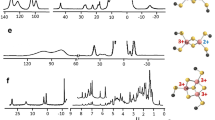
Mitochondria from respiring cells were isolated under anaerobic conditions. Microscopic images were largely devoid of contaminants, and samples consumed O2 in an NADH-dependent manner. Protein and metal concentrations of packed mitochondria were determined, as was the percentage of external void volume. Samples were similarly packed into electron paramagnetic resonance tubes, either in the as-isolated state or after exposure to various reagents. Analyses revealed two signals originating from species that could be removed by chelation, including rhombic Fe3+ (g = 4.3) and aqueous Mn2+ ions (g = 2.00 with Mn-based hyperfine). Three S = 5/2 signals from Fe3+ hemes were observed, probably arising from cytochrome c peroxidase and the a3:Cub site of cytochrome c oxidase. Three Fe/S-based signals were observed, with averaged g values of 1.94, 1.90 and 2.01. These probably arise, respectively, from the [Fe2S2]+ cluster of succinate dehydrogenase, the [Fe2S2]+ cluster of the Rieske protein of cytochrome bc 1, and the [Fe3S4]+ cluster of aconitase, homoaconitase or succinate dehydrogenase. Also observed was a low-intensity isotropic g = 2.00 signal arising from organic-based radicals, and a broad signal with g ave = 2.02. Mössbauer spectra of intact mitochondria were dominated by signals from Fe4S4 clusters (60–85% of Fe). The major feature in as-isolated samples, and in samples treated with ethylenebis(oxyethylenenitrilo)tetraacetic acid, dithionite or O2, was a quadrupole doublet with ΔE Q = 1.15 mm/s and δ = 0.45 mm/s, assigned to [Fe4S4]2+ clusters. Substantial high-spin non-heme Fe2+ (up to 20%) and Fe3+ (up to 15%) species were observed. The distribution of Fe was qualitatively similar to that suggested by the mitochondrial proteome.









Similar content being viewed by others
Notes
For a purified Fe4S4 ferredoxin the area under the doublet can be quantified to within 1–2%. Here, the uncertainties are considerably larger, primarily because more than one cluster contributes. The primary contributors to the doublet may be aconitase and dihydroxyacid dehydratase. Because species with slightly different but unresolved parameters contribute, lineshapes are heterogeneously broadened Lorentzians. We used both the Lorentzian and the Voight lineshape options of WMOSS. As Voight shapes are narrower at the base, this option yields, upon visual inspection, a lower estimate for the concentration.
In weak applied fields, the lowest three Kramers doublets of the spin sextet are generally populated at 4.2 K, yielding three Mössbauer spectra per site. Moreover, under these conditions the magnetic splittings, like the effective g values observed by EPR, are very sensitive to the rhombicity parameter E/D. Consequently, the high-spin Fe3+ ions in our sample produce broad and barely discernible features in weak fields. However, the 8.0-T spectra are fairly insensitive to D and E/D, because the large Zeeman splitting puts essentially all Fe3+ ions into the M S = −5/2 state, facilitating detection and quantification.
