Abstract
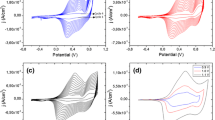
A new copolymer derived from 2,7-carbazole with thiophene is reported. This copolymer was obtained through the synthesis of 2,7-dibromo-N-methyl carbazole and 2-bromothiophene using a Kumada-type reaction to bond both rings and using two different kinds of Pd catalysts, [Pd(OAc)2] and [Pd(PPh3)4], with significant differences in their yields. The trimer that was obtained was subsequently electropolymerized using cyclic voltammetry, and the photoelectrochemical properties were determined. Polymerization using electrochemistry showed that the trimer contains two types of possible bonds for the formation of the macromolecule, which led to the formation of two distinctly different materials. The obtained materials exhibit promising photoelectric responses suggesting that they both can be explored in the designing of photovoltaic cells.








Similar content being viewed by others
References
Goetzbergera A, Heblinga C, Schock HW (2003) Photovoltaic materials, history, status and outlook. Mater Sci Eng R Rep 40:1–46
Günes S, Neugebauer H, Sariciftci NS (2007) Conjugated polymer-based organic solar cells. Chem Rev 107:1324–1338
Cheng YJ, Yang SH, Hsu CS (2009) Synthesis of conjugated polymers for solar cell applications. Chem Rev 109:5868–5923
Morin JF, Leclerc M, Adès D, Siove A (2005) Polycarbazoles: 25 years of progress. Macromol Rapid Commun 26:761–778
Boudreault PT, Beaupré S, Leclerc M (2010) Polycarbazoles for plastic electronics. Polym Chem 1:127–136
Dierschke F, Grimsdale AC, Müllen K (2003) Efficient SYNTHESIS OF 2,7-DIBROMOCARBAZOLES AS COMPONENTS FOR ELECTROACTIVE MATERIALS. Synthesis 16:2470–2472
Appukkuttan P, Van der Eycken E, Dehaen W (2005) Microwave-enhanced Cadogan cyclization: an easy access to the 2-substituted carbazoles and other fused heterocyclic systems. Synlett 01:127–133
Freeman W, Urvoy M, Criswell ME (2005) Triphenylphosphine-mediated reductive cyclization of 2-nitrobiphenyls: a practical and convenient synthesis of carbazoles. J Org Chem 70:5014–5019
Bouchard J, Wakim S, Leclerc M (2004) Synthesis of diindolocarbazoles by Cadogan reaction: route to ladder oligo(p-aniline)s. J Org Chem 69:5705–5711
Blouin N, Michaud A, Gendron D, Wakim S, Bair E, Neagu-Plesu R, Belletête M, Durocher G, Tao Y, Leclerc M (2008) Toward a rational design of poly(2,7-carbazole) derivatives for solar cells. J Am Chem Soc 130:732–742
Sato M, Tanaka S, Kaeriyama KJ (1985) Electrochemical preparation of highly conducting polythiophene films. J Chem Soc Chem Commun 11:713–714
Zotti G, Schiavon G, Zechin S, Morin JF, Leclerc M (2002) Electrochemical, conductive, and magnetic properties of 2,7-carbazole-based conjugated polymers. Macromolecules 35:2122–2128
Heinze J, Fontana-Uribe BA, Ludwings S (2010) Electrochemistry of conducting polymers, persistent models and new concepts. Chem Rev 110:4724–4771
Kawabata K, Goto H (2010) Electrosynthesis of 2,7-linked polycarbazole derivatives to realize low-bandgap electroactive polymer. Synth Met 160:2290–2298
Wei Z, Xu J, Nie G, Du Y, Pu S (2006) Low-potential electrochemical polymerization of carbazole and its alkyl derivatives. J Electroanal Chem 589:112–119
Roncali J (1992) Conjugated poly(thiophenes): synthesis, functionalization, and applications. Chem Rev 92:711–738
Bredas JL, Silbey R, Boudreaux DS, Chance RR (1983) Chain-length dependence of electronic and electrochemical properties of conjugated systems: polyacetylene, polyphenylene, polythiophene, and polypyrrole. J Am Chem Soc 105:6555–6559
Acknowledgments
The authors acknowledge financial support from FONDECYT Project No. 11080073. Acknowledges DEA/PUCV and CONICYT for doctorate scholarship for financial support of J.A.A., L.B., J.C.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aristizabal, J.A., Soto, J.P., Ballesteros, L. et al. Synthesis, electropolymerization, and photoelectrochemical characterization of 2,7-di(thiophen-2-yl)-N-methylcarbazole. Polym. Bull. 70, 35–46 (2013). https://doi.org/10.1007/s00289-012-0817-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-012-0817-8