Abstract
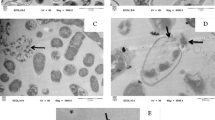
Bacitracin is a metal-dependent dodecapeptide antipeptide produced by Bacillus species. Microcalorimetry was used to study the antimicrobial activity of bacitracin and bacitracin–metal ion complexation inhibited on Staphylococcus aureus at 37°C. The affinity of metal ions binding to bacitracin was investigated by isothermal titration calorimetry and was as follows: Cu(II) ≥ Ni(II) > Co(II) > Zn(II) ≥ Mn(II). The metal ion binding affinity is not relative to the antimicrobial activity of bacitracin–metal complexation. Atomic force microscopic images revealed that the surface of S. aureus treated by bacitracin–Zn(II) was rather rough compared to that treated by bacitracin only. The central cell surface displayed small depressed grooves around the septal annulus at the onset of division. Bacitracin mainly inhibited the splitting system within the thick cross walls as seen by transmission electron microscopy (TEM). The inhibition mechanism of bacitracin may be relative to the assistance of Zn(II) coordination with the cell surface as seen by TEM. We can put forward that the activity of bacitracin only inhibited growth and division initially from the synthesis of the cell wall, especially the cell wall of the septal annulus. The divalent metal ions function to increase the adsorption of bacitracin onto the cell surface.








Similar content being viewed by others
References
Abdulrahim SMM, Haddadin SY, Odetallah NHM, Robinson RK (1999) Effect of Lactobacillus acidophilus and zinc bacitracin as dietary additives for broiler chickens. Br Poultry Sci 40:91–94
Bechtel DB, Bulla LA Jr (1976) Electron microscope study of sporulation and parasporal crystal formation in Bacillus thuringiensis. J Bacteriol 127:1472–1481
Beveridge TJ, Popkin TJ, Cole RM (1993) Electron microscopy. In: Gerhardted P (ed) Methods for general and molecular bacteriology. American Society for Microbiology, Washington DC, pp 42–71
Braga PC, Ricci D (1998) Atomic force microscopy: application to investigation of Escherichia coli morphology before and after exposure to cefodizime. Antimicrob Agents Chemother 42:18–22
Braga PC, Ricci D (2000) Detection of rokitamycin induced morphostructural alterations in Helicobacter pylori by atomic force microscopy. Chemotherapy 46:15–22
Braga PC, Ricci D (2002) Differences in the susceptibility of Streptococcus pyogenes to rokitamycin and erythromycin A revealed by morphostructural atomic force microscopy. J Antimicrob Chemother 50:457–460
Braga PC, Sasso M, Maci DS (1997) Cefodizime: effects of sub-inhibitory concentrations on adhesiveness and bacterial morphology of Staphylococcus aureus and Escherichia coli: comparison with cefotaxime and ceftriaxone. J Antimicrob Chemother 39:79–84
Calabrese EJ, Blain R (2005) The occurrence of hormetic dose responses in the toxicological literature, the hormesis database: an overview. Toxicol Appl Pharmacol 202:289–301
Chang TW, Gorbach SL, Bartlett JG, Saginur R (1980) Bacitracin treatment of antibiotic-associated colitis and diarrhea caused by Clostridium difficile toxin. Gastroenterology 78:1584–1586
Chaw KC, Manimaran M, Tay FEH (2005) Role of silver ions in destabilization of intermolecular adhesion forces measured by atomic force microscopy in Staphylococcus epidermidis biofilms. Antimicrob Agents Chemother 49:4853–4859
Dufrêne YF (2004a) Refining our perception of bacterial surfaces with the atomic force microscope. J Bacteriol 186:3283–3285
Dufrêne YF (2004b) Using nanotechniques to explore microbial surfaces. Nat Rev Microbiol 2:451–460
Epperson JD, Ming LJ (2000) Proton NMR studies of Co II complexes of the peptide antibiotic bacitracin and analogues: insight into structure–activity relationship. Biochemistry 39:4037–4045
Froyshov O (1984) The bacitracins: properties, biosynthesis, and fermentation. In: Vandamme EJ (ed) Biotechnology of industrial antibiotics. Marcel Dekker, New York, pp 665–694
Garbutt JT, Morehouse AL, Hanson AM (1961) Metal binding properties of bacitracins. J Agric Food Chem 9:285