Abbreviations
- CoQ:
-
Coenzyme Q
- DTT:
-
Dithiothreitol
- EDTA:
-
Ethylenediaminetetraacetic acid
- EGTA:
-
Ethylenebis(oxyethylenenitrilo)tetraacetic acid
- EPR:
-
Electron paramagnetic resonance
- ETF:
-
Electron transfer flavoprotein
- HEPES:
-
N-(2-Hydroxyethyl)piperazine-N′-ethanesulfonic acid
- IM:
-
Inner membrane
- IMS:
-
Intermembrane space
- NHE:
-
Normal hydrogen electrode
- OM:
-
Outer membrane
- SH buffer:
-
0.6 M sorbitol/20 mM N-(2-hydroxyethyl)piperazine-N′-ethanesulfonic acid buffer pH 7.4
- SP buffer:
-
1.2 M sorbitol/20 mM potassium phosphate buffer pH 7.4
References
Lemire BD, Oyedotun KS (2002) Biochim Biophys Acta Bioenerg 1553:102–116
Beinert H (2002) Biochim Biophys Acta Bioenerg 1553:7–22
Maguire JJ, Johnson MK, Morningstar JE, Ackrell BAC, Kearney EB (1985) J Biol Chem 260:10909–10912
Fee JA, Findling KL, Yoshida T, Hille R, Tarr GE, Hearshen DO, Dunham WR, Day EP, Kent TA, Münck E (1984) J Biol Chem 259:124–133
Emptage MH, Kent TA, Kennedy MC, Beinert H, Munck E (1983) Proc Natl Acad Sci USA 80:4674–4678
Wallace MA, Liou LL, Martins J, Clement MHS, Bailey S, Longo VD, Valentine JS, Gralla EB (2004) J Biol Chem 279:32055–32062
Barros MH, Nobrega FG (1999) Gene 233:197–203
Schiffler B, Bureik M, Reinle W, Muller EC, Hannemann F, Bernhardt R (2004) J Inorg Biochem 98:1229–1237
Xia B, Cheng H, Bandarian V, Reed GH, Markley JL (1996) Biochemistry 35:9488–9495
Mitou G, Higgins C, Wittung-Stafshede P, Conover RC, Smith AD, Johnson MK, Gaillard J, Stubna A, Münck E, Meyer J (2003) Biochemistry 42:1354–1364
Zhang S, Sanyal I, Bulboaca GH, Rich A, Flint DH (1994) Arch Biochem Biophys 309:29–35
Berkovitch F, Nicolet Y, Wan JT, Jarrett JT, Drennan CL (2004) Science 303:76–79
Cosper MM, Jameson GNL, Eidsness MK, Huynh BH, Johnson MK (2002) FEBS Lett 529:332–336
Jameson GN, Cosper MM, Hernandez HL, Johnson MK, Huynh BH (2004) Biochemistry 43:2022–2031
Hewitson KS, Ollagnier-de Choudens S, Sanakis Y, Shaw NM, Baldwin JE, Münck E, Roach PL, Fontecave M (2002) J Biol Inorg Chem 7:83–93
Kriek M, Peters L, Takahashi Y, Roach PL (2002) Protein Expr Purif 28:241–245
Miller JR, Busby RW, Jordan SW, Cheek J, Henshaw TF, Ashley GW, Broderick JB, Cronan JE, Marletta MA (2000) Biochemistry 39:15166–15178
Ollagnier-de Choudens S, Sanakis Y, Hewitson KS, Roach P, Baldwin JE, Münck E, Fontecave M (2000) Biochemistry 39:4165–4173
Velasco JA, Cansado J, Pena MC, Kawakami T, Laborda J (1993) Gene 137:179–185
Flint DH, Emptage MH, Finnegan MG, Fu WG, Johnson MK (1993) J Biol Chem 268:14732–14742
Mühlenhoff U, Gerber J, Richhardt N, Lill R (2003) EMBO J 22:4815–4825
Schilke B, Voisine C, Beinert H, Craig E (1999) Proc Natl Acad Sci USA 96:10206–10211
Jensen LT, Culotta VC (2000) Mol Cell Biol 20:3918–3927
Tong WH, Jameson GNL, Huynh BH, Rouault TA (2003) Proc Natl Acad Sci USA 100:9762–9767
Ruzicka FJ, Beinert H (1975) Biochem Biophys Res Comm 66:622–631
Ruzicka FJ, Beinert H (1975) J Biol Chem 252:8440–8445
Paulsen KE, Orville AM, Frerman FE, Lipscomb JD, Stankovich MT (1992) Biochemistry 31:11755–11761
