Abstract
Purpose
Tumor biopsies are critical for delineating pharmacodynamic effects of drugs and for optimal patient selection during oncology clinical trials of molecular targeted therapies. The purpose of this study was to identify factors related to patients’ willingness to provide study-related tumor biopsies in phase 1 clinical trials of molecularly targeted therapy.
Methods
An investigator-designed survey, that assessed biopsy willingness, demographic and clinical factors, was completed anonymously by patients with advanced cancer in a phase 1 clinic for targeted therapy. Data were analyzed using multivariate logistic regression models with odds ratios (OR) and 95 % confidence intervals (CI).
Results
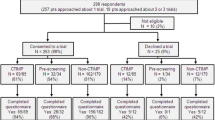
Three hundred and sixty-two patients with advanced cancer (50 % male, 56 % aged ≤60 years) participated. In univariate analyses, willingness to provide study-related biopsy was associated with male gender, white race, higher income, using the Internet for cancer-related information, and having had a biopsy previously (p < 0.05). In multivariate analyses, male gender (OR 2.41, 95 % CI 1.54, 3.78) and having had a biopsy (OR 3.71, 95 % CI 1.68, 8.15) were associated with willingness to have one biopsy; male gender (OR 1.97, 95 % CI 1.30, 3.00) and relying on the Internet as a source of information (OR 1.87, 95 % CI 1.21, 2.89) were associated with willingness to have more than one biopsy.
Conclusions
The results suggest that male gender is associated with greater stated willingness to undergo biopsy. Also, the Internet is an important source of information for patients with cancer and may strongly influence their decisions about whether to consent to biopsies in early clinical trials.
Similar content being viewed by others
References
Agulnik M, Oza AM, Pond GR, Siu LL (2006) Impact and perceptions of mandatory tumor biopsies for correlative studies in clinical trials of novel anticancer agents. J Clin Oncol 24(30):4801–4807. doi:10.1200/jco.2005.03.4496
Bernhard J, Dietrich D, Glimelius B, Hess V, Bodoky G, Scheithauer W, Herrmann R (2010) Estimating prognosis and palliation based on tumour marker CA 19–9 and quality of life indicators in patients with advanced pancreatic cancer receiving chemotherapy. Br J Cancer 103(9):1318–1324. doi:10.1038/sj.bjc.6605929
Castleton K, Fong T, Wang-Gillam A, Waqar MA, Jeffe DB, Kehlenbrink L, Gao F, Govindan R (2010) A survey of internet utilization among patients with cancer. Support Care Cancer. doi:10.1007/s00520-010-0935-5
Chabner BA (2010) New results will change the paradigm for phase 1 trials and drug approval. Oncologist 15:1023–1025
Crook JM, Gomez-Iturriaga A, Wallace K, Ma C, Fung S, Alibhai S, Jewett M, Fleshner N (2011) Comparison of health-related quality of life 5 years after SPIRIT: surgical prostatectomy versus interstitial radiation intervention trial. J Clin Oncol 29(4):362–368
Dowlati A, Haaga J, Remick SC, Spiro TP, Gerson SL, Liu L, Berger SJ, Berger NA, Willson JK (2001) Sequential tumor biopsies in early phase clinical trials of anticancer agents for pharmacodynamic evaluation. Clin Cancer Res 7(10):2971–2976
Eisenhauer EA, Twelves C, Buyse M (2006) Phase 1 Cancer Clinical Trials: A Practical Guide. Oxford University Press Inc., New York
El-Osta H, Hong D, Wheeler J, Fu S, Naing A, Falchook G, Hicks M, Wen S, Tsimberidou AM, Kurzrock R (2011) Outcomes of research biopsies in phase 1 clinical trials: the MD anderson cancer center experience. Oncologist 16(9):1292–1298
Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, O’Dwyer PJ, Lee RJ, Grippo JF, Nolop K, Chapman PB (2010) Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med 363(9):809–819
Gray SW, Armstrong K, Demichele A, Schwartz JS, Hornik RC (2009) Colon cancer patient information seeking and the adoption of targeted therapy for on-label and off-label indications. Cancer 115(7):1424–1434. doi:10.1002/cncr.24186
Helft PR, Daugherty CK (2006) Are we taking without giving in return? The ethics of research-related biopsies and the benefits of clinical trial participation. J Clin Oncol 24(30):4793–4795. doi:10.1200/jco.2006.05.7125