Giesbrecht P, Wecke J, Reinicke B (1976) On the morphogenesis of the cell wall of staphylococci. Int Rev Cytol 44:225–318
Giesbrecht P, Kersten T, Maidhof H, Wecke J (1997) Two alternative mechanisms of cell separation in staphylococci: one lytic and one mechanical. Arch Microbiol 167:239–250
Giesbrecht P, Kersten T, Maidhof H, Wecke J (1998) Staphylococcal cell wall: morphogenesis and fatal variations in the presence of penicillin. Microbiol Mol Biol Rev 62:1371–1414
Hammes WP, Otto Kandler JW (1979) The sensitivity of the pseudomurein-containing genus Methanobacterium to inhibitors of murein synthesis. Arch Microbiol 123:275–279
Harel YM, Bailone A, Bibi E (1999) Resistance to bacitracin as modulated by an Escherichia coli homologue of the bacitracin ABC transporter BcrC subunit from Bacillus licheniformis. J Bacteriol 181:6176–6178
Hirotsu Y, Nishiuchi Y, Shiba T (1978) Total synthesis of bacitracin F. Peptide Chem 9:171–176
Johansen CTG, Gram L (1996) Changes in cell morphology of Listeria monocytogenes and Shewanella putrefaciens resulting from the action of protamine. Appl Environ Microbiol 62:1058–1064
Konigsberg W, Craig LC (1962) On bacitracin F. J Org Chem 27:934–938
Konz D, Klens A, Schörgendorfer K, Marahiel MA (1997) The bacitracin biosynthesis operon of Bacillus licheniformis ATCC 10716: molecular characterization of three multi-modular peptide synthetases. Chem Biol 4:927–937
Kotra LP, Golemi D, Amro NA, Liu GY, Mobashery S (1999) Dynamics of the lipopolysaccharide assembly on the surface of Escherichia coli. J Am Chem Soc 121:8707–8711
Lyon BR, Skurray R (1987) Antimicrobial resistance of Staphylococcus aureus: genetic basis. Microbiol Rev 51:88–134
MacKenzie FM, Greig P, Morrison D, Edwards G, Gould IM (2002) Identification and characterization of teicoplanin-intermediate Staphylococcus aureus blood culture isolates in NE Scotland. J Antimicrob Chemother 50:689–697
Matias VRF, Beveridge TJ (2006) Native cell wall organization shown by cryo-electron microscopy confirms the existence of a periplasmic space in Staphylococcus aureus. J Bacteriol 188:1011–1021
Matsuzaki K, Murase O, Tokuda H, Funakoshi S, Fujii N, Miyajima K (1994) Orientational and aggregational states of magainin 2 in phospholipid bilayers. Biochemistry 33:3342–3349
Meincken M, Holroyd DL, Rautenbach M (2005) Atomic force microscopy study of the effect of antimicrobial peptides on the cell envelope of Escherichia coli. Antimicrob Agents Chemother 49:4085–4092
Ming LJ, Epperson JD (2002) Metal binding and structure–activity relationship of the metalloantibiotic peptide bacitracin. J Inorg Biochem 91:46–58
Monroe S, Polk R (2000) Antimicrobial use and bacterial resistance. Curr Opin Microbiol 3:496–501
Nagaraja TG, Chengappa MM (1998) Liver abscesses in feedlot cattle: a review. J Anim Sci 76:287–298
O’Neill NM, Vine GJ, Beezer AE, Bishop AH, Hadgraft J, Labetoulle C, Walker M, Bowler PG (2003) Antimicrobial properties of silver-containing wound dressings: a microcalorimetric study. Int J Pharm 263:61
Pancholi V, Fischetti VA (1988) Isolation and characterization of the cell-associated region of group a streptococcal M6 protein. J Bacteriol 170:2618–2624
Paul TR, Venter A, Blaszczak LC, Parr JRTR, Labischinski H, Beveridge TJ (1995) Localization of penicillin-binding proteins to the splitting system of Staphylococcus aureus septa by using a mercury-penicillin v derivative. J Bacteriol 177:3631–3640
Podlesek Z, Comino A, Herzog-Velikonja B, Zgur-Bertok D, Komel R, Grabnar M (1995) Bacillus licheniformis bacitracin resistance ABC transporter: relationship to mammalian multidrug resistance. Mol Microbiol 16:969–976
Podlesek Z, Comino A, Herzog-Velikonja B, Grabnar M (2000) The role of the bacitracin ABC transporter in bacitracin resistance and collateral detergent sensitivity. FEMS Microbiol Lett 188:103–106
Rao S, Venkateswerlu G (1989) Metal ion influence on the growth inhibition of Neurospora crassa by bacitracin. Microbiology 19:253–258