Hunte C, Koepke J, Lange C, Rossmanith T, Michel H (2000) Structure 8:669–684
Bonagura CA, Bhaskar B, Shimizu H, Li HY, Sundaramoorthy M, McRee DE, Goodin DB, Poulos TL (2003) Biochemistry 42:5600–5608
Mowat CG, Miles CS, Munro AW, Cheesman MR, Quaroni LG, Reid GA, Chapman SK (2000) J Biol Inorg Chem 5:584–592
Kojima N, Palmer G (1983) J Biol Chem 258:4908–4913
Brautigan DL, Feinberg BA, Hoffman BM, Margoliash E, Peisach J, Bumberg WE (1977) J Biol Chem 252:574–582
Barros MH, Carlson CG, Glerum DM, Tzagoloff A (2001) FEBS Lett 492:133–138
Hederstedt L, Lewin A, Throne-Holst M (2005) J Bacteriol 187:8361–8369
Glerum DM, Muroff I, Jin C, Tzagoloff A (1997) J Biol Chem 272:19088–19094
Bureik M, Schiffler B, Hiraoka Y, Vogel F, Bernhardt R (2002) Biochemistry 41:2311–2321
Petrova VY, Drescher D, Kujumdzieva AV, Schmitt MJ (2004) Biochem J 380:393–400
Ding H, Harrison K, Lu J (2005) J Biol Chem 280:30432–30437
Lesuisse E, Santos R, Matzanke BF, Knight SAB, Camadro JM, Dancis A (2003) Hum Mol Genet 12:879–889
Sellers VM, Wu CK, Dailey TA, Dailey HA (2001) Biochemistry 40:9821–9827
Ferreira GC, Franco R, Mangravita A, George GN (2002) Biochemistry 41:4809–4818
Wu CK, Dailey HA, Rose JP, Burden A, Sellers VM, Wang BC (2001) Nat Struct Biol 8:156–160
Foury F, Roganti T (2002) J Biol Chem 277:24475–24483
Li LT, Kaplan J (1997) J Biol Chem 272:28485–28493
Moraes CT, Diaz F, Barrientos A (2004) Biochim Biophys Acta Bioenerg 1659:153–159
Dumont ME, Cardillo TS, Hayes MK, Sherman F (1991) Mol Cell Biol 11:5487–5496
Chloupkova M, LeBard LS, Koeller DM (2003) J Mol Biol 331:155–165
Lill R, Kispal G (2001) Res Microbiol 152:331–340
Lange H, Lisowsky T, Gerber J, Mühlenhoff U, Kispal G, Lill R (2001) EMBO Rep 2:715–720
Stenmark P, Grünler J, Mattsson J, Sindelar PJ, Nordlund P, Berthold DA (2001) J Biol Chem 276:33297–33300
Berthold DA, Voevodskaya N, Stenmark P, Gräslund A, Nordlund P (2002) J Biol Chem 277:43608–43614
Neese F, Zumft WG, Antholine WE, Kroneck PMH (1996) J Am Chem Soc 118:8692–8699
Barros MH, Johnson A, Tzagoloff A (2004) J Biol Chem 279:49883–49888
Palumaa P, Kangur L, Voronova A, Sillard R (2004) Biochem J 382:307–314
Hiser L, Di Valentin M, Hamer AG, Hosler JP (2000) J Biol Chem 275:619–623
Sturtz LA, Diekert K, Jensen LT, Lill R, Culotta VC (2001) J Biol Chem 276:38084–38089
Cobine PA, Ojeda LD, Rigby KM, Winge DR (2004) J Biol Chem 279:14447–14455
Luk E, Jensen LT, Culotta VC (2003) J Biol Inorg Chem 8:803–809
Hirabaya T, Harada T (1971) Biochem Biophys Res Commun 45:1369
Sharp RE, White P, Chapman SK, Reid GA (1994) Biochemistry 33:5115–5120
Arscott LD, Gromer S, Schirmer RH, Becker K, Williams CH (1997) Proc Natl Acad Sci USA 94:3621–3626
Staples CR, Ameyibor E, Fu WG, Gardet-Salvi L, Stritt-Etter EL, Knaff DB, Johnson MK (1996) Biochemistry 35:11425–11434
Garrib A, McMurray WC (1986) J Biol Chem 261:8042–8048
Huh WK, Kim ST, Yang KS, Seok YJ, Hah YC, Kang SO (1994) Eur J Biochem 225:1073–1079
Joo HS, Kim SS (1998) J Biochem Mol Biol 31:37–43
