Summary
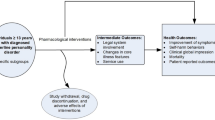
Twenty subjects with binge eating disorder were randomly assigned to flexible-dose fluvoxamine or placebo for 12 weeks. A significant reduction in binge frequency, Beck Depression Inventory scores and the eating concern, shape concern and weight concern subscales of the Eating Disorder Examination were noted for both fluvoxamine (n = 9) and placebo (n = 11) groups. There were no significant differences between fluvoxamine and placebo for any treatment outcome variables. The findings from this small trial contribute to the inconsistent results of antidepressant studies in binge eating disorder.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received August 7, 2002; accepted January 26, 2003 Published online April 23, 2003
RID="*"
ID="*" This study was presented as a poster at the 9th International Conference on Eating Disorders, May, 2000, New York, NY. Supported in part by a grant from Solvay Pharmaceuticals.
Correspondence: Teri Pearlstein, M.D., Assistant Professor, Department of Psychiatry and Human Behavior, Brown Medical School, Director, Women's Behavioral Health Program, Women and Infants Hospital, 101 Dudley Street, Providence, RI 02905, U.S.A.; e-mail: Teri_Pearlstein@brown.edu
Rights and permissions
About this article
Cite this article
Pearlstein, T., Spurell, E., Hohlstein, L. et al. A double-blind, placebo-controlled trial of fluvoxamine in binge eating disorder: a high placebo response. Arch Womens Ment Health 6, 147–151 (2003). https://doi.org/10.1007/s00737-003-0172-8
Issue Date:
DOI: https://doi.org/10.1007/s00737-003-0172-8