Rogers HJ, Perkins HR, Ward JB (1980) Microbial cell walls and membranes. Chapman and Hall, London
Schneewind O, Fowler A, Faull KF (1995) Structure of the cell wall anchor of surface proteins in Staphylococcus aureus. Science 268:103–106
Scogin DA, Mosberg HI, Storm DR, Gennis RB (1980) Binding of nickel and zinc ions to bacitracin A. Biochemistry 19:3348–3352
Seebauer EG, Duliba EP, Scogin DA, Gennis RB, Belford RL (1983) EPR evidence on the structure of the copper-bacitracin A complex. J Am Chem Soc 105:4926–4929
Sin DW, Ho C, Wong Y, Ho S, Ip AC (2005) Analysis of major components of residual bacitracin and colistin in food samples by liquid chromatography tandem mass spectrometry. Anal Chim Acta 535:23–31
Stone KJ, Strominger JL (1971) Mechanism of action of bacitracin: complexation with metal ion and C55 -isoprenyl pyrophosphate. Proc Natl Acad Sci USA 68:3223–3227
Storm DR (1974) Mechanism of bacitracin action: a specifc lipidpeptide interaction. Ann NY Acad Sci 235:387–398
Storm DR, Strominger JL (1967) Bacitracin: an inhibitor of the dephosphorylation of lipid pyrophosphate, an intermediate in the biosynthesis of the peptidoglycan of bacterial cell walls. Proc Natl Acad Sci USA 57:767–773
Storm DR, Strominger JL (1973) Complex formation between bacitracin peptides and isoprenyl pyrophosphates. J Biol Chem 248:3940–3945
Suurkuusk J, Wadso I (1982) A multichannel microcalorimetry system. Chem Sci 20:155–163
Tollersrud T, Berge T, Andersen RS, Lund A (2001) Imaging the surface of Staphylococcus aureus by atomic force microscopy. APMIS 109:541–545
Toscano WA Jr, Storm DR (1982) Bacitracin. Pharmacol Ther 16:199–210
Touhami A, Jericho MH, Boyd JM, Beveridge TJ (2006) Nanoscale characterization and determination of adhesion forces of Pseudomonas aeruginosa pili by using atomic force microscopy. J Bacteriol 188:370–377
Tsuji K, Robertson JH (1975) Improved high-performance liquid chromatographic method for polypeptide antibiotics and its application to study the effects of treatments to reduce microbial levels in bacitracin powder. J Chromatogr 112:663–672
Verbelen C, Dupres V, Menozzi FD, Raze D, Baulard AR, Hols P, Dufrêne YF (2006) Ethambutol induced alterations in Mycobacterium bovis BCG imaged by atomic force microscopy. FEMS Microbiol Lett 264:192–197
Wu M, Maier E, Benz R, Hancock REW (1999) Mechanism of interaction of different classes of cationic antimicrobial peptides with planar bilayers and with the cytoplasmic membrane of Escherichia coli. Biochemistry 38:7235–7242
Xie CL, Tang HK, Song ZH, Qu SS, Liao YT, Liu HS (1988) Microcalorimetry study of bacterial growth. Thermochim Acta 123:33–41
Yamada S, Sugai M, Komatsuzawa H, Nakashima S, Oshida T, Matsumoto A, Suginaka H (1996) An autolysin ring associated with cell separation of Staphylococcus aureus. J Bacteriol 178:1565–1571
Zhao R, Liu Y, Xie ZX, Shen P, Qu SS (2000) A microcalorimetric method for studying the biological effects of La3+ on Escherichia coli. J Biochem Biophys Methods 46:1–9
Zhou H, Fan T, Zhang XD, Guo Q, Ogawa H (2007) Novel bacteria-templated sonochemical route for the in situ one-step synthesis of ZnS hollow nanostructures. Chem Mater 19:2144–2146
Zhu JC, Liu Y, Wong WK, Zhou B, Yin J (2006) Investigation of antibacterial activity of two kinds of novel Schiff bases on Escherichia coli by microcalorimetry. Chin J Chem 24:1295–1300
Acknowledgments
We gratefully acknowledge the financial support of the National Natural Science Foundation of China (grants 30570015, 20621502, 20873096), the Research Foundation of the Chinese Ministry of Education ([2006]8-IRT0543) and the Natural Science Foundation of Hubei Province (2005ABC002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qi, ZD., Lin, Y., Zhou, B. et al. Characterization of the Mechanism of the Staphylococcus aureus Cell Envelope by Bacitracin and Bacitracin-Metal Ions. J Membrane Biol 225, 27–37 (2008). https://doi.org/10.1007/s00232-008-9130-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-008-9130-8