Shan XY, Wang LQ, Hoffmaster R, Kruger WD (1999) J Biol Chem 274:32613–32618
Robinson KM, Lemire BD (1996) J Biol Chem 271:4061–4067
Gin P, Hsu AY, Rothman SC, Jonassen T, Lee PT, Tzagoloff A, Clarke CF (2003) J Biol Chem 278:25308–25316
Oxelmark E, Marchini A, Malanchi I, Magherini F, Jaquet L, Hajibagheri MAN, Blight KJ, Jauniaux JC, Tommasino M (2000) Mol Cell Biol 20:7784–7797
Sands RH, Beinert H (1960) Biochem Biophys Res Commun 3:47–52
Hartzell CR, Beinert H (1974) Biochim Biophys Acta 368:318–338
Bulteau AL, Ikeda-Saito M, Szweda LI (2003) Biochemistry 42:14846–14855
Lin CIP, Ohnishi T, Clejan L, Beattie DS (1983) Eur J Biochem 137:179–183
Phillips JD, Graham LA, Trumpower BL (1993) J Biol Chem 268:11727–11736
Merbitz-Zahradnik T, Zwicker K, Nett JH, Link TA, Trumpower BL (2003) Biochemistry 42:13637–13645
Shergill JK, Cammack R (1994) Biochim Biophys Acta 1185:43–49
Shergill JK, Cammack R, Chen JH, Fisher MJ, Madden S, Rees HH (1995) Biochem J 307:719–728
Albract SPJ, Subramanian J (1977) Biochim Biophys Acta 462:36–48
Gavin CE, Gunter KK, Gunter TE (1999) Neurotoxicology 20:445–453
Guldutuna S, Zimmer G, Leuschner M, Bhatti S, Elze A, Beisinger B, Hofmann M, Leuschner U (1999) Biochim Biophys Acta 1453:396–406
Tuckey RC, McKinley AJ, Headlam MJ (2001) Eur J Biochem 268:2338–2343
Albracht SPJ, Leeuwerik FJ, VanSwol B (1979) FEBS Lett 104:197–200
Albracht SPJ (1980) Biochim Biophys Acta 612:11–28
Albracht SPJ, VanVerseveld HW, Hagen WR, Kalkman ML (1980) Biochim Biophys Acta 593:173–186
Diekert K, de Kroon AIPM, Kispal G, Lill R (2000) In: Pon LA, Schon EA (eds) Methods in yeast genetics, vol 65. Academic, San Diego
Cleland WW (1964) Biochemistry 3:480
Barondeau DP, Roberts LM, Lindahl PA (1994) J Am Chem Soc 116:3442–3448
Wright R (2000) Microsc Res Tech 51:496–510
Pelley JW, Garner CW, Little GH (1978) Anal Biochem 86:341–343
Toyoshima S, Wantanabe F, Saido H, Miyatake K, Nakano Y (1995) J Nutr 125:2846–2850
Sapan CV, Lundlad RL, Price NC (1999) Biotechnol Appl Biochem 29:99–108
Dineley KE, Richards LL, Votyakova TV, Reynolds IJ (2005) Mitochondria 5:55–65
Lund P, Wiggins D (1990) Biochim Biophys Acta 1018:98–102
Hackenbrock CR (1968) J Cell Biol 37:345–369
Zischka H, Weber G, Weber PJA, Posch A, Braun RJ, Bühringer D, Schneider U, Nissum M, Meitinger T, Ueffing M, Eckerskorn C (2003) Proteomics 3:906–916
Shaw JM, Nunnari J (2002) Trends Cell Biol 12:178–184
Egner A, Jakobs S, Hell SW (2002) Proc Natl Acad Sci USA 99:3370–3375
Polcic P, Sabova L, Kolarov J (1997) FEBS Lett 412:207–210
Soubannier V, Vaillier J, Paumard P. Coulary B, Schaeffer J, Velours J (2002) J Biol Chem 277:10739–10745
Boyle GM, Roucou X, Nagley P, Devenish RJ, Prescott M (1999) Eur J Biochem 262:315–323
Wickman HH, Klein MP, Shirley DA (1964) Phys Rev 152:345–357
Mayhew SG (1978) Eur J Biochem 85:535–547
Lindahl PA, Day EP, Kent TA, Orme-Johnson WH, Münck E (1985) J Biol Chem 260:1160–1173
Andrew CR, Kemper LJ, Busche TL, Tiwari AM, Kecskes MC, Stafford JM, Croft LC, Lu S, Moenne-Loccoz P, Huston W, Moir JWB, Eady RR (2005) Biochemistry 44:8664–8672
