Summary.
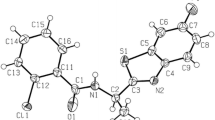
This work describes a two-step, one-pot synthetic method for the formal aza-[3 + 3] cycloaddition between N-alkyl substituted enaminones and benzoyl isothiocyanate, which afforded 4-thioxopyrimidines in reasonable yields. Reaction of acyclic enaminone with a sterically hindered group attached to the nitrogen atom afforded pyridine-2-thione, yet in low yield. The antibacterial, antifungal, and trypanocidal activities of the thioxopyrimidines were evaluated and five compounds exhibited moderate activity against Candida albicans, Micrococcus luteus, and Trypanosoma cruzi. The solid state structures of a thioxopyrimidine, an organic disulfide, and a 1,2,4-triazole were determined by X-ray diffraction analysis.
Similar content being viewed by others
References
a) Cocco MT, Congiu C, Onnis V, Piras R (2001) Farmaco 56: 741; b) Cocco MT, Congiu C, Onnis V (1995) Farmaco 50: 73
For representative papers see: a) Aggarwal V, Ila H, Junjappa H (1982) Synthesis, 65; b) Carney RWJ, Wojtkunski J, deStevens G (1964) J Org Chem 29: 2887
a) Hsung RP, Wei L-L, Sklenicka HM, Douglas CJ, McLaughlin MJ, Mulder JA, Yao LJ (1999) Org Lett 1: 509; b) Hickmott PW, Sheppard G (1971) J Chem Soc C 2112; c) Palvannan K, Stille JR (1992) J Org Chem 57: 5319; For reviews see: d) Harrity JPA, Provoost O (2005) Org Biomol Chem 3: 1349; e) Hsung RP, Kurdyumov AV, Sydorenko N (2005) Eur J Org Chem, 23
For reviews see: a) Ferrar HMC, Pereira FLC (2004) Quim Nova 27: 89; b) Kascheres C (2003) J Braz Chem Soc 14: 945; c) Negri G, Kascheres C, Kascheres AJ (2004) J Heterocyclic Chem 41: 461
a) Goerdeler J, Pohland WH (1963) Chem Ber 96: 526; b) Goerdeler J, Gnad J (1965) Chem Ber 98: 1531; c) deStevens G, Smolisnky B, Dorfman L (1964) J Org Chem 29: 1115; d) Erian AW (1999) J Prakt Chem, 341; e) Greenhill JV, Hanaee J, Steel PJ (1990) J Chem Soc Perkin Trans 1, 1869; f) Carney RWJ, Wojtkunski J, Fechtig B, Puckett RT, Biffar B, deStevens G (1971) J Org Chem 36: 2602; g) Uher M, Ilavský D, Foltín J, Škvarenivá K (1981) Collect Czech Chem Commun 46: 3128; h) El-Dean AMK, Abdel-Monea ME (2002) J Chin Chem Soc 49: 1057
For 1H NMR data of 3a at 50°C see: Macháčev V, El-Bahaie S, Št\(\breve {\rm e}\)rba V (1981) Collect Czech Chem Commun 46: 256
Consumption of 3 is easily followed because the solution changes its color from red to yellow
LJ Farrugia (1997) J Appl Cryst 30 565 Occurrence Handle10.1107/S0021889897003117 Occurrence Handle1:CAS:528:DyaK2sXnt1KgsLg%3D
L Missio HS Braibante MEF Braibante (1996) J Heterocyclic Chem 33 1243 Occurrence Handle1:CAS:528:DyaK28XlvVaktL8%3D Occurrence Handle10.1002/jhet.5570330441
a) Braibante MEF, Braibante HS, Missio L, Andricopulo A (1994) Synthesis, 898; b) Braibante MEF, Braibante, HS, Salvatore SJSA (1990) Quim Nova 13: 67; c) Braibante HS, Braibante MEF, Rosso GB, Oriques DA (2003) J Braz Chem Soc 14: 994; d) Braibante MEF, Braibante HS, Morel AF, Costa CC, Lima MG (2006) J Braz Chem Soc 17: 184
C Kusucu B Rapino L McDermott S Hadley (2004) J Clin Microbiol 42 1224 Occurrence Handle10.1128/JCM.42.3.1224-1227.2004 Occurrence Handle1:CAS:528:DC%2BD2cXivF2ktrg%3D
Enraf-Nonius (1993) CAD-4/PC 1.2 Enraf-Nonius, Delft, The Netherlands
GM Sheldrick (1997) SHELXS97 and SHELXL9 Program for Crystal Structure Refinement University of Göttingen Germany
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cunha, S., Bastos, R., Silva, P. et al. Synthesis and Structural Studies of 4-Thioxopyrimidines with Antimicrobial Activities. Monatsh. Chem. 138, 111–119 (2007). https://doi.org/10.1007/s00706-006-0577-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-006-0577-y