Abstract
Background
Retinoids play a key role in fetal lung development. It has been suggested that the maternal–fetal retinol transport is disrupted by trophoblastic apoptosis. The mechanism underlying nitrofen-induced apoptosis in placenta is not fully understood. Neutrophil gelatinase-associated lipocalin (NGAL) is expressed in the fetal part of the maternal–fetal interface. NGAL is part of the immune barrier and serves primarily as a transport protein transferring biologically hazardous molecules in a safe and controlled way. It has been shown that over-activation of NGAL induces apoptosis. We hypothesized that increased placental NGAL expression induces trophoblastic apoptosis in the nitrofen model of CDH.
Methods
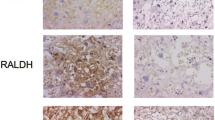
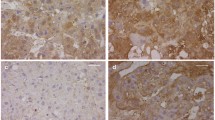
Pregnant rats were exposed to either olive oil or nitrofen on day 9 of gestation (D9). Placenta harvested on D21 and divided into two groups: control and nitrofen with CDH. Immunohistochemistry was performed to evaluate trophoblasts (by cytokeratin expression), NGAL expression, and apoptotic trophoblastic cells (using TUNEL assay). Total RNA was extracted from each placenta and the relative mRNA expression levels of NGAL were analyzed using RT-PCR.
Results
Immunohistochemistry showed NGAL immunoreactivity both in control and CDH in the fetal part of the fetal–maternal interface of placenta. Markedly increased NGAL expression was detected in CDH group compared to controls. Relative mRNA expression levels of NGAL gene were significantly increased in the CDH group compared to control in the placenta (5.924 ± 0.93 vs. 1.895 ± 0.54, p < 0.001). Markedly increased numbers of apoptotic trophoblastic cells were seen in the maternal–fetal interface in the CDH group compared to controls.
Conclusions
NGAL activation may lead to increased trophoblastic apoptosis in the maternal–fetal interface in the nitrofen model of CDH. These changes may therefore cause disturbance in maternal–fetal retinol transport affecting fetal lung morphogenesis.




Similar content being viewed by others
References
Colvin J, Bower C, Dickinson JE, Sokol J (2005) Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics 116(3):e356–e363
Stege G, Fenton A, Jaffray B (2003) Nihilism in the 1990 s: the true mortality of congenital diaphragmatic hernia. Pediatrics 112(3 Pt 1):532–535
Gosche JR, Islam S, Boulanger SC (2005) Congenital diaphragmatic hernia: searching for answers. Am J Surg 190(2):324–332
Robinson PD, Fitzgerald DA (2007) Congenital diaphragmatic hernia. Paediatr Respir Rev 8(4):323–334 quiz 334-325
Montedonico S, Nakazawa N, Puri P (2008) Congenital diaphragmatic hernia and retinoids: searching for an etiology. Pediatr Surg Int 24(7):755–761
Noble BR, Babiuk RP, Clugston RD, Underhill TM, Sun H, Kawaguchi R, Walfish PG, Blomhoff R, Gundersen TE, Greer JJ (2007) Mechanisms of action of the congenital diaphragmatic hernia-inducing teratogen nitrofen. Am J Physiol Lung Cell Mol Physiol 293(4):L1079–L1087
Kutasy B, Gosemann JH, Doi T, Fujiwara N, Friedmacher F, Puri P (2012) Nitrofen interferes with trophoblastic expression of retinol-binding protein and transthyretin during lung morphogenesis in the nitrofen-induced congenital diaphragmatic hernia model. Pediatr Surg Int 28(2):143–148
Gosemann JH, Doi T, Kutasy B, Friedmacher F, Dingemann J, Puri P (2012) Alterations of peroxisome proliferator-activated receptor gamma and monocyte chemoattractant protein 1 gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 47(5):847–851
Friedmacher F, Doi T, Gosemann JH, Fujiwara N, Kutasy B, Puri P (2012) Upregulation of fibroblast growth factor receptor 2 and 3 in the late stages of fetal lung development in the nitrofen rat model. Pediatr Surg Int 28(2):195–199
Clagett-Dame M, DeLuca HF (2002) The role of vitamin A in mammalian reproduction and embryonic development. Annu Rev Nutr 22:347–381
Beurskens LW, Tibboel D, Lindemans J, Duvekot JJ, Cohen-Overbeek TE, Veenma DC, de Klein A, Greer JJ, Steegers-Theunissen RP (2010) Retinol status of newborn infants is associated with congenital diaphragmatic hernia. Pediatrics 126(4):712–720
Gallot D, Marceau G, Coste K, Hadden H, Robert-Gnansia E, Laurichesse H, Dechelotte PJ, Labbe A, Dastugue B, Lemery D, Sapin V (2005) Congenital diaphragmatic hernia: a retinoid-signaling pathway disruption during lung development? Birth Defects Res A Clin Mol Teratol 73(8):523–531
Nakazawa N, Montedonico S, Takayasu H, Paradisi F, Puri P (2007) Disturbance of retinol transportation causes nitrofen-induced hypoplastic lung. J Pediatr Surg 42(2):345–349
Ruttenstock E, Doi T, Dingemann J, Puri P (2011) Prenatal administration of retinoic acid upregulates insulin-like growth factor receptors in the nitrofen-induced hypoplastic lung. Birth Defects Res B Dev Reprod Toxicol 92(2):148–151