Miyoshi H, Tokutake N, Imaeda Y, Akagi T, Iwamura H (1995) Biochim Biophys Acta 1229:149–154
Babcock M, deSilva D, Oaks R, DavisKaplan S, Jiralerspong S, Montermini L, Pandolfo M, Kaplan J (1997) Science 276:1709–1712
Foury F, Cazzalini O (1997) FEBS Lett 411:373–377
Tangeras A, Flatmark T, Backstrom D, Ehrenberg A (1980) Biochim Biophys Acta 589:162–175
Kispal G, Csere P, Prohl C, Lill R (1999) EMBO J 18:3981–3989
Petrat F, de Groot H, Rauen U (2001) Biochem J 356:61–69
Petrat F, Weisheit D, Lensen M, de Groot H, Sustmann R, Rauen U (2002) Biochem J 362:137–147
Sturm B, Bistrich U, Schranzhofer M, Sarsero JP, Rauen U, Scheiber-Mojdehkar B, de Groot H, Ioannou P, Petrat F (2005) J Biol Chem 280:6701–6708
Lange H, Kispal G, Lill R (1999) J Biol Chem 274:18989–18996
Agar JN, Krebs C, Frazzon J, Huynh BH, Dean DR, Johnson MK (2000) Biochemistry 39:7856–7862
Franco R, Moura JJG, Moura I, Huynh BH, Forbes WS, Ferreira GC (1995) J Biol Chem 270:26352–26357
Haile DJ, Rouault TA, Harford JB, Kennedy MC, Blondin GA, Beinert H, Klausner RD (1992) Proc Natl Acad Sci USA 89:11735–11739
Henze K, Martin W (2003) Nature 426:127–128
Mühlenhoff U, Lill R (2000) Biochim Biophys Acta Bioenerg 1459:370–382
Kilpatrick LK, Kennedy MC, Beinert H, Czernuszewicz RC, Qiu D, Spiro TG (1994) J Am Chem Soc 116:4053–4061
Jackson TA, Karapetian A, Miller AF, Brunold TC (2004) J Am Chem Soc 126:12477–12491
Un S, Tabares LC, Cortez N, Hiraoka BY, Yamakura F (2004) J Am Chem Soc 126:2720–2726
Srinivasan C, Liba A, Imlay JA, Valentine JS, Gralla EB (2000) J Biol Chem 275:29187–29192
Werth MT, Sices H, Cecchini G, Schroder I, Lasage S, Gunsalus RP, Johnson MK (1992) FEBS Lett 299:1–4
Hirst J, Sucheta A, Ackrell BAC, Armstrong FA (1996) J Am Chem Soc 118:5031–5038
Sucheta A, Ackrell BAC, Cochran B, Armstrong FA (1992) Nature 356:361–362
Link TA, Hagen WR, Pierik AJ, Assmann C, von Jagow G (1992) Eur J Biochem 208:685–691
Beinert H, Shaw RW (1977) Biochim Biophys Acta 462:121–130
Aasa R, Albracht SPJ, Falk KE, Lanne B, Vanngard T (1976) Biochim Biophys Acta 422:260–272
Gorbikova EA, Vuorilehto K, Wikstrom M, Verkhovsky MI (2006) Biochemistry 45:5641–5649
Nicholls P, Elliott WB (1974) In: Worwood M, Jacobs A (eds) Iron in biochemistry and medicine. Academic, New York
Wittenberg BA, Kampa L, Wittenberg JB, Blumberg WE, Peisach J (1968) J Biol Chem 243:1863–1870
Coulson AFW, Erman JE, Yonetani T (1971) J Biol Chem 246:917–924
Goodin DB, McRee DE (1993) Biochemistry 32:3313–3324
Bellei M, Jakopitsch C, Battistuzzi G, Sola M, Obinger C (2006) Biochemistry 45:4768–4774
Cassanova N, O’Brien KM, Stahl BT, McClure T, Poyton RO (2005) J Biol Chem 280:7645–7653
Reynolds MF, Shelver D, Kerby RL, Parks RB, Roberts GP, Burstyn JN (1998) J Am Chem Soc 120:9080–9081
Lett CM, Guillemette JG (2002) Biochem J 362:281–287
Yu L, Dong J-H, Yu C-A (1986) Biochim Biophys Acta Bioenerg 852:203–211
Tegoni M, Silvestrini MC, Guigliarelli B, Asso M, Brunori M, Bertrand P (1998) Biochemistry 37:12761–12771
