Abstract
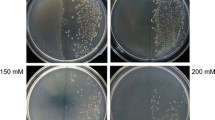
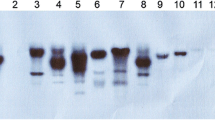
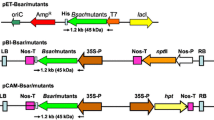
Glyphosate is a non-selective broad-spectrum herbicide that inhibits 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS). This is a key enzyme in the aromatic amino acid biosynthesis pathway of microorganisms and plants. The manipulation of bacterial EPSPS gene in order to reduce its affinity for glyphosate, followed by its transfer to plants is one of the most effective approaches for the production of glyphosate-tolerant plants. In this study, we chose to focus on amino acid residues glycine96 and alanine183 of the E. coli (k12) EPSPS enzyme. These two amino acids are important residues for glyphosate binding. We used site directed mutagenesis (SDM) to induce point mutations in the E. coli EPSPS gene, in order to convert glycine96 to alanine (Gly96Ala) and alanine183 to threonine (Ala183Thr). After confirming the mutation by sequencing, the altered EPSPS gene was transferred to rapeseed (Brassica napus L.) via Agrobacterium-mediated transformation. The transformed explants were screened in shoot induction medium containing 25 mg L−1 kanamycin. Glyphosate tolerance was assayed in putative transgenic plants. Statistical analysis of data showed that there was a significant difference between the transgenic and control plants. It was observed that transgenic plants were resistant to glyphosate at a concentration of 10 mM whereas the non-transformed control plants were unable to survive 1 mM glyphosate. The presence and copy numbers of the transgene were confirmed with PCR and Southern blotting analysis, respectively.







Similar content being viewed by others
References
Australia New Zealand Food Authority (ANZFA) (1999) Guidelines for the safety assessment of foods to be included in Standard A18 – Food Produced Using Gene Technology
Baerson SR, Rodriguez DJ, Tran M, Feng Y, Biest NA, Dill GM (2002) Glyphosate- resistance goose grass. Identification of a mutation in the target enzyme 5-enolpyruvylshikimate-3-phosphate synthase. Plant Physiol 129:1265–1275
Bhalla PL, Smith N (1998) Agrobacterium tumefaciens-mediated transformation of cauliflower, Brassica oleracea var. botrytis. Mol Breed 4:531–541
Boocock MR, Coggins JR (1983) Kinetics of 5-Enolpyruvylshikimate-3-phosphate synthase inhibition by glyphosate. FEBS Lett 154(1):127–133
Cajacob CA, Feng P, Heck J, Alibhai M, Sammons R, Padgette S (2004) Engineering resistance to herbicides. In: Christou P, Klee H (eds) Handbook of plant biotechnology. Wiley, New York, 353–372
Comai LD, Facciotti D, Hiatt WR, Thompson G, Rose RE, Stalker DM (1985) Expression in plants of a mutant aroA gene from Salmonella typhimurium confers tolerance to glyphosate. Nature 317:741–744
Eichholtz DA, Alan D, Gasser CS, Scott C, Kishore GM, Murthy G (2001) United States Patent No. 6,225,114
Franz JE, Mao MK, Sikorski JA (1997) Glyphosate's molecular mode of action. In: Glyphosate: a unique global herbicide. American Chemical Society, Washington, DC, pp 521–642
Fry J, Barnason A, Horsch R (1987). Transformation of Brassica napus with Agrobacterium based vectors. Plant Cell Rep 6:321–325
Gruys KJ, Sikorski JA (1999) Inhibitors of tryptophan, phenylalanine, and tyrosine biosynthesis as herbicides. In: Singh B (ed) Plant amino acids. Marcel Dekker, New York, pp 357–384
Haslam E (1993) Shikimic acid: metabolism and metabolites. Wiley, Chi Chester, UK
Hoekema A, Hirsch PR, Hooykaas PJJ, Schilperoort RA (1983) A binary plant vector strategy based on separation of vir- and T-region of Agrobacterium tumefaciens Ti-plasmid. Nature 303:179–180
Höfgen R, Willmitzer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877
Holt JS, Poweles SB, Holtum AM (1993) Mechanisms and organic aspects of herbicide resistance. Annu Rev Plant Physiol Plant Mol Biol 44:203–229
James C (2003) ISAAA Briefs: Preview, Global Status of Commercialized Transgenic Crops, In ISAAA Briefs 30, ISAAA, Ithaca, NY