Izzedine H, Isnard-Bagnis C, Launay-Vacher V, Mercadal L, Tostivint I, Rixe O, Brocheriou I, Bourry E, Karie S, Saeb S, Casimir N, Billemont B, Deray G (2006) Gemcitabine-induced thrombotic microangiopathy: a systematic review. Nephrol Dial Transplant 21(11):3038–3045. doi:10.1093/ndt/gfl507
Jimenez-Gordo AM, Feliu J, Martinez B, de-Castro J, Rodriguez-Salas N, Sastre N, Vilches Y, Espinosa E, Rodriguez-Aizcorbe JR, Gonzalez-Baron M (2009) Descriptive analysis of clinical factors affecting terminally ill cancer patients. Support Care Cancer 17(3):261–269. doi:10.1007/s00520-008-0460-y
Kluger HM, Dudek AZ, McCann C, Ritacco J, Southard N, Jilaveanu LB, Molinaro A, Sznol M (2011) A phase 2 trial of dasatinib in advanced melanoma. Cancer 117(10):2202–2208. doi:10.1002/cncr.25766
Kwak EL, Bang YJ, Camidge DR, Shaw AT, Solomon B, Maki RG, Ou SH, Dezube BJ, Janne PA, Costa DB, Varella-Garcia M, Kim WH, Lynch TJ, Fidias P, Stubbs H, Engelman JA, Sequist LV, Tan W, Gandhi L, Mino-Kenudson M, Wei GC, Shreeve SM, Ratain MJ, Settleman J, Christensen JG, Haber DA, Wilner K, Salgia R, Shapiro GI, Clark JW, Iafrate AJ (2010) Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 363(18):1693–1703. doi:10.1056/NEJMoa1006448
Langford A, Resnicow K, An L (2010) Clinical trial awareness among racial/ethnic minorities in HINTS 2007: sociodemographic, attitudinal, and knowledge correlates. J Health Commun 15(Suppl 3):92–101. doi:10.1080/10810730.2010.525296
Maida V, Peck J, Ennis ME, Brar N, Maida AR (2010) Preferences for active and aggressive intervention among patients with advanced cancer. BMC Cancer 10:1–10
Mego M, De Giorgi U, Dawood S, Wang X, Valero V, Andreopoulou E, Handy B, Ueno NT, Reuben JM, Cristofanilli M (2011) Characterization of metastatic breast cancer patients with nondetectable circulating tumor cells. Int J Cancer 129(2):417–423. doi:10.1002/ijc.25690
Meischke H, Bowen D, Kuniyuki A (2001) Awareness of genetic testing for breast cancer risk among women with a family history of breast cancer: effect of women’s information sources on their awareness. Cancer Detect Prev 25(4):319–327
Naing A, Stephen SK, Frenkel M, Chandhasin C, Hong DS, Lei X, Falchook G, Wheler JJ, Fu S, Kurzrock R (2011) Prevalence of complementary medicine use in a phase 1 clinical trials program. Cancer 117:5142–5150
Park JW, Kerbel RS, Kelloff GJ, Barrett JC, Chabner BA, Parkinson DR, Peck J, Ruddon RW, Sigman CC, Slamon DJ (2004) Rationale for biomarkers and surrogate end points in mechanism-driven oncology drug development. Clin Cancer Res 10(11):3885–3896. doi:10.1158/1078-0432.ccr-03-0785
Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, Bergethon K, Shaw AT, Gettinger S, Cosper AK, Akhavanfard S, Heist RS, Temel J, Christensen JG, Wain JC, Lynch TJ, Vernovsky K, Mark EJ, Lanuti M, Iafrate AJ, Mino-Kenudson M, Engelman JA (2011) Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med 3(75):75ra26. doi:10.1126/scitranslmed.3002003
Shabanloei R, Golchin M, Esfahani A, Dolatkhah R, Rasoulian M (2010) Effects of music therapy on pain and anxiety in patients undergoing bone marrow biopsy and aspiration. AORN J 91(6):746–751. doi:10.1016/j.aorn.2010.04.001
Sherrill B, Amonkar MM, Sherif B, Maltzman J, O’Rourke L, Johnston S (2010) Quality of life in hormone receptor-positive HER-2 + metastatic breast cancer patients during treatment with letrozole alone or in combination with lapatinib. Oncologist 15(9):944–953
Stewart DJ, Kurzrock R (2009) Cancer: the road to amiens. J Clin Oncol 27:328–333
Yu EY, Wilding G, Posadas E, Gross M, Culine S, Massard C, Morris MJ, Hudes G, Calabro F, Cheng S, Trudel GC, Paliwal P, Sternberg CN (2009) Phase II study of dasatinib in patients with metastatic castration-resistant prostate cancer. Clin Cancer Res 15(23):7421–7428. doi:10.1158/1078-0432.ccr-09-1691
Acknowledgments
We thank Joann Aaron, MS, Scientific Editor, Department of Investigational Cancer Therapeutics, MD Anderson Cancer Center, for scientific editing of the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, D.S., George, G.C., Iwuanyanwu, E.C. et al. Factors related to biopsy willingness in patients with advanced cancer in a phase 1 clinic for molecularly targeted therapy. J Cancer Res Clin Oncol 139, 963–970 (2013). https://doi.org/10.1007/s00432-013-1404-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-013-1404-6