Anemüller S, Bill E, Schäfer G, Trautwein A, Teixeira M (1992) Eur J Biochem 210:133–138
Lester RL, Crane FL (1959) J Biol Chem 234:2169–2175
Waldeck AR, Stowell MHB, Lee HK, Hung SC, Matsson M, Hederstedt L, Ackrell BAC, Chan SI (1997) J Biol Chem 272:19373–19382
Lukoyanov D, Berry SM, Lu Y, Antholine WE, Scholes CP (2002) Biophys J 82:2758–2766
Gamelin DR, Randall DW, Hay MT, Houser RP, Mulder TC, Canters GW, de Vries S, Tolman WB, Lu Y, Solomon EI (1998) J Am Chem Soc 120:5246–5263
Nittis T, George GN, Winge DR (2001) J Biol Chem 276:42520–42526
Heaton DN, George GN, Garrison G, Winge DR (2001) Biochemistry 40:743–751
Ludovico P, Sansonetty F, Corte-Real M (2001) Microbiology 147:3335–3343
Sickmann A, Reinders J, Wagner Y, Joppich C, Zahedi R, Meyer HE, Schonfisch B, Perschil I, Chacinska A, Guiard B et al (2003) Proc Natl Acad Sci USA 100:13207–13212
Huh W-K, Falvo JV, Gerke LC, Carroll AS, Howson RW, Weissman JS, O’Shea EK (2003) Nature 425:686–691
Prokisch H, Scharfe C, Camp DG, Xiao WZ, David L, Andreoli C, Monroe ME, Moore RJ, Gritsenko MA, Kozany C, Hixson KK, Mottaz HM, Zischka H, Ueffing M, Herman ZS, Davis RW, Meitinger T, Oefner PJ, Smith RD, Steinmetz LM (2004) PLoS Biol 2:795–804
Forster J, Famili I, Fu P, Palsson BO, Nielsen J (2003) Genome Res 13(2):244–253
Ohlmeier S, Kastaniotis AJ, Hiltunen JK, Bergmann U (2004) J Biol Chem 6:3956–3979
Ghaemmaghami S, Huh WK, Bower K, Howson RW, Belle A, Dephoure N, O’Shea EK, Weissman JS (2003) Nature 425:737–741
Biswas SK, Yamaguchi M, Naoe N, Takashima T, Takeo K (2003) J Electron Microsc 52:133–143
Trumpower B (1990) J Biol Chem 265:11409–11412
Murphy MEP, Nall BT, Brayer GD (1992) J Mol Biol 227:160–176
Daum G et al (1982) J Biol Chem 257:13028–13033
Goodin DB, Davidson MG, Roe JA, Mauk AG, Smith M (1991) Biochemistry 30:4953–4962
Capeillereblandin G, Bray RC, Iwatsubo M, Labeyrie F (1975) Eur J Biochem 54:549–566
Ebert CE, Ghosh M, Wang YD, Beattie DS (2003) Biochim Biophys Acta 1607:65–78
Orme-Johnson NR, Hansen RE, Beinert H (1971) Biochem Biophys Res Commun 45:871
Salerno JC (1984) J Biol Chem 259:2331–2336
Svensson-Ek M, Abramson J, Larsson G, Tornroth S, Brzezinski P, Iwata S (2002) J Mol Biol 321(2):329–339
Babcock GT, Vickery LE, Palmer G (1978) J Biol Chem 253:2400–2411
Cheesman MR, Oganesyan VS, Watmough NJ, Butler CS, Thomson AJ (2004) J Am Chem Soc 126:4157–4166
Beinert H, Ackrell BAC, Kearney EB, Singer TP (1974) Biochem Biophys Res Commun 58:564–572
Peterson J, Vibat C, Gennis RB (1994) FEBS Lett 355:155–156
Svensson B, Andersson KK, Hederstedt L (1996) Eur J Biochem 238:287–295
Jonassen T, Proft M, Randez-Gil F, Schultz JR, Marbois BN, Entian KD, Clarke CF (1998) J Biol Chem 273:3351–3357
Rea S (2001) FEBS Lett 509:389–394
Liu KE, Lippard SJ (1991) J Biol Chem 266:12836–12839
Fox BG, Hendrich MP, Surerus KK, Anderson KK, Froland WA, Lipscomb JD, Münck E (1993) J Am Chem Soc 115:3688–3701
Regev-Rudzki N, Karniely S, Ben-Haim NN, Pines O (2005) Mol Biol Cell 16:4163–4171
Tong JJ, Feinberg BA (1994) J Biol Chem 269:24920–24927