Kishore GM, Brundage L, Kolk K, Padgette SR, Rochester D, Huynh QK, della-Cioppa G (1986) Isolation, purification and characterization of a glyphosate tolerant mutant E. coli EPSP synthase. Proc Fed Am Soc Exp Biol 45:1506
Kishore GM, Padgette SR, Fraley RT (1992) History of herbicide-tolerant crops, methods of development and current state of the art-emphasis on Glyphosate tolerance. Weed Technol 6:626–634
Kuiper HA, Kleter GA, Noordam MY (2000) Risks of the release of transgenic herbicide-resistant plants with respect to humans, animals, and the environment. Crop Prot 19:773–778
Malik J, Barry G, Kishore G (1989) The herbicide glyphosate. BioFactors 2:17–25
Marzabadi MR, Gruys KJ, Pansegran PD, Walker MC, Yuen HK, Sikorski JA (1996) An EPSP synthase inhibitor joining shikimate-3-phosphate with glyphosate: synthesis and ligand binding studies. Biochemistry 35:4199–4210
McDowell LM, Schmidt A, Cohen ER, Studelska DR, Schaefer J (1996) Structural constraints on the ternary complex of 5-enolpyruvylshikimic acid-3-phosphate synthase from rotational-echo double-resonance NMR. J Mol Biol 256:160–171
Meilan R, Han KH, Ma C, DiFazio SP, Eaton JA, Hoien EA, Stanton BJ, Crockett RP, Taylor ML, James RR, Skinner JS, Jouanian L, Pilate G, Strauss SH (2002) The CP4 transgene provides high level of tolerance to Roundup® herbicide in field-grown hybrid poplars. Canad J Res 32:967–976
Moloney MM, Walker JM, Sharma KK (1989) High efficiency transformation of Brassica napus using Agrobacterium vectors. Plant Cell Rep 8:238–242
Murray MG, Thampson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acid Res 8:4321–4325
Padgette SR, Re DB, Gasser CS, Eichholtz DA, Frazier RB, Hironaka CM, Levine EB, Shah DM, Fraley RT, Kishore GM (1991) Site-directed mutagenesis of a conserved region of the 5-enolpyruvylshikimic acid-3-phosphate synthase active site. J Biol Chem 266:22361–22369
Ream JE, Yuen HK, Frazier RB, Sikorski JA (1992) EPSP synthase: binding studies using isothermal titration microcalorimetry and equilibrium dialysis and their implication for ligand recognition and kinetic mechanism. Biochemistry 31:5528–5534
Sambrook J, Russel DW (2001) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory Press. New York, pp 12.1–12.114
Sost D, Amrhein N (1990) Substitution of Gly96 to Ala in the 5-enolpyruvylshikimate-3-phosphate synthase of Klebsiella pneumoniae results in a greatly reduced affinity for the herbicide glyphosate. Arc Biochem Biophys 282(2):433–436
Sharma KK (1987) Control of organ differentiation from somatic tissues and pollen embryogenesis in anther culture of B. juncea. Ph.D. thesis, Department of Botany, University of Delhi
Stalker DM, Hiatt WR, Comai LD (1985) A single amino acid substitution in the enzyme 5-enolpyruvylshikimic acid-3-phosphate synthase confers resistance to herbicide glyphosate. J Biol Chem 260:4724–4728
Stam M, Mol JNM, Kooper JM (1997) The silence of genes in transgenic plants. Ann Bot 79:3–12
Steinruken HC, Amrhein N (1980) The herbicide glyphosate is a potent inhibitor of 5-enolpyruvylshikimic acid-3-phosphate synthase. Biochem Biophy Res Commun 94:1207–1212
Wang HY, Li YF, Xie LX, Xu P (2003) Expression of bacterial aroA mutant, aroA-M1, encoding 5-enolpyruvylshikimste-3-phosphate synthase for the production of glyphosate-resistant tobacco plants. J Plant Res 116:455–460
Acknowledgments
Special thanks to Dr. P. Shariati and Dr. Christopher Preston for critical reading of this manuscript. The National Institute for Genetic Engineering and Biotechnology (NIGEB), Iran, has financially supported this research project no. 155.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Schmidt
Rights and permissions
About this article
Cite this article
Kahrizi, D., Salmanian, A.H., Afshari, A. et al. Simultaneous substitution of Gly96 to Ala and Ala183 to Thr in 5-enolpyruvylshikimate-3-phosphate synthase gene of E. coli (k12) and transformation of rapeseed (Brassica napus L.) in order to make tolerance to glyphosate. Plant Cell Rep 26, 95–104 (2007). https://doi.org/10.1007/s00299-006-0208-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-006-0208-4