Quadro L, Hamberger L, Gottesman ME, Wang F, Colantuoni V, Blaner WS, Mendelsohn CL (2005) Pathways of vitamin A delivery to the embryo: insights from a new tunable model of embryonic vitamin A deficiency. Endocrinology 146(10):4479–4490
Quadro L, Hamberger L, Gottesman ME, Colantuoni V, Ramakrishnan R, Blaner WS (2004) Transplacental delivery of retinoid: the role of retinol-binding protein and lipoprotein retinyl ester. Am J Physiol Endocrinol Metab 286(5):E844–E851
Tadesse S, Luo G, Park JS, Kim BJ, Snegovskikh VV, Zheng T, Hodgson EJ, Arcuri F, Toti P, Parikh CR, Guller S, Norwitz ER (2011) Intra-amniotic infection upregulates neutrophil gelatinase-associated lipocalin (NGAL) expression at the maternal–fetal interface at term: implications for infection-related preterm birth. Reprod Sci 18(8):713–722
Bahmani P, Halabian R, Rouhbakhsh M, Roushandeh AM, Masroori N, Ebrahimi M, Samadikuchaksaraei A, Shokrgozar MA, Roudkenar MH (2010) Neutrophil gelatinase-associated lipocalin induces the expression of heme oxygenase-1 and superoxide dismutase 1, 2. Cell Stress Chaperones 15(4):395–403
Nelson AM, Zhao W, Gilliland KL, Zaenglein AL, Liu W, Thiboutot DM (2008) Neutrophil gelatinase-associated lipocalin mediates 13-cis retinoic acid-induced apoptosis of human sebaceous gland cells. J Clin Invest 118(4):1468–1478
Dingemann J, Doi T, Ruttenstock E, Puri P (2010) Abnormal platelet-derived growth factor signaling accounting for lung hypoplasia in experimental congenital diaphragmatic hernia. J Pediatr Surg 45(10):1989–1994
Morriss-Kay GM, Ward SJ (1999) Retinoids and mammalian development. Int Rev Cytol 188:73–131
Sapin V, Ward SJ, Bronner S, Chambon P, Dolle P (1997) Differential expression of transcripts encoding retinoid binding proteins and retinoic acid receptors during placentation of the mouse. Dev Dyn 208(2):199–210
Clugston RD, Zhang W, Greer JJ (2010) Early development of the primordial mammalian diaphragm and cellular mechanisms of nitrofen-induced congenital diaphragmatic hernia. Birth Defects Res A Clin Mol Teratol 88(1):15–24
Kling DE, Cavicchio AJ, Sollinger CA, Schnitzer JJ, Kinane TB, Newburg DS (2010) Nitrofen induces apoptosis independently of retinaldehyde dehydrogenase (RALDH) inhibition. Birth Defects Res B Dev Reprod Toxicol 89(3):223–232
Manson JM (1986) Mechanism of nitrofen teratogenesis. Environ Health Perspect 70:137–147
Brown TJ, Manson JM (1986) Further characterization of the distribution and metabolism of nitrofen in the pregnant rat. Teratology 34(2):129–139
Devireddy LR, Gazin C, Zhu X, Green MR (2005) A cell-surface receptor for lipocalin 24p3 selectively mediates apoptosis and iron uptake. Cell 123(7):1293–1305
Berger T, Togawa A, Duncan GS, Elia AJ, You-Ten A, Wakeham A, Fong HE, Cheung CC, Mak TW (2006) Lipocalin 2-deficient mice exhibit increased sensitivity to Escherichia coli infection but not to ischemia-reperfusion injury. Proc Natl Acad Sci U S A 103(6):1834–1839
Bachman MA, Miller VL, Weiser JN (2009) Mucosal lipocalin 2 has pro-inflammatory and iron-sequestering effects in response to bacterial enterobactin. PLoS Pathog 5(10):e1000622
Zelko IN, Mariani TJ, Folz RJ (2002) Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic Biol Med 33(3):337–349
Gonzalez-Reyes S, Martinez L, Martinez-Calonge W, Fernandez-Dumont V, Tovar JA (2006) Effects of antioxidant vitamins on molecular regulators involved in lung hypoplasia induced by nitrofen. J Pediatr Surg 41(8):1446–1452
Gonzalez-Reyes S, Martinez L, Martinez-Calonge W, Fernandez-Dumont V, Tovar JA (2006) Effects of nitrofen and vitamins A, C and E on maturation of cultured human H441 pneumocytes. Biol Neonate 90(1):9–16
Islam S, Narra V, Cote GM, Manganaro TF, Donahoe PK, Schnitzer JJ (1999) Prenatal vitamin E treatment improves lung growth in fetal rats with congenital diaphragmatic hernia. J Pediatr Surg 34(1):172–176 discussion 176–177
Thebaud B, Tibboel D, Rambaud C, Mercier JC, Bourbon JR, Dinh-Xuan AT, Archer SL (1999) Vitamin A decreases the incidence and severity of nitrofen-induced congenital diaphragmatic hernia in rats. Am J Physiol 277(2 Pt 1):L423–L429
Fisher JC, Kling DE, Kinane TB, Schnitzer JJ (2002) Oxidation-reduction (redox) controls fetal hypoplastic lung growth. J Surg Res 106(2):287–291
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kutasy, B., Gosemann, J.H., Duess, J.W. et al. Increased trophoblastic apoptosis mediated by neutrophil gelatinase-associated lipocalin (NGAL) activation in the nitrofen model of congenital diaphragmatic hernia. Pediatr Surg Int 29, 25–31 (2013). https://doi.org/10.1007/s00383-012-3203-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-012-3203-6