Smith AD, Jameson GNL, Dos Santos PC, Agar JN, Naik S, Krebs C, Frazzon J, Dean DR, Huynh BH, Johnson MK (2005) Biochemistry 44:12955–12969
Pelzer W, Muhlenhoff U, Diekert K, Siegmund K, Kispal G, Lill R (2000) FEBS Lett 476:134–139
Picciocchi A, Douce R, Alban C (2003) J Biol Chem 278:24966–24975
Ugulava NB, Gibney BR, Jarrett JT (2001) Biochemistry 40:8343–8351
Duin EC, Lafferty ME, Crouse BR, Allen RM, Sanyal I, Flint DH, Johnson MK (1997) Biochemistry 36:11811–11820
Hoja U, Marthol S, Hofmann J, Stegner S, Schulz R, Meier S, Greiner E, Schweizer E (2004) J Biol Chem 279:21779–21786
Ollagnier-de Choudens S, Fontecave M (1999) FEBS Lett 453:25–28
Cosper MM, Jameson GNL, Hernandez HL, Krebs C, Huynh BH, Johnson MK (2004) Biochemistry 43:2007–2021
Branda SS, Cavadini P, Adamec J, Kalousek F, Taroni F, Isaya G (1999) J Biol Chem 274:22763–22769
He YN, Alam SL, Proteasa SV, Zhang Y, Lesuisse E, Dancis A, Stemmler TL (2004) Biochemistry 43:16254–16262
Bou-Abdallah F, Adinolfi S, Pastore A, Laue TM, Chasteen ND (2004) J Mol Biol 341:605–615
Dawson JH, Bracete AM, Huff AM, Kadkhodayan S, Zeitler CM, Sono M, Chang CK, Loewen PC (1991) FEBS Lett 295:123–126
Ioannidis N, Cooper CE, Poole RK (1992) Biochem J 288:649–655
Macheroux P, Hill S, Austin S, Eydmann T, Jones T, Kim SO, Poole R, Dixon R (1998) Biochem J 332:413–419
Acknowledgements
We thank the following people: Art Johnson and Holly Cargill (Department of Biochemistry and Biophysics, Texas A&M University) for instructions on isolating mitochondria; Rola Barhoumi (Image Analysis Laboratory, Texas A&M University) and Anne Ellis (Microscopy and Imaging Center, Texas A&M University) for collecting microscopic images; Jinny Johnson (Protein Chemistry Laboratory, Texas A&M University) for performing amino acid analyses; David P. Giedroc (Department of Biochemistry and Biophysics, Texas A&M University) for access to his atomic absorption spectrophotometer; William James (Department of Chemistry, Texas A&M University) for training on and assistance with the inductively coupled plasma mass spectrometer; Shelly Henderson Possi for help in isolating some batches and in measuring O2 consumption; Tanner Freeman for preparing one of the EPR samples; and Roland Lill for helpful discussion.
This study was supported by the Robert A. Welch Foundation (A1170) and The National Institutes of Health [GM077387 (M.P.H.), EB001475 (E.M.) and The Chemistry Biology Interface training program (B.N.H. and J.G)].
Author information
Authors and Affiliations
Corresponding author
Additional information
Note added in proof: It now appears less likely that Isa1p and Isa2p contain Fe/S clusters similar to those in other Fe/S scaffold proteins.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hudder, B.N., Morales, J.G., Stubna, A. et al. Electron paramagnetic resonance and Mössbauer spectroscopy of intact mitochondria from respiring Saccharomyces cerevisiae . J Biol Inorg Chem 12, 1029–1053 (2007). https://doi.org/10.1007/s00775-007-0275-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-007-0275-1