Abstract
Purpose
To evaluate cisplatin (CDDP) pharmacokinetics after its intravenous (IV) or intrahepatic arterial administration (IHA) in healthy pigs with or without embolization by absorbable gelatine.
Material and Methods
We analysed plasmatic and hepatic drug concentration in four groups of six mini-pigs each according to the modality of administration of CDDP (1 mg/kg): IV, IHA, IHA with partial embolization using absorbable gelatine (IHA-Pe), and IHA with complete embolization (IHA-Te). Unbounded plasmatic and hepatic platinum concentrations were measured. Concentration and pharmacokinetics parameters were compared using analysis of variance.
Results
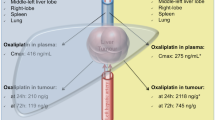
For all groups, there was a rapid and biexponential decrease in free platinum concentration. Plasmatic terminal half-life (T1/2) was significantly decreased after embolization at 191, 178, 42, and 41 min after IV, IHA, IHA-Pe, and IHA-Te administration, respectively. Maximal plasmatic concentration and systemic exposure to CDDP (AUC24) values were significantly decreased after embolization (Cmax p = 0.0075; AUC24 p = 0.0053). Hepatic CDDP concentration rapidly peaked and then decreased progressively. After 24 h, the residual concentration represented 45, 47, 60, and 63 % of Cmax, respectively, after IV, IHA, IHA-Pe, and IHA-Te. Hepatic T1/2 and AUC∞ values were increased after embolization, but the differences were not statistically significant.
Conclusion
This preliminary study confirms the feasibility of a pig model to study systemic and hepatic CDDP pharmacokinetics. Systemic exposure is lower after embolization, which could minimize systemic toxicity. Hepatic T1/2 elimination and hepatic exposition values are increased with IHA compared with IV administration.





Similar content being viewed by others
References
Bruix J, Sherman M (2005) Management of hepatocellular carcinoma. Hepatology 42(5):1208–1236
Llovet JM et al (2002) Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 359(9319):1734–1739
Lo CM et al (2002) Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 35(5):1164–1171
Daniels JR, Wallman M (2010) Subselective intra-arterial chemotherapy infusion in the treatment of hepatocellular carcinoma. Semin Oncol 37(2):83–88
Egawa H et al (1990) Effects of intra-arterial chemotherapy with a new lipophilic anticancer agent, estradiol-chlorambucil (KM2210), dissolved in lipiodol on experimental liver tumor in rats. J Surg Oncol 44(2):109–114
Konno T (1990) Targeting cancer chemotherapeutic agents by use of lipiodol contrast medium. Cancer 66(9):1897–1903
Nakamura H et al (1989) Transcatheter oily chemoembolization of hepatocellular carcinoma. Radiology 170(3 Pt 1):783–786
Sasaki Y et al (1987) A new approach to chemoembolization therapy for hepatoma using ethiodized oil, cisplatin, and gelatin sponge. Cancer 60(6):1194–1203
Marelli L et al (2007) Transarterial therapy for hepatocellular carcinoma: which technique is more effective? A systematic review of cohort and randomized studies. Cardiovasc Intervent Radiol 30(1):6–25
Chen PJ et al (2010) Issues and controversies of hepatocellular carcinoma-targeted therapy clinical trials in Asia: experts’ opinion. Liver Int 30(10):1427–1438
Stewart DJ et al (1983) Clinical pharmacology of intra-arterial cis-diamminedichloroplatinum(II). Cancer Res 43(2):917–920
Johnson PJ et al (1991) Pharmacokinetics and toxicity of intra-arterial adriamycin for hepatocellular carcinoma: effect of coadministration of lipiodol. J Hepatol 13(1):120–127
Dzodic R et al (2004) Pharmacokinetic advantage of intra-arterial hepatic oxaliplatin administration: comparative results with cisplatin using a rabbit VX2 tumor model. Anticancer Drugs 15(6):647–650
Cagol PP, Pasqual E, Bacchetti S (2006) Potential advantages of loco-regional intra-arterial chemotherapy. In Vivo 20(6A):777–779
Kelsen DP et al (1982) Pharmacokinetics of cisplatin regional hepatic infusions. Am J Clin Oncol 5(2):173–178
Ding JW et al (1992) Pharmacokinetics of mitomycin C following hepatic arterial chemoembolization with gelfoam. HPB Surg 5(3):161–167 (discussion 167–169)
Civalleri D et al (1991) Liver and tumor uptake and plasma pharmacokinetic of arterial cisplatin administered with and without starch microspheres in patients with liver metastases. Cancer 68(5):988–994
Sadahiro S et al (2003) Pharmacokinetics of 5-fluorouracil following hepatic intra-arterial infusion in a VX2 hepatic metastasis model. Jpn J Clin Oncol 33(8):377–381
Barone R (ed) (1996) Anatomie comparée des mamifères domestiques, 5th edn. Angiologie, Vigot, Paris
Zhang L et al (2004) Study of hepatic function matching between Banna minipig inbred and humans. Transplant Proc 36(8):2492–2494
Charte nationale portant sur l’expérimenation animale (2008) http://www.enseignementsup-recherche.gouv.fr/cid28541/la-charte-nationale-portant-sur-l-ethique-de-l-experimentation-animale.html
Shin SW (2009) The current practice of transarterial chemoembolization for the treatment of hepatocellular carcinoma. Korean J Radiol 10(5):425–434
Pera MF Jr, Harder HC (1977) Analysis for platinum in biological material by flameless atomic absorption spectrometry. Clin Chem 23(7):1245–9124
Bannister SJ et al (1978) Atomic absorption spectrophotometry of free circulating platinum species in plasma derived from cis-dichlorodiammineplatinum(II). Clin Chem 24(6):877–880
Litterst CL et al (1976) Distribution and disposition of platinum following intravenous administration of cis-diamminedichloroplatinum(II) (NSC 119875) to dogs. Cancer Res 36(7 Pt 1):2340–2344
Vexler AM et al (1995) Reduction of the systemic toxicity of cisplatin by intra-arterial hepatic route administration for liver malignancies. Int J Cancer 60(5):611–615
Kruh GD (2003) Lustrous insights into cisplatin accumulation: copper transporters. Clin Cancer Res 9(16 Pt 1):5807–5809
Raoul JL et al (1992) Chemoembolization of hepatocellular carcinomas. A study of the biodistribution and pharmacokinetics of doxorubicin. Cancer 70(3):585–590
Acknowledgments
We thank Sylvie Massoulier, Carine Nicolas, and Denis Gourcy for their contributions during the experiment.
Conflict of interest
All the authors declared that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chabrot, P., Cardot, JM., Guibert, P. et al. Cisplatin Pharmacokinetics in Nontumoral Pig Liver Treated With Intravenous or Transarterial Hepatic Chemoembolization. Cardiovasc Intervent Radiol 35, 1467–1474 (2012). https://doi.org/10.1007/s00270-012-0386-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-012-0386-0